Revolutionizing 4-Aryl-4,5-Dihydrofuran Production: A Green, Base-Catalyzed Route for Scalable Manufacturing
Revolutionizing 4-Aryl-4,5-Dihydrofuran Production: A Green, Base-Catalyzed Route for Scalable Manufacturing
The synthesis of 4-aryl-4,5-dihydrofuran derivatives represents a critical challenge in modern organic chemistry, particularly for the development of complex pharmaceutical intermediates and bioactive natural products. As detailed in patent CN101824010B, a groundbreaking methodology has been established that shifts the paradigm from hazardous heavy metal catalysis to a benign, base-promoted cyclization strategy. This innovation addresses the urgent industry demand for sustainable manufacturing processes that do not compromise on yield or purity. By utilizing readily available aryl-substituted nitroethylenes and 1,3-dicarbonyl compounds, this technology enables the direct construction of the dihydrofuran core in a single operational step. For R&D directors and process chemists, this represents a significant leap forward in synthetic efficiency, offering a robust pathway to access structurally diverse heterocyclic scaffolds essential for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4-aryl-4,5-dihydrofuran skeleton has relied heavily on transition metal-mediated oxidative cyclizations, which pose severe logistical and environmental burdens. Traditional protocols often employ stoichiometric amounts of toxic heavy metals such as thallium(III) acetate or lead(IV) acetate in conjunction with strong acids like perchloric acid. As illustrated in the legacy reaction schemes below, these methods not only suffer from moderate yields but also generate substantial quantities of hazardous waste that require costly disposal procedures.
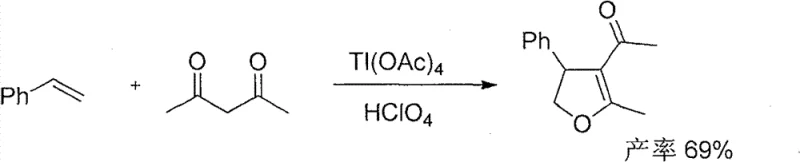
Furthermore, alternative routes utilizing manganese acetate or multi-step sequences involving sulfonium ylides introduce additional complexities, including stringent temperature controls and difficult purification steps to remove metal residues. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for material delivery, creating bottlenecks for supply chain managers who require consistent, high-volume availability of key intermediates without regulatory compliance risks associated with heavy metal limits in final API products.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data introduces a streamlined, one-pot synthesis that completely bypasses the need for transition metal catalysts. By leveraging the nucleophilic activation of 1,3-dicarbonyl compounds via simple inorganic bases like potassium carbonate in dimethyl sulfoxide (DMSO), the reaction proceeds through a concerted Michael addition-intramolecular cyclization cascade. This approach transforms the synthesis into a operationally simple process where reagents are mixed and heated to mild temperatures ranging from 30°C to 80°C.
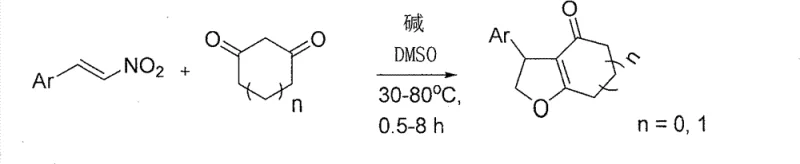
The result is a dramatic improvement in process mass intensity (PMI) and overall throughput. The elimination of toxic reagents simplifies the workup procedure to a standard acid quench and extraction, thereby removing the need for specialized scavenging resins or complex chromatographic separations often required to meet strict pharmaceutical purity specifications. This shift not only enhances safety profiles for plant operators but also aligns perfectly with green chemistry principles, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the dual role of the base and the solvent system in facilitating the cascade reaction. Initially, the alkali catalyst deprotonates the active methylene group of the 1,3-dicarbonyl compound, generating a stabilized enolate nucleophile. This species then undergoes a conjugate addition (Michael addition) to the electron-deficient beta-carbon of the aryl-substituted nitroethylene. The nitro group serves as a powerful electron-withdrawing anchor, activating the alkene for nucleophilic attack while simultaneously stabilizing the resulting anionic intermediate.
Following the initial carbon-carbon bond formation, the proximity of the carbonyl oxygen allows for an intramolecular nucleophilic attack on the adjacent carbon bearing the nitro group, or via a nitro-elimination pathway depending on the specific substitution pattern, ultimately closing the five-membered furan ring. The use of DMSO as the solvent is critical, as its high polarity stabilizes the charged intermediates and transition states, accelerating the reaction kinetics without the need for extreme thermal energy. This mechanistic pathway ensures high regioselectivity, predominantly forming the 4-aryl isomer over other potential regioisomers, which is crucial for maintaining a clean impurity profile in the final product.
