Advanced Metal-Free Synthesis of Styrylquinoline Derivatives for Commercial Scale-Up
The chemical manufacturing landscape is undergoing a significant transformation driven by the urgent need for sustainable and economically viable synthetic routes, as exemplified by the groundbreaking technology disclosed in patent CN114436954A. This specific intellectual property introduces a highly efficient method for the cross-deaminoalkenylation of methyl-substituted nitrogen heterocyclic compounds and benzylamines, addressing critical bottlenecks in the production of complex pharmaceutical intermediates. Unlike traditional approaches that rely heavily on scarce and expensive transition metals, this innovation leverages a readily available iodine and organic acid catalytic system to achieve remarkable conversion rates. For R&D Directors and Procurement Managers alike, this represents a pivotal shift towards processes that offer both high atom economy and simplified operational workflows without compromising on the structural integrity of the final molecule. The ability to synthesize valuable styrylquinoline derivatives under such accessible conditions opens new avenues for scaling up production while maintaining rigorous quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of C(sp3)-H bonds in nitrogen heterocycles has been dominated by methodologies requiring sophisticated transition metal catalysts such as Manganese, Platinum, or Nickel complexes, which introduce substantial complexity to the manufacturing process. As illustrated in prior art examples, reactions often necessitate harsh conditions, including temperatures exceeding 140°C and the use of strong bases like potassium tert-butoxide, which can compromise the stability of sensitive functional groups on the substrate. Furthermore, the reliance on heavy metal catalysts creates a significant burden on downstream processing, as extensive purification steps are mandatory to reduce metal residues to parts-per-million levels acceptable for pharmaceutical applications. These legacy methods not only inflate the cost of goods sold due to expensive catalyst loading but also generate hazardous waste streams that require specialized treatment, thereby increasing the environmental footprint and operational risk for large-scale facilities.
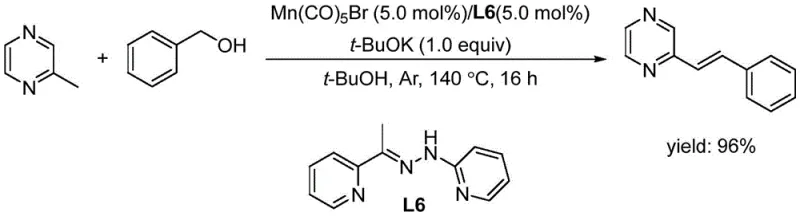
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the novel approach detailed in the patent utilizes a metal-free catalytic system comprising molecular iodine and trifluoroacetic acid in a toluene solvent medium. This methodology operates under significantly milder thermal conditions, typically between 80°C and 110°C, which reduces energy consumption and minimizes the thermal degradation of reactants. The elimination of transition metals fundamentally alters the impurity profile of the reaction, removing the need for costly metal scavenging resins or complex chromatographic separations dedicated solely to metal removal. For supply chain leaders, this translates to a more robust and predictable manufacturing timeline, as the availability of iodine and organic acids is far more stable than that of specialized organometallic complexes. The operational simplicity allows for easier technology transfer from laboratory to pilot plant, accelerating the time to market for new drug candidates relying on these key intermediate structures.
Mechanistic Insights into Iodine-Catalyzed Cross-Deaminoalkenylation
The mechanistic pathway of this transformation involves a radical-mediated process initiated by the interaction of molecular iodine with the benzylamine substrate under an oxygen atmosphere. This oxidative condition facilitates the generation of reactive imine intermediates in situ, which subsequently undergo coupling with the methyl-substituted nitrogen heterocycle at the activated C(sp3)-H position. The presence of the organic acid is crucial for protonating intermediate species and stabilizing the transition state, ensuring high regioselectivity towards the desired cross-coupled product rather than homocoupling side reactions. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters, as the balance between oxidant concentration and acid strength directly influences the reaction kinetics and overall yield. The specificity of this system allows for a broad tolerance of substituents on both the heterocyclic and benzylamine rings, enabling the synthesis of a diverse library of analogues without the need for protecting group strategies that add synthetic steps.
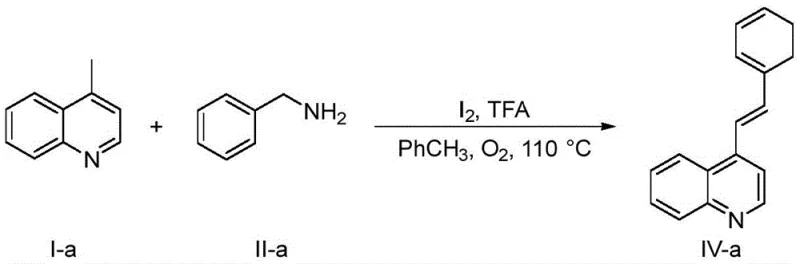
Controlling the impurity profile in this metal-free system is inherently more straightforward compared to metal-catalyzed variants, primarily because the catalyst itself is volatile or easily washed away during aqueous workup. The primary side reactions involve the self-dehydroalkenylation of the nitrogen heterocycle, but the patent data indicates that this pathway is minimized under the optimized toluene solvent conditions, keeping side product formation below critical thresholds. For Quality Assurance teams, this means that the final active pharmaceutical ingredient (API) intermediate exhibits a cleaner spectral profile with fewer unknown peaks, simplifying the validation process for regulatory filings. The absence of heavy metals also alleviates concerns regarding genotoxic impurities, which are a major focus in modern drug safety assessments, thereby reducing the analytical burden on QC laboratories. This inherent purity advantage positions the technology as a preferred route for GMP manufacturing environments where consistency and cleanliness are paramount.
