Advanced Metal-Free Synthesis of Isothiochromene Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex heterocyclic scaffolds. Patent CN113929658A introduces a groundbreaking methodology for the preparation of isothiochromene derivatives, a class of compounds renowned for their significant biological activities and utility as key intermediates in drug discovery. This innovation addresses critical bottlenecks in traditional synthesis by replacing expensive and toxic noble metal catalysts with graphitic carbon nitride (g-C3N4), a stable and recyclable organic semiconductor. By leveraging visible-light photocatalysis, this protocol achieves high atom utilization and excellent yields under remarkably mild conditions, positioning it as a transformative solution for reliable isothiochromene derivative supplier networks aiming to modernize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isothiochromene core has relied heavily on transition metal-catalyzed cyclization reactions, often utilizing homogeneous systems based on copper, ruthenium, or other precious metals. While these methods can effectively construct the bicyclic system, they suffer from severe drawbacks that hinder their application in GMP-compliant pharmaceutical manufacturing. The primary concern is the contamination of the final product with heavy metal residues, which necessitates complex and costly purification steps to meet stringent regulatory limits defined by guidelines such as ICH Q3D. Furthermore, homogeneous catalysts are notoriously difficult to separate from the reaction mixture and cannot be easily recycled, leading to significant waste generation and inflated operational costs. The reaction conditions are frequently harsh, requiring strong oxidants or elevated temperatures that limit substrate compatibility and pose safety risks during commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a heterogeneous photocatalytic system that fundamentally alters the reaction landscape. By employing graphitic carbon nitride (g-C3N4) as the catalyst, the process eliminates the risk of metal contamination entirely, thereby simplifying the purification workflow and ensuring a cleaner impurity profile. The reaction proceeds in absolute ethanol, a green and economically favorable solvent, under mild conditions with only 5% oxygen content, removing the need for hazardous strong oxidants. This approach not only enhances the safety profile of the manufacturing process but also significantly broadens the substrate scope, allowing for the incorporation of diverse functional groups such as halogens, alkyls, and alkoxy groups without degradation. The heterogeneous nature of the catalyst facilitates easy separation via filtration, enabling potential reuse and drastically reducing the environmental footprint of the synthesis.
Mechanistic Insights into g-C3N4 Photocatalytic Cyclization
The core of this technological advancement lies in the unique electronic properties of graphitic carbon nitride, which acts as a metal-free photocatalyst to drive the oxidative cyclization of N-aryl thioamides with alkynes. Upon irradiation, the g-C3N4 semiconductor absorbs light energy to generate electron-hole pairs, initiating a redox cycle that activates the molecular oxygen present in the reaction atmosphere. This activation generates reactive oxygen species that facilitate the radical addition of the thioamide sulfur atom to the alkyne triple bond, followed by intramolecular cyclization to form the isothiochromene ring system. This mechanism bypasses the need for stoichiometric chemical oxidants, relying instead on atmospheric oxygen as the terminal oxidant, which is a hallmark of green chemistry principles. The efficiency of this electron transfer process ensures high conversion rates while maintaining the integrity of sensitive functional groups on the aromatic rings.
From a quality control perspective, the absence of transition metals in the catalytic cycle provides a distinct advantage in managing the impurity profile of the final active pharmaceutical ingredient (API) intermediate. Traditional metal-catalyzed routes often generate organometallic byproducts or require aggressive chelating agents for metal scavenging, which can introduce new impurities or complicate the crystallization process. The g-C3N4 mediated pathway avoids these issues, resulting in a product with high purity that requires minimal downstream processing. Additionally, the mild reaction temperature prevents thermal decomposition of the substrate or product, further enhancing the overall yield and consistency of the batch. This mechanistic clarity offers R&D teams confidence in the reproducibility and robustness of the process when transferring from laboratory scale to pilot plant operations.
How to Synthesize Isothiochromene Derivatives Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and high efficiency, making it accessible for both laboratory research and industrial production. The process begins with the formation of an amide intermediate, followed by thionation to generate the key thioamide precursor, and concludes with the photocatalytic cyclization step. Each stage is optimized for maximum yield and minimal waste, utilizing common reagents and standard equipment. The detailed standardized synthesis steps see the guide below, which breaks down the precise molar ratios, solvent choices, and reaction times required to achieve the reported high-quality outcomes.
