Advanced Pd-Catalyzed Synthesis of High-Optical-Activity Axial Chiral Allene Compounds for Commercial Scale-up
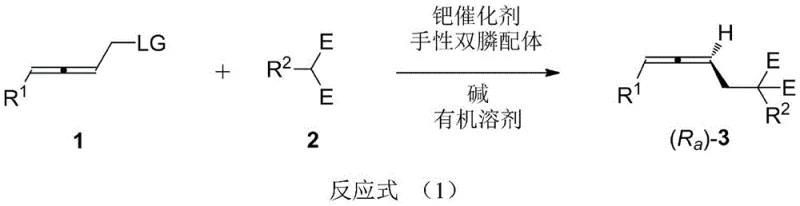
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral building blocks essential for modern drug discovery and agrochemical development. A significant breakthrough in this field is documented in Chinese Patent CN108976123B, which discloses a robust and highly stereoselective method for constructing axial chiral allene compounds. Unlike traditional approaches that often struggle with substrate limitations or require harsh conditions, this invention utilizes a palladium-catalyzed system combined with specialized chiral diphosphine ligands to achieve exceptional enantioselectivity. The technology addresses a critical gap in fine chemical manufacturing by providing a direct route to 1,3-disubstituted axial chiral allenes, which are pivotal scaffolds in numerous bioactive molecules. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages and scalability of this patent is crucial for optimizing supply chains and reducing development timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,3-disubstituted axial chiral allenes has relied heavily on copper-catalyzed asymmetric allenylation of terminal alkynes or reactions involving exo-2,3-dienol derivatives. While these methods have contributed to the field, they suffer from inherent inefficiencies that hinder large-scale commercial adoption. For instance, copper-catalyzed pathways frequently necessitate the addition of at least equivalent amounts of chiral agents, such as diphenylprolinol, which drastically increases raw material costs and complicates downstream purification processes. Furthermore, existing palladium-catalyzed methods reported in literature often require the introduction of sterically bulky groups on the substrate or nucleophile to enforce stereocontrol. This structural constraint severely limits the scope of accessible compounds, making it difficult to synthesize allenes containing valuable, smaller functional groups that are often required for subsequent derivatization into complex drug candidates. These limitations create bottlenecks in the manufacturing of high-purity OLED material or pharmaceutical precursors where structural diversity is paramount.
The Novel Approach
The methodology outlined in patent CN108976123B represents a paradigm shift by eliminating the need for bulky steric groups while maintaining superior stereocontrol. By employing a specific combination of a palladium catalyst, a chiral diphosphine ligand, and a base in an organic solvent, the reaction achieves high stereoselectivity directly from 2,3-allenyl functional group compounds and nucleophiles. This approach is characterized by its operational simplicity and mild reaction conditions, typically proceeding at temperatures as low as 5°C. The versatility of this system is evidenced by its compatibility with a wide range of electron-withdrawing groups, including esters, sulfones, and cyano groups, allowing for the synthesis of diverse allene architectures without the need for specialized, hard-to-source starting materials. This flexibility significantly enhances the feasibility of cost reduction in electronic chemical manufacturing and pharmaceutical intermediate production by streamlining the synthetic route.
Mechanistic Insights into Palladium-Catalyzed Asymmetric Allylic Alkylation
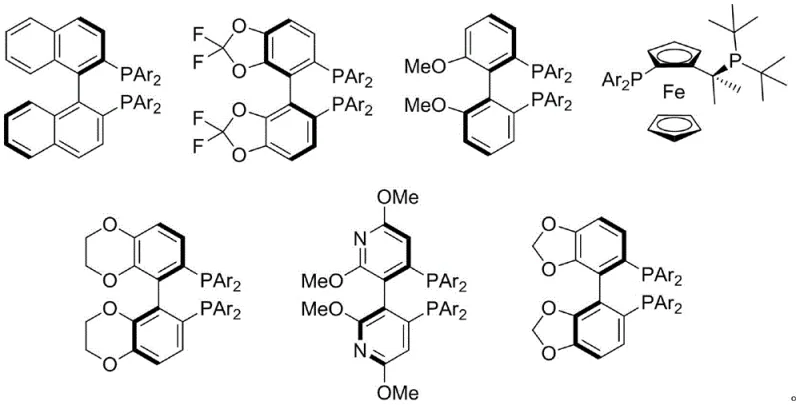
The core of this technological advancement lies in the precise interplay between the palladium catalyst and the chiral environment created by the diphosphine ligand. The reaction likely proceeds through a standard palladium(0)/palladium(II) catalytic cycle, where the oxidative addition of the 2,3-allenyl substrate generates a pi-allyl palladium intermediate. The chirality is induced during the nucleophilic attack on this intermediate, which is tightly controlled by the steric and electronic properties of the ligand. The patent highlights the use of ligands such as DTBM-SEGPHOS and its derivatives, which possess a rigid backbone and bulky substituents that effectively differentiate the enantiotopic faces of the allyl system. This ensures that the nucleophile attacks from a specific trajectory, resulting in the formation of the desired axial chirality with high fidelity. The ability to tune the ligand structure allows chemists to optimize the enantiomeric excess for specific substrates, achieving values consistently between 90% and 96% ee.
