Advanced FeCl3-Catalyzed Cyclization Strategy for High-Purity Coumestan Intermediates Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access bioactive scaffolds efficiently, and the synthesis of Coumestan derivatives represents a critical area of development due to their potent biological activities ranging from phytoestrogenic to hepatoprotective effects. Patent CN101817830B introduces a groundbreaking approach that leverages anhydrous iron trihalides, specifically FeCl3 or FeBr3, to facilitate the direct oxidative cyclization of 3-aryl-4-hydroxycoumarin compounds into the coveted coumestan core structure. This innovation addresses long-standing challenges in heterocyclic chemistry by enabling the direct formation of a carbon-oxygen bond between the aromatic ring's sp2 carbon and the side-chain oxygen atom without the need for prior halogenation. For R&D directors and process chemists, this represents a paradigm shift from multi-step, precious-metal-dependent sequences to a more atom-economical and operationally simple protocol that utilizes abundant earth-abundant metals. The strategic importance of this technology lies in its ability to streamline the production of high-value intermediates used in drug discovery and agrochemical applications, thereby enhancing the overall efficiency of the supply chain for these specialized molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the coumestan skeleton has relied heavily on transition metal-catalyzed cross-coupling reactions, particularly those involving palladium, which necessitate the pre-functionalization of the aromatic ring with halogen atoms such as bromine or iodine. This traditional requirement introduces significant inefficiencies, including additional synthetic steps for halogenation, the use of expensive and often toxic palladium catalysts, and the subsequent need for rigorous purification to remove trace heavy metal residues that are strictly regulated in pharmaceutical products. Furthermore, conventional methods often suffer from limited substrate scope and sensitivity to functional groups, requiring harsh conditions or specialized ligands that drive up the cost of goods sold and complicate the manufacturing process. The reliance on precious metals also exposes the supply chain to volatility in metal prices and availability, creating potential bottlenecks for large-scale production campaigns. These cumulative factors result in a process that is not only economically burdensome but also environmentally less sustainable due to the generation of hazardous waste associated with heavy metal usage and complex workup procedures.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a single-electron transfer (SET) oxidation mechanism mediated by inexpensive anhydrous ferric chloride or ferric bromide to achieve direct C-O bond formation. This novel approach bypasses the need for pre-halogenation entirely, allowing the reaction to proceed directly from readily available 3-aryl-4-hydroxycoumarin precursors in a single pot. The reaction conditions are remarkably mild, operating effectively in common aprotic solvents like 1,2-dichloroethane or dichloromethane at temperatures ranging from room temperature to reflux, which significantly reduces energy consumption and operational complexity. By replacing palladium with iron, the process drastically lowers the catalyst cost and eliminates the regulatory burden associated with heavy metal clearance, resulting in a cleaner final product profile. The simplicity of the workup, which involves basic filtration and standard column chromatography, further enhances the practicality of this method for both laboratory-scale optimization and industrial manufacturing, offering a compelling alternative for the reliable coumestan derivatives supplier seeking to optimize their production capabilities.
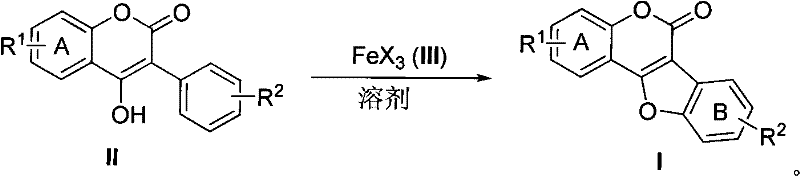
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the unique ability of anhydrous iron(III) species to act as a Lewis acid and a single-electron oxidant simultaneously, facilitating the intramolecular cyclization through a radical or cationic pathway. Upon coordination with the hydroxyl group and the aromatic system of the 3-aryl-4-hydroxycoumarin substrate, the iron center promotes the activation of the inert sp2 C-H bond on the aromatic ring, enabling a direct nucleophilic attack by the oxygen atom to close the furan ring. This mechanism avoids the high-energy barriers typically associated with direct C-H functionalization and proceeds with high regioselectivity to form the 6H-benzofuro[3,2-c][1]benzopyran-6-one core. The use of a stoichiometric or excess amount of the iron salt, typically in a molar ratio of 2.5:1 to 5:1 relative to the substrate, ensures complete conversion and drives the equilibrium towards the desired cyclic product. Understanding this mechanistic nuance is crucial for process chemists aiming to replicate the high yields reported in the patent, as the precise control of water content and solvent dryness is paramount to maintaining the activity of the anhydrous iron catalyst throughout the reaction duration.
