Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates Production
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly those serving as core scaffolds for bioactive molecules. Patent CN112239456B introduces a significant advancement in this domain by disclosing a highly efficient preparation method for substituted 2,3-dihydroquinolone compounds. This specific class of heterocycles is critically important, as evidenced by their presence in potent antitumor agents and analgesic drugs, such as the structures depicted in the biological activity overview. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary starting materials. By replacing hazardous carbon monoxide gas with a solid surrogate, this innovation addresses critical safety and scalability concerns inherent in traditional carbonylation processes, positioning it as a superior choice for the manufacturing of high-value pharmaceutical intermediates.
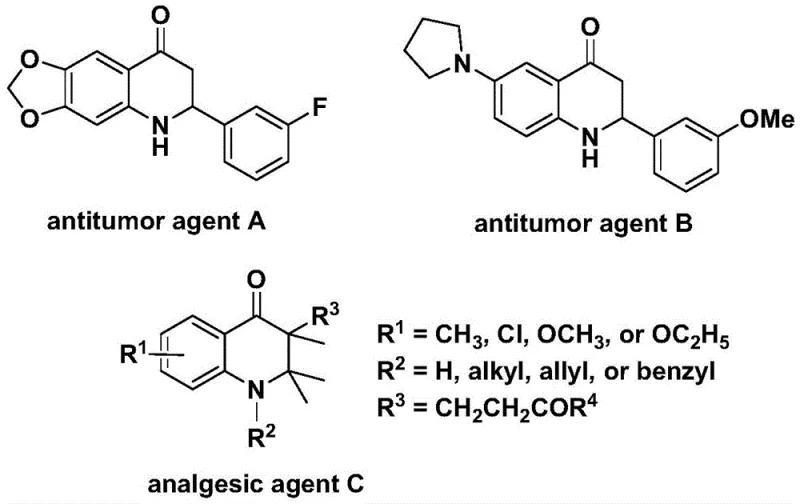
The strategic value of this synthesis lies in its ability to access complex molecular architectures through a convergent approach. The 2,3-dihydroquinolone skeleton is a privileged structure in medicinal chemistry, yet its construction often requires multi-step sequences or harsh conditions. The method described in CN112239456B streamlines this process into a single catalytic cycle that builds the six-membered lactam ring while simultaneously installing diverse substituents at the 2- and 3-positions. This capability allows medicinal chemists and process engineers to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies or to optimize the production of specific drug candidates. For a reliable pharmaceutical intermediate supplier, mastering such versatile C-H functionalization and carbonylation techniques is essential for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has relied heavily on the direct use of carbon monoxide gas. While effective in laboratory settings, the use of gaseous CO presents severe logistical and safety challenges for commercial manufacturing. High-pressure reactors are mandatory to maintain sufficient CO concentration, which drastically increases capital expenditure and operational complexity. Furthermore, carbon monoxide is extremely toxic, requiring rigorous leak detection systems and specialized ventilation infrastructure to protect personnel. Beyond safety, traditional methods often suffer from poor atom economy or require pre-functionalized substrates that are expensive and difficult to source. Many existing protocols also struggle with functional group tolerance, leading to complex impurity profiles that necessitate costly and yield-reducing purification steps. These limitations create bottlenecks in the supply chain, increasing lead times and reducing the overall reliability of production for key API intermediates.
The Novel Approach
The methodology outlined in patent CN112239456B offers a transformative solution by employing 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide substitute. This innovative substitution fundamentally changes the risk profile of the reaction, allowing it to proceed in standard glassware or stainless steel reactors without the need for high-pressure gas handling. The reaction utilizes a palladium catalyst system, specifically bis(acetylacetone)palladium paired with a dppp ligand, which facilitates the oxidative addition into the carbon-iodine bond of the aniline derivative. This is followed by the insertion of CO generated in situ from the TFBen, and subsequent coordination and insertion of the olefin. The result is a clean, efficient cyclization that constructs the target 2,3-dihydroquinolone core with high regioselectivity. This approach not only enhances safety but also simplifies the workflow, making it ideally suited for cost reduction in pharmaceutical intermediate manufacturing.
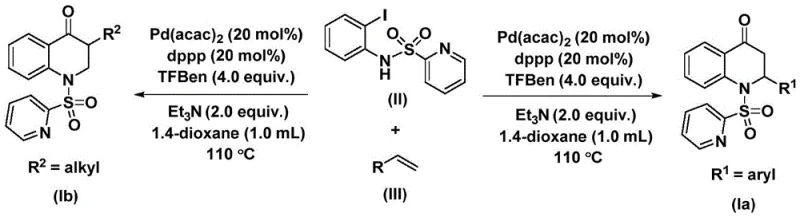
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
Understanding the catalytic cycle is paramount for R&D directors aiming to optimize this process for large-scale production. The reaction initiates with the generation of the active palladium(0) species from the Pd(acac)2 precursor. This active catalyst undergoes oxidative addition into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, forming a stable aryl-palladium(II) intermediate. This step is crucial as it activates the aromatic ring for subsequent transformations. Following this, the carbon monoxide molecule, released thermally from the TFBen additive, inserts into the palladium-carbon bond. This insertion forms an acyl-palladium intermediate, effectively installing the carbonyl functionality required for the lactam ring. The precision of this step ensures that the carbonyl group is positioned correctly relative to the nitrogen atom, setting the stage for ring closure.
