Advanced Rhodium-Catalyzed Synthesis of Isoxazolidine Scaffolds for Commercial Pharmaceutical Manufacturing
Introduction to Novel Isoxazolidine Synthesis Technology
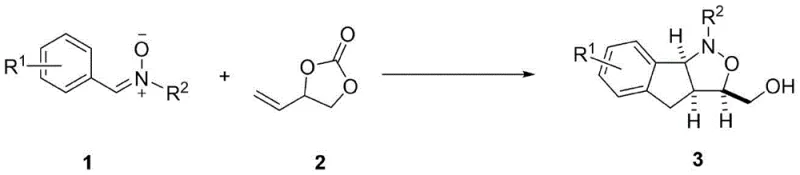
The pharmaceutical industry continuously seeks efficient pathways to access privileged scaffolds that serve as the backbone for bioactive molecules. Patent CN115215814A introduces a groundbreaking synthetic methodology for constructing isoxazolidine compounds, a structural motif frequently encountered in natural products with significant biological activities such as antitumor and anti-inflammatory properties. This technology leverages advanced transition metal catalysis to achieve what was previously difficult through conventional organic synthesis. By utilizing a rhodium-based catalytic system, the process enables the direct coupling of nitrone compounds with vinyl carbonates, bypassing the need for pre-functionalized substrates. This represents a paradigm shift in how chemists approach the construction of nitrogen-oxygen heterocycles, offering a streamlined route that aligns perfectly with the demands of modern process chemistry for reliability and efficiency.
The core innovation lies in the ability to activate inert carbon-hydrogen bonds under relatively mild thermal conditions. Traditional methods often require stoichiometric amounts of hazardous reagents or extreme temperatures that can degrade sensitive functional groups. In contrast, this rhodium-catalyzed approach operates effectively at temperatures ranging from 60°C to 80°C, preserving the integrity of complex molecular architectures. For research and development teams focused on lead optimization, this means faster iteration cycles and the ability to explore chemical space more broadly without being constrained by synthetic feasibility. The robustness of this method suggests it is highly suitable for the production of high-purity pharmaceutical intermediates where consistency and quality are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoxazolidine rings has been plagued by significant challenges that hinder scalability and cost-effectiveness in industrial settings. Conventional routes often rely on 1,3-dipolar cycloadditions which can suffer from poor regioselectivity, leading to mixtures of isomers that are difficult and expensive to separate. Furthermore, many traditional protocols necessitate the use of highly reactive dipolarophiles that are unstable or toxic, posing safety risks during large-scale manufacturing. The requirement for multiple protection and deprotection steps to manage functional group compatibility further elongates the synthetic sequence, driving up material costs and waste generation. These inefficiencies create bottlenecks in the supply chain, making it difficult for procurement managers to secure reliable sources of complex intermediates at competitive prices.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by employing a catalytic C-H activation strategy that fundamentally simplifies the synthetic logic. Instead of relying on pre-activated halides or unstable dipoles, the process utilizes readily available nitrone compounds and vinyl carbonates as building blocks. The rhodium catalyst facilitates a highly selective cyclization that constructs the isoxazolidine core with precise stereocontrol, eliminating the formation of unwanted byproducts. This atom-economical approach not only reduces the consumption of raw materials but also simplifies the downstream purification process. For supply chain heads, this translates to a more resilient manufacturing process with fewer unit operations, thereby reducing the risk of production delays and ensuring a steady flow of critical intermediates for drug development pipelines.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
At the heart of this technology is a sophisticated catalytic cycle driven by the pentamethylcyclopentadienyl rhodium(III) complex, typically generated in situ from [Cp*RhCl2]2 and a silver salt like AgSbF6. The mechanism initiates with the coordination of the nitrone nitrogen to the cationic rhodium species, followed by the concerted metalation-deprotonation (CMD) of the adjacent aromatic C-H bond. This step generates a stable five-membered rhodacycle intermediate, which is the key to the reaction's high regioselectivity. The subsequent insertion of the vinyl carbonate alkene into the rhodium-carbon bond sets the stage for the formation of the new carbon-carbon bond. Finally, reductive elimination or nucleophilic attack by the oxygen atom closes the isoxazolidine ring, regenerating the active catalyst. Understanding this mechanistic pathway is crucial for R&D directors aiming to troubleshoot potential scale-up issues or adapt the chemistry to novel substrates.