From an impurity control perspective, this iron-mediated pathway offers distinct advantages by minimizing the formation of side products commonly seen in palladium-catalyzed homocoupling or dehalogenation reactions. The absence of halogenated intermediates reduces the risk of generating poly-halogenated byproducts or biaryl impurities that are difficult to separate from the final API intermediate. Moreover, the reaction demonstrates excellent tolerance to various substituents on the aromatic rings, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like bromine, without significant degradation of the yield or purity. This robustness suggests that the catalytic cycle is resilient to electronic variations in the substrate, making it a versatile platform for synthesizing a diverse library of coumestan analogs. For quality assurance teams, this translates to a more predictable impurity profile and simplified analytical method development, ensuring that the high-purity coumestan derivatives meet the stringent specifications required for downstream biological testing and clinical applications.
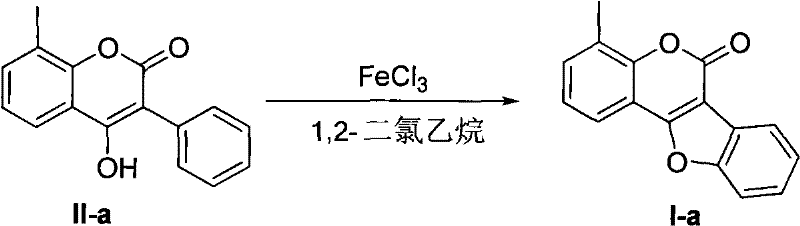
How to Synthesize Coumestan Derivatives Efficiently
The practical implementation of this synthesis route is designed to be accessible to standard organic chemistry laboratories equipped with basic reflux and filtration setups, requiring no specialized high-pressure or inert atmosphere equipment beyond standard drying techniques for solvents. The protocol begins with the dissolution of the 3-aryl-4-hydroxycoumarin starting material in a dry aprotic solvent, followed by the controlled addition of the anhydrous iron trihalide catalyst under stirring to ensure homogeneous mixing and heat dissipation. Reaction monitoring is typically conducted via thin-layer chromatography (TLC) to determine the optimal endpoint, which varies depending on the specific substrate and reaction temperature, ranging from 2 hours at reflux to 12 hours at room temperature. Once the reaction is deemed complete, the mixture is cooled, and the iron salts are removed via filtration through Celite, followed by a straightforward aqueous workup or direct concentration to isolate the crude product. The detailed standardized synthesis steps for specific derivatives, including precise stoichiometric ratios and purification parameters, are outlined in the guide below to assist technical teams in replicating these results accurately.
- Dissolve the 3-aryl-4-hydroxycoumarin precursor in dry 1,2-dichloroethane and add anhydrous FeCl3 at room temperature.
- Heat the reaction mixture to reflux for approximately 6 hours until conversion is complete as monitored by TLC.
