Advanced Metal-Free Synthesis of Cyclic Thiourea Compounds for Commercial Pharmaceutical Manufacturing
Advanced Metal-Free Synthesis of Cyclic Thiourea Compounds for Commercial Pharmaceutical Manufacturing
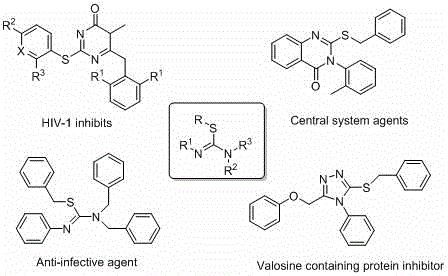
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective synthetic routes for complex heterocyclic building blocks. A significant breakthrough in this domain is detailed in patent CN113735777B, which discloses a novel method for preparing cyclic thiourea compounds. This technology addresses critical pain points in modern organic synthesis by eliminating the reliance on expensive transition metal catalysts. Instead, it utilizes a metal hydride-mediated coupling strategy that operates under mild conditions. For R&D directors and procurement managers, this represents a paradigm shift towards greener, more economical manufacturing of high-value intermediates used in HIV-1 inhibitors, anti-infective agents, and central nervous system drugs. The ability to synthesize these scaffolds without heavy metal contamination is particularly valuable for meeting stringent regulatory purity standards in API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of C-S bonds in thiourea derivatives has relied heavily on transition metal catalysis, often involving palladium or copper complexes. While effective, these conventional methods suffer from significant drawbacks that hinder large-scale commercial viability. The primary issue is the high cost and scarcity of noble metal catalysts, which directly impacts the cost of goods sold (COGS). Furthermore, sulfur-containing substrates are notorious for poisoning metal catalysts, leading to inconsistent reaction rates and the need for excessive catalyst loading. From a supply chain perspective, the requirement for rigorous exclusion of air and moisture in many metal-catalyzed protocols adds complexity to reactor operations. Additionally, the removal of trace metal residues from the final product requires additional purification steps, such as scavenging or recrystallization, which reduces overall yield and increases waste generation. These factors collectively create a bottleneck for the efficient manufacturing of high-purity pharmaceutical intermediates.
The Novel Approach
The methodology described in patent CN113735777B offers a transformative solution by employing a metal-free C-S coupling strategy. This approach utilizes readily available cyclic thioureas and diiodobenzene as substrates, reacting them in the presence of a metal hydride base such as sodium hydride (NaH) or potassium hydride (KH). The reaction proceeds smoothly at room temperature in a mixed solvent system of tetrahydrofuran (THF) and dimethylacetamide (DMA). This eliminates the need for high-temperature reflux or specialized pressure equipment. By avoiding transition metals entirely, the process inherently prevents metal contamination, simplifying downstream purification. The operational simplicity is further enhanced by the tolerance of various functional groups on the aromatic ring, including halogens, alkyls, and alkoxy groups. This versatility allows for the rapid synthesis of diverse libraries of S-arylated imidazoles, accelerating the drug discovery process while maintaining a lean manufacturing footprint.
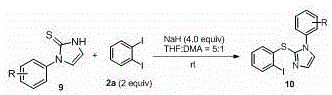
Mechanistic Insights into Metal Hydride-Mediated C-S Coupling
The core of this innovation lies in the nucleophilic substitution mechanism facilitated by the strong base. In the absence of a metal catalyst, the metal hydride serves to deprotonate the cyclic thiourea, generating a highly reactive nucleophilic species. This anionic intermediate then attacks the electron-deficient aryl iodide, specifically targeting the carbon-iodine bond. The use of diiodobenzene is strategic; the first iodine atom acts as the leaving group for the C-S bond formation, while the second iodine atom remains intact on the aromatic ring. This retained iodine functionality is crucial for subsequent derivatization, allowing chemists to perform further cross-coupling reactions to build even more complex molecular architectures. The reaction kinetics are favorable at room temperature, suggesting a low activation energy barrier for this specific nucleophilic aromatic substitution pathway. This mechanistic understanding confirms that the process is not only chemically efficient but also robust against minor variations in reaction parameters, a key requirement for industrial reproducibility.
Impurity control is another critical aspect where this mechanism excels. In traditional metal-catalyzed routes, side reactions such as homocoupling of the aryl halide or desulfurization can occur, leading to difficult-to-remove impurities. The metal-free nature of this protocol minimizes these pathways. The primary byproducts are typically inorganic salts derived from the base and the leaving iodide, which are easily removed during the aqueous workup phase. The patent data indicates high regioselectivity, ensuring that the sulfur atom attaches exclusively to the intended position on the imidazole ring. This high selectivity translates directly to higher crude purity, reducing the burden on purification teams. For quality control laboratories, this means simpler analytical profiles and faster release times for batches, enhancing overall supply chain agility.