The final stages of the catalytic cycle involve the interaction with the olefin partner. The alkene coordinates to the palladium center and inserts into the acyl-palladium bond, creating a new carbon-carbon bond and extending the carbon chain. This forms an alkyl-palladium intermediate which is poised for the final ring-closing event. Reductive elimination then occurs, releasing the substituted 2,3-dihydroquinolone product and regenerating the palladium(0) catalyst to continue the cycle. This mechanism explains the high substrate compatibility observed in the patent data; the mild nature of the palladium cycle tolerates various electronic environments on both the aryl iodide and the olefin. Whether the substituent is an electron-donating methyl group or an electron-withdrawing chlorine atom, the catalytic turnover remains efficient, ensuring consistent yields across a broad range of derivatives.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it accessible for both laboratory research and pilot plant operations. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in an aprotic organic solvent such as dioxane. The mixture is then heated to a moderate temperature of 110°C for a duration of 48 hours. This thermal energy drives the decomposition of the CO surrogate and sustains the catalytic turnover. Upon completion, the workup is straightforward, involving filtration to remove palladium black and inorganic salts, followed by standard chromatographic purification. The detailed standardized synthesis steps see the guide below.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, TFBen (CO source), N-pyridine sulfonyl-o-iodoaniline, and olefin in dioxane solvent.
- Heat the reaction mixture to 110°C and stir for 48 hours under inert atmosphere to facilitate the carbonylation cyclization.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible operational improvements and risk mitigation. The shift from gaseous CO to a solid surrogate eliminates the need for specialized high-pressure infrastructure, thereby reducing capital investment and maintenance costs associated with reactor systems. This simplification allows for greater flexibility in manufacturing sites, as the reaction can be performed in facilities that may not be equipped for handling toxic gases. Furthermore, the use of commercially available starting materials, such as readily sourced olefins and aniline derivatives, ensures a stable and continuous supply chain. The robustness of the reaction conditions means that batch-to-batch variability is minimized, leading to more predictable production schedules and reduced downtime.
- Cost Reduction in Manufacturing: The elimination of high-pressure gas equipment and the associated safety compliance measures results in significant overhead savings. Additionally, the high efficiency of the palladium catalyst system reduces the amount of raw material waste, improving the overall mass balance of the process. The ability to use standard solvents like dioxane, which are easily recovered and recycled, further contributes to lower operational expenditures. By streamlining the synthetic route into a single pot transformation, labor costs and processing time are also optimized compared to multi-step alternatives.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents rather than compressed gases mitigates the risks associated with transportation and storage hazards. This stability ensures that raw material inventory can be maintained safely on-site without the regulatory burdens of hazardous gas cylinders. The broad substrate scope allows for the sourcing of diverse olefin and aniline precursors from multiple vendors, preventing single-source bottlenecks. This flexibility is critical for maintaining continuity of supply for key pharmaceutical intermediates in a volatile global market.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, having been demonstrated to work effectively from milligram to gram scales in the patent examples. The absence of toxic gas emissions aligns with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing lines. The clean reaction profile reduces the burden on waste treatment facilities, as the effluent contains fewer hazardous byproducts. This environmental compatibility supports sustainable manufacturing goals and enhances the corporate social responsibility profile of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of using TFBen over carbon monoxide gas in this synthesis?
A: Using 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the need for high-pressure gas equipment and specialized safety protocols required for toxic CO gas, significantly enhancing operational safety and simplifying the reaction setup for industrial scale-up.
Q: What types of substituents are compatible with this palladium-catalyzed protocol?
A: The method demonstrates excellent substrate compatibility, tolerating various functional groups on the aryl ring including methyl, tert-butyl, methoxy, fluorine, and chlorine at ortho, meta, or para positions, as well as linear, branched alkyl, and silyl groups on the olefin component.
Q: How does this method impact the purity profile of the final heterocyclic product?
A: The use of a specific palladium catalyst system with dppp ligand and the controlled release of CO from TFBen minimizes side reactions such as homocoupling or over-carbonylation, resulting in cleaner crude reaction mixtures that facilitate easier purification and higher final purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and securing the supply of essential medicines. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like the one in CN112239456B can be successfully translated into industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle complex organometallic reactions safely and efficiently, leveraging technologies such as solid CO surrogates to maximize safety and yield.
We invite you to collaborate with us to leverage this innovative synthesis for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you optimize your supply chain and bring your life-saving therapies to market faster and more cost-effectively.