- Filter off solids, wash with solvent, concentrate the filtrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iron-catalyzed methodology presents a transformative opportunity to reduce the total cost of ownership for coumestan-based projects while mitigating supply risks associated with critical raw materials. The substitution of expensive palladium catalysts with commodity-grade iron salts results in a drastic reduction in direct material costs, which is particularly impactful when scaling production from kilograms to metric tons. Furthermore, the elimination of the halogenation step shortens the overall synthetic timeline, reducing labor costs, solvent consumption, and reactor occupancy time, all of which contribute to substantial cost savings in pharmaceutical intermediates manufacturing. The simplicity of the purification process also means lower consumption of silica gel and eluents, further driving down the variable costs per batch. These economic benefits are compounded by the increased reliability of the supply chain, as iron salts are globally available and not subject to the same geopolitical constraints or price volatility as precious metals, ensuring consistent production schedules and delivery timelines for clients.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive iron reagents fundamentally alters the cost structure of the synthesis, removing the need for costly ligand systems and extensive metal scavenging processes that are mandatory for palladium routes. This shift allows manufacturers to allocate resources more efficiently, potentially passing savings on to customers or reinvesting in process optimization and capacity expansion. Additionally, the higher atom economy of the direct cyclization reduces the volume of chemical waste generated per unit of product, lowering waste disposal fees and environmental compliance costs. The cumulative effect of these factors is a significantly more competitive pricing model for the final coumestan derivatives, enhancing the market position of suppliers who adopt this technology.
- Enhanced Supply Chain Reliability: By relying on widely available and stable reagents like anhydrous ferric chloride and common chlorinated solvents, the manufacturing process becomes far less susceptible to disruptions caused by raw material shortages. This stability is crucial for maintaining continuous production flows, especially for long-term contracts where delivery consistency is a key performance indicator. The robustness of the reaction conditions also means that the process can be easily transferred between different manufacturing sites or scaled up in existing facilities without requiring major capital investment in new equipment. This flexibility ensures that the reliable coumestan derivatives supplier can respond swiftly to fluctuations in market demand, securing their role as a strategic partner for global pharmaceutical and agrochemical companies.
- Scalability and Environmental Compliance: The operational simplicity of the method, characterized by ambient pressure and moderate temperatures, makes it inherently safer and easier to scale compared to high-energy or high-pressure alternatives. The reduced use of toxic heavy metals aligns with increasingly stringent environmental regulations and corporate sustainability goals, facilitating smoother regulatory approvals and audits. The straightforward workup procedure minimizes the generation of complex waste streams, simplifying effluent treatment and reducing the environmental footprint of the manufacturing process. These attributes make the technology ideal for the commercial scale-up of complex pharmaceutical intermediates, ensuring that production can grow in tandem with clinical or commercial demand without encountering significant technical or regulatory hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity on its practical application and benefits for industry stakeholders. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this route for their own portfolios. Understanding these nuances is essential for leveraging the full potential of the iron-catalyzed cyclization method in a commercial setting.
Q: What are the primary advantages of this FeCl3 method over traditional Pd-catalyzed routes?
A: This method eliminates the need for pre-halogenation of the aromatic ring and avoids expensive palladium catalysts, significantly reducing raw material costs and simplifying the purification process by removing heavy metal residues.
Q: What solvents are compatible with this oxidative cyclization process?
A: The patent specifies the use of dry aprotic solvents, specifically anhydrous dichloromethane or 1,2-dichloroethane, which facilitate the single-electron transfer mechanism required for the C-O bond formation.
Q: Is this synthesis scalable for industrial production of coumestan intermediates?
A: Yes, the process utilizes inexpensive iron salts and standard organic solvents with simple workup procedures like filtration and column chromatography, making it highly suitable for commercial scale-up without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumestan Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the FeCl3-catalyzed cyclization in delivering high-quality intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of coumestan derivatives meets the highest standards of quality and consistency required by our partners. By integrating this advanced technology into our production capabilities, we can offer superior cost-performance ratios and faster turnaround times, positioning ourselves as a preferred partner for complex organic synthesis projects.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements and cost structures. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this iron-catalyzed method for your supply chain. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your needs, helping you make informed decisions that drive value and efficiency in your drug development pipeline. Contact us now to explore a partnership that combines cutting-edge chemistry with reliable supply chain execution.