How to Synthesize 4-Styrylquinoline Efficiently
To implement this synthesis effectively, process engineers must adhere to specific stoichiometric ratios and environmental controls outlined in the patent embodiments to maximize yield and reproducibility. The procedure involves mixing the methyl-substituted nitrogen heterocycle and benzylamine with precise amounts of iodine and trifluoroacetic acid in toluene, followed by heating under an oxygen atmosphere for an extended period. Detailed standardized synthesis steps are provided in the guide below to ensure operators can replicate the high yields observed in the experimental data consistently across different batch sizes. Proper attention to the quenching phase using sodium thiosulfate is essential to neutralize excess iodine before extraction, ensuring the safety and stability of the isolated product. Adhering to these protocols guarantees that the commercial output meets the stringent specifications required for downstream pharmaceutical applications.
- Mix methyl-substituted nitrogen heterocycle, benzylamine, iodine, and organic acid in toluene solvent under an oxygen atmosphere.
- Heat the reaction mixture to a temperature range of 80-110°C and maintain stirring for 18 to 24 hours to ensure complete conversion.
- Quench the reaction with water, remove residual iodine with sodium thiosulfate, and purify the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free alkenylation technology offers profound advantages for procurement strategies and supply chain resilience in the fine chemical sector. The substitution of expensive transition metal catalysts with commodity chemicals like iodine drastically reduces the raw material cost base, allowing for more competitive pricing models in long-term supply agreements. Additionally, the simplified workup procedure reduces the consumption of solvents and purification media, leading to substantial cost savings in waste management and utility usage over the lifecycle of the product. For supply chain heads, the reliance on widely available reagents mitigates the risk of disruption caused by geopolitical issues affecting the supply of rare earth metals or specialized ligands. This stability ensures continuous production capability, which is critical for meeting the just-in-time delivery requirements of multinational pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a significant cost driver from the bill of materials, while the milder reaction conditions lower energy expenditures associated with heating and cooling cycles. Furthermore, the reduced need for complex purification steps to remove metal residues decreases the consumption of chromatography silica and specialized scavenging agents, contributing to a leaner manufacturing cost structure. These cumulative efficiencies allow manufacturers to offer more attractive pricing without sacrificing margin, enhancing competitiveness in the global marketplace for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing iodine and common organic acids is far less volatile than procuring specialized organometallic complexes, which often have limited suppliers and long lead times. This accessibility ensures that production schedules are not held hostage by raw material shortages, providing a reliable flow of goods to customers. The robustness of the reaction also means that variations in reagent quality have less impact on the final outcome, reducing the rate of batch failures and ensuring consistent supply continuity even during periods of market fluctuation.
- Scalability and Environmental Compliance: The process is inherently scalable due to its use of common solvents and standard reactor configurations, facilitating a smooth transition from kilogram to ton-scale production without significant re-engineering. Environmentally, the absence of heavy metal waste simplifies compliance with increasingly strict environmental regulations, reducing the liability and cost associated with hazardous waste disposal. This green chemistry profile aligns with the sustainability goals of major pharmaceutical companies, making the supplier a more attractive partner for eco-conscious procurement initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cross-deaminoalkenylation technology in industrial settings. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for potential partners. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. The responses cover aspects ranging from catalyst recovery to substrate scope limitations, ensuring a comprehensive overview of the technology's capabilities.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method utilizes molecular iodine and organic acids as catalysts, completely eliminating the need for costly transition metals like Manganese or Platinum, which significantly simplifies downstream purification and reduces raw material costs.
Q: What are the typical reaction conditions for this cross-deaminoalkenylation?
A: The reaction operates under mild thermal conditions ranging from 80°C to 110°C in a toluene solvent system with an oxygen atmosphere, offering a safer and more energy-efficient profile compared to traditional high-temperature protocols.
Q: How does this method impact impurity profiles in pharmaceutical intermediates?
A: By avoiding heavy metal catalysts, the process inherently prevents metal residue contamination, resulting in a cleaner crude product that requires less aggressive purification steps to meet stringent pharmaceutical purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Styrylquinoline Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this advanced metal-free synthesis for the commercial production of high-value pharmaceutical intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patent can be realized at an industrial level with consistent quality. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch meets the exacting standards required for drug substance manufacturing. Our commitment to process excellence means we can deliver complex molecules like 4-styrylquinoline derivatives with the reliability and transparency that global supply chains demand.
We invite you to engage with our technical procurement team to discuss how this innovative route can optimize your specific project requirements and reduce overall development timelines. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique chemical needs, ensuring a partnership built on scientific rigor and commercial value.