Impurity control is inherently built into this mechanism due to the high specificity of the C-H activation step. Unlike radical processes that can lead to indiscriminate functionalization, the directed nature of the rhodium catalyst ensures that reaction occurs only at the intended position ortho to the nitrone directing group. This minimizes the formation of regioisomeric impurities that often complicate regulatory filings for new drug applications. Additionally, the use of silver additives helps to scavenge halides and maintain the cationic nature of the rhodium center, further enhancing catalytic turnover and stability. For quality assurance teams, this inherent selectivity means that the crude product profile is much cleaner, reducing the burden on analytical laboratories and accelerating the release of batches for clinical use.
How to Synthesize Isoxazolidine Compounds Efficiently
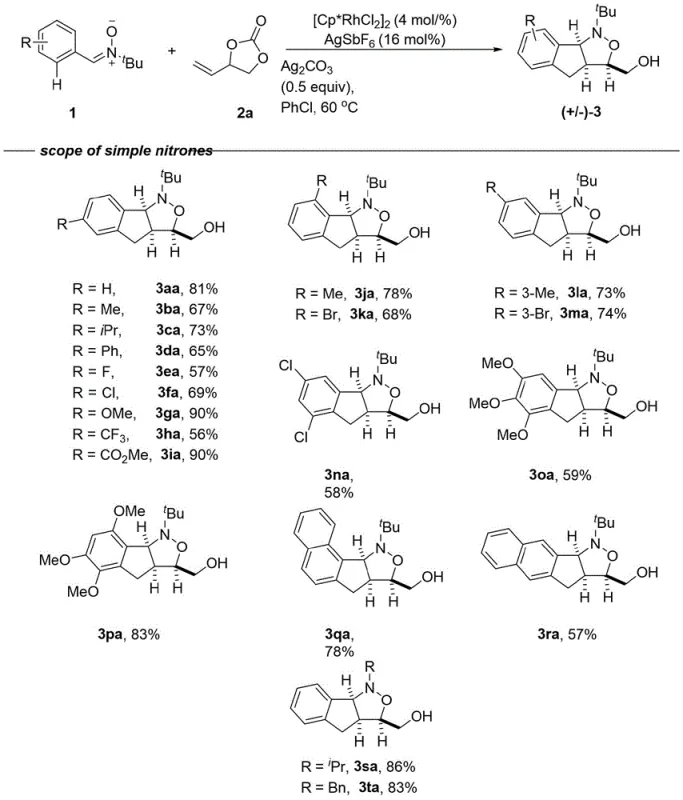
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The standard protocol involves mixing the nitrone substrate with a slight excess of the vinyl carbonate coupling partner in a suitable organic solvent such as chlorobenzene or trifluoroethanol. The catalyst system, comprising the rhodium dimer and silver hexafluoroantimonate, is added along with a base like silver carbonate to neutralize acidic byproducts. The reaction is typically conducted under an inert atmosphere to protect the catalyst from oxidation. While the general procedure is robust, optimization of temperature and concentration may be necessary for sterically hindered substrates. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the nitrone compound (1 equiv), vinyl carbonate derivative (1-1.5 equiv), [Cp*RhCl2]2 catalyst (4 mol%), AgSbF6 (16 mol%), and Ag2CO3 additive (0.5 equiv) in an organic solvent such as chlorobenzene.
- Seal the reaction vessel under an inert atmosphere (argon or nitrogen) to prevent oxidation and moisture interference, ensuring the stability of the sensitive rhodium catalyst species throughout the process.
- Heat the mixture to a temperature between 60°C and 80°C for approximately 12 to 24 hours, then isolate the final isoxazolidine product via silica gel column chromatography after solvent removal.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic technology offers compelling advantages that directly address the pain points of modern pharmaceutical manufacturing. The reliance on earth-abundant and commercially available starting materials significantly lowers the barrier to entry for production. Nitrones and vinyl carbonates are stable, easy to handle, and can be sourced from multiple suppliers, mitigating the risk of single-source dependency. The mild reaction conditions reduce energy consumption compared to high-temperature processes, contributing to lower operational expenditures. Furthermore, the simplified workup procedure, often requiring only standard column chromatography or crystallization, minimizes solvent usage and waste disposal costs. These factors combine to create a cost structure that is highly competitive for the production of high-value intermediates.
