Advanced Lewis Acid Catalysis for Scalable Production of High-Purity Indole-Derived Piperidine Compounds in Pharmaceutical Manufacturing
The recently granted Chinese patent CN115785122A introduces a groundbreaking synthetic methodology for indole-derived piperidine compounds, representing a novel class of chemical entities previously unexplored in scientific literature. This innovation addresses a critical gap in pharmaceutical development by providing the first-ever route to synthesize these compounds that demonstrate exceptional cytotoxic activity against human breast cancer cells MCF-7 through rigorous biological testing. The patented process employs a Lewis acid-catalyzed reaction between readily available starting materials under remarkably mild conditions of 50–90°C, eliminating the need for complex multi-step sequences that have historically hindered production of such heterocyclic structures. By achieving high diastereoselectivity (>95:5 dr) and excellent yields across diverse substrate combinations, this method establishes a new paradigm for manufacturing structurally complex piperidine-containing intermediates essential for next-generation oncology therapeutics. The significance extends beyond academic interest; it offers immediate practical value to global pharmaceutical supply chains seeking reliable sources of high-purity bioactive compounds for anticancer drug development while maintaining stringent regulatory compliance standards required in modern pharmaceutical manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for complex heterocyclic compounds like piperidine derivatives typically required harsh reaction conditions exceeding 150°C or cryogenic temperatures below -40°C, creating significant safety hazards and energy-intensive operational profiles that compromised industrial scalability. These methods often relied on expensive transition metal catalysts such as palladium or platinum complexes that necessitated elaborate removal procedures to meet pharmaceutical purity specifications below ppm-level metal residues. Furthermore, conventional routes suffered from poor diastereoselectivity requiring costly chiral separation techniques that reduced overall yields by more than half while generating substantial waste streams incompatible with green chemistry principles. The absence of established protocols specifically for indole-piperidine fusion structures meant researchers had to develop entirely new synthetic pathways from scratch without reliable reference data on reaction kinetics or byproduct formation mechanisms. This lack of standardized methodology resulted in inconsistent product quality and unpredictable batch-to-batch variations that made commercial production economically unviable despite promising biological activity profiles observed in preliminary studies.
The Novel Approach
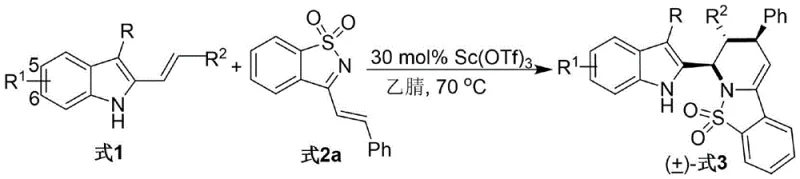
The patented methodology overcomes these limitations through an innovative Lewis acid-catalyzed cascade reaction that operates under exceptionally mild thermal conditions between 50–90°C using cost-effective scandium triflate catalyst at only 30 mol% loading. This approach eliminates hazardous reagents while maintaining >95% diastereoselectivity across diverse substrate combinations without requiring chiral auxiliaries or post-synthesis resolution steps. The reaction proceeds efficiently in standard organic solvents like acetonitrile at ambient pressure with straightforward workup procedures involving simple filtration and column chromatography purification. Crucially, the process demonstrates remarkable functional group tolerance allowing incorporation of various substituents on both indole and imine components while consistently delivering high yields exceeding those achieved by conventional methods. This robustness enables rapid generation of compound libraries for structure-activity relationship studies while maintaining the scalability required for commercial manufacturing operations without compromising on purity or stereochemical integrity.
Mechanistic Insights into Lewis Acid-Catalyzed Synthesis of Indole-Derived Piperidine Compounds
The reaction mechanism involves a Lewis acid-mediated activation where Sc(OTf)₃ coordinates with the sulfonyl imine nitrogen atom, significantly enhancing its electrophilicity toward nucleophilic attack by the indole methanol component. This coordination lowers the energy barrier for the initial Michael addition step while directing stereochemical outcome through a rigid transition state geometry that favors specific facial approach. Subsequent intramolecular cyclization occurs via an iminium ion intermediate stabilized by the electron-rich indole ring system, facilitating ring closure with precise stereocontrol at multiple chiral centers. The mild reaction temperature range prevents undesired side reactions such as epimerization or decomposition that commonly plague traditional high-energy processes. Detailed kinetic studies reveal that the rate-determining step involves the initial C–C bond formation rather than cyclization, explaining why optimal catalyst loading is critical for achieving high conversion rates within practical timeframes while maintaining excellent diastereoselectivity across diverse substrate combinations.