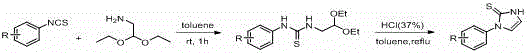
How to Synthesize Cyclic Thiourea Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates. The process begins with the preparation of the cyclic thiourea precursor, which can be synthesized from isothiocyanates and aminoacetaldehyde diethyl acetal under acidic conditions. Once the precursor is obtained, the key C-S coupling step is executed by suspending sodium hydride in anhydrous THF. The cyclic thiourea is added dropwise to ensure controlled gas evolution and heat management. Following the addition of the diiodobenzene electrophile, the mixture is stirred at ambient temperature. Monitoring via thin-layer chromatography (TLC) allows for precise determination of the reaction endpoint, typically ranging from 2 to 10 hours depending on the electronic nature of the substituents. The detailed standardized synthesis steps for scaling this process are provided in the guide below.
- Suspend metal hydride (NaH or KH) in anhydrous THF and stir under inert atmosphere.
- Dropwise add the cyclic thiourea substrate dissolved in DMA to the suspension at room temperature.
- Add diiodobenzene solution, stir at room temperature until TLC indicates completion, then quench and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers substantial strategic benefits. The most immediate impact is seen in the reduction of raw material costs. By replacing expensive palladium or copper catalysts with commodity chemicals like sodium hydride and diiodobenzene, the direct material cost per kilogram of product is significantly lowered. Furthermore, the elimination of metal scavengers and the associated filtration media reduces consumable expenses. The simplified workflow also translates to reduced labor costs, as the process does not require the specialized handling procedures associated with air-sensitive catalysts. This efficiency gain allows manufacturing facilities to allocate resources to other critical production lines, optimizing overall plant throughput.
- Cost Reduction in Manufacturing: The economic model of this synthesis is driven by the substitution of high-value catalysts with low-cost bases. Transition metal catalysts often account for a significant portion of the variable cost in fine chemical synthesis. By removing this line item entirely, the margin for error in pricing becomes much more comfortable. Additionally, the room temperature operation eliminates the energy costs associated with heating reactors to high temperatures or maintaining cryogenic conditions. The simplified purification process, which relies on standard extraction and chromatography rather than complex metal removal technologies, further drives down the cost of goods. These cumulative savings make the final API intermediate much more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Sodium hydride, THF, DMA, and diiodobenzene are commodity chemicals produced by multiple suppliers globally, reducing the risk of single-source dependency. In contrast, specialized ligands and catalysts often have longer lead times and are subject to geopolitical supply constraints. The robustness of the reaction conditions also means that the process is less susceptible to delays caused by equipment failures related to heating or cooling systems. This reliability ensures consistent delivery schedules to downstream customers, fostering stronger long-term partnerships and reducing the need for safety stock inventory.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with green chemistry principles. The absence of heavy metals simplifies waste treatment protocols, as the effluent does not require specialized processing to remove toxic metal ions. This reduces the environmental compliance burden and associated disposal fees. The scalability of the process is evident from its mild conditions; exotherms are manageable, and the reaction does not generate hazardous gases beyond the hydrogen evolved from the hydride, which is easily vented. This makes the transition from pilot scale to multi-ton commercial production straightforward, enabling rapid response to market demand surges without extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. They are designed to clarify the operational feasibility and strategic value of implementing this route in a commercial setting. Understanding these details is essential for making informed decisions about process adoption and supplier selection.
Q: What are the primary advantages of this metal-free synthesis method?
A: The method eliminates the need for expensive transition metal catalysts like palladium or copper, significantly reducing raw material costs and avoiding heavy metal contamination in the final API intermediate.
Q: What are the typical reaction conditions required?
A: The reaction proceeds efficiently at room temperature using a THF/DMA solvent system with sodium hydride as the base, offering excellent functional group tolerance and operational simplicity.
Q: Can this process be scaled for industrial production?
A: Yes, the use of commercially available reagents, mild temperatures, and the absence of air-sensitive catalysts make this protocol highly suitable for commercial scale-up and continuous manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic Thiourea Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis technology described in CN113735777B. As a leading CDMO partner, we possess the technical expertise to translate this laboratory-scale innovation into a robust commercial manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of cyclic thiourea intermediate meets the highest industry standards for pharmaceutical applications. Our commitment to quality ensures that your downstream synthesis remains uninterrupted and compliant.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your time-to-market with our reliable Cyclic Thiourea supply solutions.