- Cost Reduction in Manufacturing: The elimination of pre-functionalized starting materials and the reduction in synthetic steps lead to substantial cost savings. By avoiding the use of expensive organometallic reagents or hazardous halogenated precursors, the overall material cost is drastically reduced. The high catalytic efficiency means that only small amounts of the precious rhodium metal are required, and recovery strategies can be implemented to further optimize economics. This efficiency allows for the production of complex isoxazolidine derivatives at a fraction of the cost associated with legacy methods, providing a clear margin advantage for generic drug manufacturers and CDMOs alike.
- Enhanced Supply Chain Reliability: The robustness of the reaction across a wide range of substrates ensures consistent output regardless of minor variations in raw material quality. The protocol has been demonstrated to work effectively with nitrones derived from complex natural products and pharmaceuticals, indicating high tolerance to functional groups. This versatility means that a single manufacturing platform can be adapted to produce a diverse library of intermediates, simplifying inventory management and logistics. For supply chain heads, this flexibility translates to shorter lead times and the ability to respond rapidly to changing market demands without the need for extensive process re-validation.
- Scalability and Environmental Compliance: The use of benign solvents and the absence of toxic byproducts align well with green chemistry principles and increasingly stringent environmental regulations. The process generates minimal waste, reducing the burden on effluent treatment facilities. Scalability is supported by the homogeneous nature of the catalytic system, which allows for efficient heat and mass transfer in larger reactors. This makes the technology suitable for commercial scale-up of complex pharmaceutical intermediates, ensuring that supply can meet the demands of late-stage clinical trials and commercial launch without technical bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this rhodium-catalyzed synthesis. These insights are derived directly from the experimental data and scope studies presented in the patent documentation. They are intended to provide clarity for technical teams evaluating the feasibility of adopting this route for their specific projects. Understanding these nuances is essential for successful technology transfer and process optimization.
Q: What are the key advantages of this Rh-catalyzed method over traditional isoxazolidine synthesis?
A: This method utilizes mild reaction conditions (60-80°C) and avoids harsh reagents, offering excellent regio- and stereoselectivity. It allows for the direct functionalization of C-H bonds, significantly reducing the number of synthetic steps required compared to classical cyclization methods.
Q: Can this synthesis protocol accommodate complex natural product derivatives?
A: Yes, the patent demonstrates high substrate tolerance, successfully applying the methodology to nitrones derived from complex natural products and pharmaceutical molecules such as Gemfibrozil, Ibuprofen, and Tocopherol, yielding functionalized isoxazolidines with good efficiency.
Q: What downstream transformations are possible with the resulting isoxazolidine products?
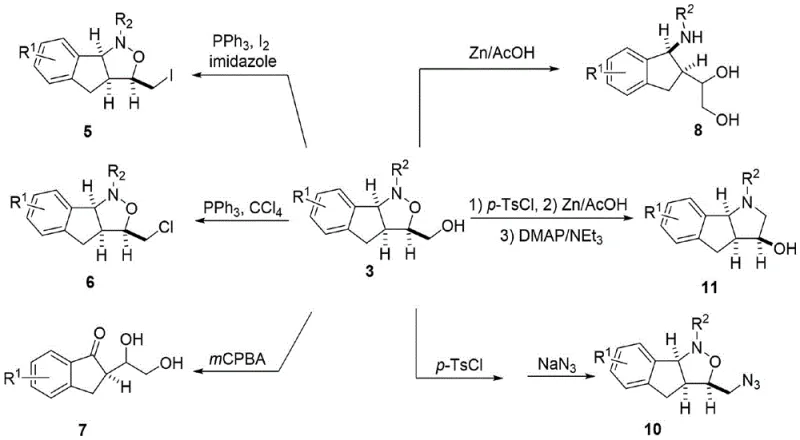
A: The synthesized isoxazolidine scaffold serves as a versatile intermediate that can be converted into various bioactive derivatives, including iodo/chloro-substituted compounds, azides, and ring-opened amino alcohols, facilitating late-stage functionalization for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazolidine Compounds Supplier
As the demand for complex heterocyclic intermediates grows, partnering with an experienced CDMO is critical for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We understand the complexities of transition metal catalysis and have the expertise to optimize these reactions for maximum efficiency and safety on an industrial scale.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target isoxazolidine derivatives. Let us help you accelerate your drug development timeline with our superior manufacturing capabilities and commitment to quality excellence.