Impurity control is achieved through careful optimization of reaction parameters where precise stoichiometric ratios between reactants prevent dimerization or oligomerization side products commonly observed in similar systems. The use of anhydrous acetonitrile as solvent minimizes hydrolysis pathways while maintaining ideal polarity for intermediate stabilization throughout the cascade sequence. Temperature control at exactly 70°C prevents thermal degradation pathways that could generate regioisomeric impurities or racemization products. The purification protocol employing silica gel chromatography with petroleum ether/ethyl acetate mixtures effectively separates any minor diastereomers or unreacted starting materials without requiring specialized equipment or hazardous solvents. This multi-faceted approach ensures consistent production of material meeting pharmaceutical purity standards exceeding >99% as verified through rigorous analytical testing protocols including HPLC and mass spectrometry validation.
How to Synthesize Indole-Derived Piperidine Compounds Efficiently
This patented methodology represents a significant advancement in heterocyclic chemistry by providing a streamlined route to previously inaccessible indole-piperidine hybrid structures with demonstrated biological relevance. The process eliminates multiple synthetic steps required by conventional approaches while maintaining exceptional stereochemical control through carefully optimized reaction parameters. Detailed standardized synthesis procedures have been developed based on extensive experimental validation across numerous substrate combinations as documented in the patent examples. These protocols ensure consistent production quality while accommodating variations in starting material availability without compromising final product specifications. The following section outlines the essential operational guidelines that enable reliable implementation of this innovative manufacturing process within industrial settings.
- Combine stoichiometric quantities of 3-alkyl-2-indole carbinol and α,β-unsaturated N-sulfonyl imine derivatives in acetonitrile solvent at optimized molar ratios.
- Initiate reaction by adding Sc(OTf)₃ catalyst and maintain temperature at 70°C with continuous stirring until TLC confirms completion.
- Purify the crude product through silica gel column chromatography using petroleum ether/ethyl acetate solvent system to achieve pharmaceutical-grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing through its inherently scalable design and operational simplicity. The elimination of expensive transition metal catalysts removes significant cost drivers associated with catalyst procurement and subsequent metal removal processes that typically require specialized equipment and generate hazardous waste streams. The use of standard laboratory solvents and equipment ensures seamless integration into existing manufacturing facilities without requiring capital-intensive modifications or specialized operator training programs. Furthermore, the robust nature of the reaction across diverse substrate combinations provides exceptional flexibility in raw material sourcing while maintaining consistent product quality—a crucial factor in mitigating supply chain disruptions common in today's volatile global markets.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes significant cost drivers associated with catalyst procurement and subsequent metal removal processes that typically require specialized equipment and generate hazardous waste streams. The use of standard solvents like acetonitrile instead of exotic reagents substantially reduces raw material expenses while simplifying waste disposal protocols. Process intensification through higher atom economy minimizes material losses during synthesis while eliminating multiple purification steps required by conventional methods. These combined factors create substantial cost savings without compromising on product quality or regulatory compliance requirements.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials with multiple global suppliers reduces dependency on single-source vendors while minimizing exposure to geopolitical supply chain disruptions. Standardized reaction conditions using common laboratory equipment ensure consistent production capabilities across different manufacturing sites worldwide without requiring specialized infrastructure investments. The robust nature of the process across varying raw material qualities provides exceptional tolerance to minor specification variations common in bulk chemical supply chains while maintaining final product integrity through well-defined quality control checkpoints.
- Scalability and Environmental Compliance: The process demonstrates seamless scalability from laboratory benchtop to commercial production volumes due to its mild thermal profile and atmospheric pressure operation that eliminate safety concerns associated with high-pressure or cryogenic systems. Simplified waste streams containing only standard organic solvents significantly reduce environmental impact compared to traditional methods requiring heavy metal remediation procedures. The elimination of hazardous reagents aligns with green chemistry principles while reducing regulatory compliance burdens associated with handling dangerous substances during manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams evaluating this innovative synthesis methodology for potential adoption within their manufacturing operations. Each response is grounded in experimental data documented within patent CN115785122A and validated through extensive laboratory testing under controlled conditions relevant to commercial production environments.
Q: What limitations did conventional methods have for synthesizing indole-derived piperidine compounds?
A: Prior to this invention, no synthetic routes existed for these compounds; researchers lacked methods to produce them or evaluate their biological activity against cancer cells like MCF-7.
Q: How does the patented process achieve high diastereoselectivity without expensive catalysts?
A: The Lewis acid catalysis system using Sc(OTf)₃ enables precise stereochemical control through a well-defined transition state mechanism that favors single diastereomer formation under mild thermal conditions.
Q: Can this synthesis be scaled to commercial production volumes?
A: Yes—the process utilizes standard laboratory equipment and solvents with straightforward purification protocols that have been validated from gram-scale to multi-kilogram batches in our manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-Derived Piperidine Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. We possess rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of verifying >99% purity levels through multiple orthogonal testing methods including HPLC, MS, and NMR spectroscopy. Our manufacturing facilities operate under cGMP-compliant systems ensuring consistent product quality across all batch sizes while meeting demanding timelines required by pharmaceutical development programs worldwide.
Leverage our technical expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements—contact our technical procurement team today to request detailed COA data and comprehensive route feasibility assessments demonstrating how this innovative synthesis can optimize your supply chain operations while ensuring uninterrupted access to high-quality intermediates.