Impurity control is another critical aspect where this mechanism excels. The high regioselectivity and stereoselectivity minimize the formation of branched byproducts or racemic mixtures, which are common issues in less optimized catalytic systems. The use of mild bases like potassium carbonate further suppresses side reactions such as elimination or isomerization that could degrade the allene functionality. For supply chain heads, this means a cleaner crude product profile, which translates to simplified purification steps and higher overall yields. The robustness of the catalytic system against various functional groups ensures that the process remains stable even when scaling up, reducing the risk of batch-to-batch variability that often plagues complex chiral syntheses.
How to Synthesize Axial Chiral Allene Compounds Efficiently
The synthesis protocol described in the patent offers a straightforward pathway for producing these valuable intermediates in a laboratory or pilot plant setting. The process involves the sequential addition of reagents under inert atmosphere to prevent catalyst deactivation, followed by a controlled temperature ramp to initiate the reaction. The simplicity of the workup procedure, which typically involves filtration through a short silica plug and standard column chromatography, makes it highly attractive for rapid process development. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- In a glovebox, sequentially add the chiral diphosphine ligand and base (e.g., K2CO3) into a dry reaction tube, then remove the tube and add the palladium catalyst, nucleophilic reagent, and organic solvent under nitrogen protection.
- Stir the mixture at room temperature for 30 minutes, then cool the reaction system to the set temperature (e.g., 5°C) and stir for an additional 10 minutes to ensure thermal equilibrium.
- Add the 2,3-allenyl functional group compound dissolved in solvent to the reaction mixture, stir until completion monitored by TLC, then filter through silica gel and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the significant simplification of the supply chain for chiral building blocks. By utilizing readily available starting materials and avoiding the need for stoichiometric chiral auxiliaries, the overall cost of goods sold (COGS) can be drastically reduced. This is particularly relevant for the commercial scale-up of complex polymer additives or agrochemical intermediates where margin pressures are high. The elimination of expensive chiral reagents not only lowers direct material costs but also reduces the waste disposal burden associated with their removal, contributing to a more sustainable and compliant manufacturing process.
- Cost Reduction in Manufacturing: The catalytic nature of the process means that expensive chiral information is introduced via a ligand used in small molar percentages rather than as a stoichiometric reagent. This fundamentally alters the cost structure, allowing for substantial cost savings compared to traditional resolution methods or stoichiometric chiral pool syntheses. Furthermore, the high yields and selectivity reduce the loss of valuable raw materials, ensuring that every kilogram of input translates efficiently into saleable product. The mild reaction conditions also imply lower energy consumption for heating or cooling, adding another layer of operational cost efficiency.
- Enhanced Supply Chain Reliability: The broad substrate scope of this method means that manufacturers are not locked into sourcing niche, bulky precursors that may have volatile availability. The ability to use simple alkyl chains or common functionalized groups as substrates ensures a stable and continuous supply of raw materials. This reliability is critical for maintaining production schedules for high-purity pharmaceutical intermediates, preventing delays that could impact downstream drug formulation timelines. The robustness of the reaction also minimizes the risk of batch failures, ensuring consistent delivery performance to customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents like THF and inorganic bases that are easy to handle on a multi-kilogram scale. The high selectivity reduces the generation of hazardous waste streams associated with separating difficult impurities, aligning with increasingly stringent environmental regulations. This ease of scale-up facilitates the transition from gram-scale R&D to ton-scale commercial production without the need for extensive process re-engineering, thereby accelerating time-to-market for new products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral allene synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for decision-makers evaluating this route for their own production needs.
Q: What are the advantages of this Pd-catalyzed method over traditional Cu-catalyzed allenylation?
A: Unlike traditional Cu-catalyzed methods that often require stoichiometric amounts of chiral agents or bulky substituents to achieve selectivity, this Pd-catalyzed protocol operates under mild conditions with catalytic amounts of chiral ligands, offering broader substrate versatility and excellent stereoselectivity (90-96% ee) without structural limitations.
Q: What is the typical enantiomeric excess (ee) achieved with this synthesis method?
A: The method consistently delivers high optical activity, with experimental data showing enantiomeric excess values ranging from 90% to 96% ee across various substrates, including the first high-enantioselective synthesis of (R)-traumatic lactone at 98% ee.
Q: Can this method be applied to the synthesis of natural products?
A: Yes, the resulting high-optical-activity allene products serve as versatile intermediates for constructing chiral gamma-allenoic acids, esters, and gamma-butyrolactones, successfully demonstrated by the total synthesis of the natural product (R)-traumatic lactone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Allene Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN108976123B for the future of fine chemical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest standards required by global regulatory bodies. We understand that the successful commercialization of chiral intermediates requires not just chemical expertise but also a deep understanding of supply chain dynamics and cost optimization strategies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Whether you need specific COA data for audit purposes or detailed route feasibility assessments to evaluate the integration of this chemistry into your existing portfolio, we are here to support your goals. Contact us today to discuss how we can become your trusted partner in delivering high-value chiral solutions.