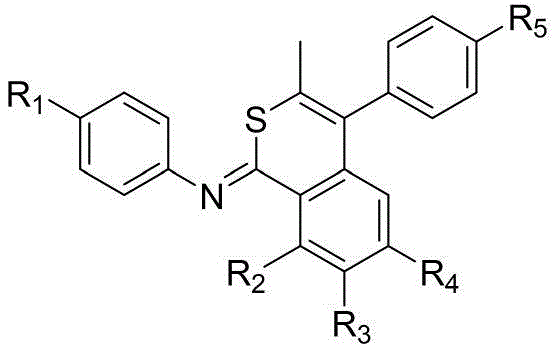
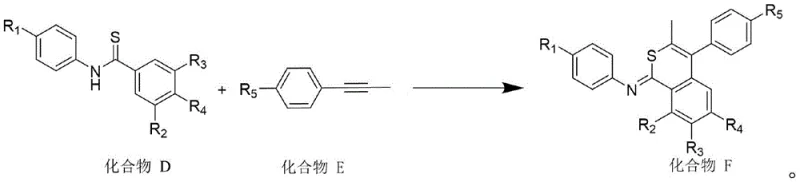
- Synthesize Compound C by reacting aniline derivatives (Compound A) with benzoyl chloride derivatives (Compound B) in ethyl acetate using triethylamine as a base.
- Convert Compound C to the corresponding thioamide (Compound D) using Lawesson's reagent in toluene at 135°C under nitrogen protection.
- Perform the final cyclization by reacting Compound D with an alkyne (Compound E) in absolute ethanol using graphitic carbon nitride (g-C3N4) as a photocatalyst under 5% oxygen content.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route presents a compelling value proposition centered on cost optimization and supply security. The elimination of noble metal catalysts removes a significant variable cost driver, as prices for metals like palladium and ruthenium are subject to volatile market fluctuations. Furthermore, the use of ethanol as a primary solvent replaces more expensive or hazardous organic solvents, contributing to substantial cost savings in raw material procurement. The simplified workup procedure, characterized by the ease of filtering the solid photocatalyst, reduces the consumption of auxiliary materials and shortens the overall production cycle time, thereby enhancing throughput capacity without requiring additional capital investment in specialized equipment.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally lowers the cost of goods sold (COGS) by eliminating the need for expensive precious metal catalysts and the associated metal scavenging resins often required for purification. The process utilizes inexpensive, commodity-grade chemicals such as urea-derived g-C3N4 and absolute ethanol, which are readily available in the global market at stable prices. By avoiding the use of strong oxidants and harsh reaction conditions, the process also reduces energy consumption and maintenance costs associated with corrosion-resistant reactor linings, leading to a more economically efficient manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials, such as substituted anilines and benzoyl chlorides, ensures a robust and resilient supply chain that is less susceptible to disruptions compared to routes dependent on specialized organometallic reagents. The simplicity of the reaction setup allows for flexible production scheduling and rapid scale-up, enabling suppliers to respond quickly to fluctuating market demands. Additionally, the stability of the g-C3N4 catalyst allows for potential storage and reuse strategies, further securing the continuity of supply and reducing the risk of production delays caused by catalyst shortages.
- Scalability and Environmental Compliance: This green synthesis method aligns perfectly with increasingly stringent environmental regulations, as it minimizes the generation of hazardous waste and avoids the discharge of heavy metals into the environment. The use of ethanol, a biodegradable and low-toxicity solvent, simplifies waste treatment protocols and reduces the regulatory burden associated with solvent recovery and disposal. The mild reaction conditions enhance process safety, lowering the risk of thermal runaways and making the technology highly suitable for large-scale commercial production in facilities with standard safety infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits and operational parameters of the technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: Why is the g-C3N4 photocatalytic method superior to traditional noble metal catalysis for isothiochromene synthesis?
A: Traditional methods often rely on homogeneous noble metal catalysts like copper or ruthenium, which leave toxic metal residues that are difficult to remove and unfavorable for pharmaceutical applications. The g-C3N4 method is heterogeneous, metal-free, and operates under mild conditions, ensuring high purity and easier downstream processing.
Q: What are the specific reaction conditions required for the final cyclization step?
A: The final cyclization step utilizes absolute ethanol as a green solvent and requires a 5% oxygen content atmosphere. The reaction proceeds at mild temperatures over 15-20 hours using graphitic carbon nitride as the photocatalyst, avoiding the need for harsh oxidants or extreme heat.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up due to its simple operation, use of inexpensive and readily available raw materials, and the ease of separating the heterogeneous catalyst. The absence of strong oxidants and the use of common solvents like ethanol further enhance its safety and economic viability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isothiochromene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free photocatalytic technology in advancing the synthesis of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isothiochromene derivative delivered meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite forward-thinking partners to collaborate with us to leverage this innovative synthesis route for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this green chemistry approach can optimize your budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both scientific success and commercial profitability in your drug development programs.