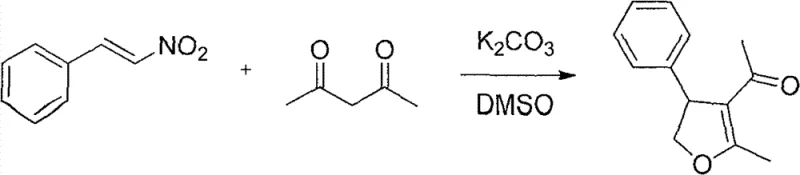
Moreover, the tolerance of this mechanism towards various electronic environments on the aromatic ring is noteworthy. Whether the aryl group contains electron-donating substituents like methoxy or methyl groups, or electron-withdrawing halogens such as chlorine and bromine, the cyclization proceeds efficiently. This broad substrate scope indicates that the rate-determining step is likely robust against steric and electronic variations, providing process chemists with a versatile platform for synthesizing a wide library of analogues for structure-activity relationship (SAR) studies without needing to re-optimize conditions for each new derivative.
How to Synthesize 4-Aryl-4,5-Dihydrofuran Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, precise control over reaction parameters is essential to maximize yield and minimize side reactions. The patent data outlines a generalized protocol that balances reagent stoichiometry and thermal input to achieve optimal conversion. While the specific details of the standardized operating procedure are proprietary to the full technical documentation, the fundamental steps involve the careful mixing of the nitroalkene and diketone precursors in dry DMSO, followed by the controlled addition of the base catalyst.
- Combine aryl-substituted nitroethylene and 1,3-dicarbonyl compound (e.g., acetylacetone) in dimethyl sulfoxide (DMSO) solvent.
- Add an alkali catalyst such as potassium carbonate (K2CO3) with a molar ratio preferably around 0.6 relative to the nitro compound.
- Stir the reaction mixture at temperatures between 30°C and 80°C for 0.5 to 8 hours until conversion is complete, followed by standard acid workup and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this base-catalyzed synthesis offers profound advantages for procurement strategies and supply chain resilience. The most immediate impact is the drastic simplification of the raw material portfolio. By replacing expensive and regulated heavy metal salts with commodity chemicals like potassium carbonate and acetylacetone, manufacturers can significantly mitigate supply risk and price volatility. This shift ensures a more stable sourcing channel, as the key reagents are produced globally in massive quantities for various industrial applications, reducing the likelihood of shortages that could disrupt production schedules.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of costly downstream processing steps. In traditional heavy metal routes, a significant portion of the manufacturing budget is allocated to metal scavenging, waste treatment, and extensive purification to meet residual metal limits. By removing these requirements entirely, the new method reduces the consumption of auxiliary materials and lowers the burden on waste management infrastructure. Furthermore, the one-pot nature of the reaction minimizes labor hours and equipment occupancy time, leading to substantial operational expenditure savings that can be passed down to the customer.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions directly translates to improved supply chain reliability. Unlike sensitive organometallic reactions that may require inert atmospheres or cryogenic cooling, this base-catalyzed process operates under ambient pressure and moderate temperatures. This tolerance makes the process less susceptible to minor fluctuations in plant utilities or operator error, ensuring consistent batch-to-batch quality. For supply chain heads, this predictability is invaluable, as it allows for more accurate forecasting and reduces the need for excessive safety stock holdings.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the simplicity of this homogeneous reaction system facilitates smooth scale-up from gram to ton scales. The use of DMSO, a high-boiling polar aprotic solvent, allows for flexible temperature control, while the absence of toxic heavy metals simplifies regulatory compliance and environmental permitting. This alignment with increasingly stringent global environmental standards future-proofs the manufacturing asset, ensuring long-term viability and reducing the risk of shutdowns due to non-compliance issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the practical implications for potential partners and licensees.
Q: What are the primary advantages of this base-catalyzed method over traditional heavy metal routes?
A: This method eliminates the use of toxic and expensive heavy metal catalysts like thallium acetate or lead acetate. It operates under milder conditions using inexpensive inorganic bases like potassium carbonate, significantly reducing environmental hazards and downstream purification costs associated with heavy metal removal.
Q: Can this synthesis protocol accommodate diverse aromatic substrates?
A: Yes, the process demonstrates excellent substrate universality. It successfully tolerates various substituents on the aromatic ring, including electron-withdrawing groups like halogens (Cl, Br) and electron-donating groups like methoxy or methyl, as well as heteroaromatic systems like furan, yielding the corresponding dihydrofuran derivatives efficiently.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The one-pot nature of the reaction, combined with the use of commercially available solvents like DMSO and stable solid bases, simplifies operational complexity. The absence of sensitive reagents requiring low temperatures or inert atmospheres makes it highly amenable to scale-up for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aryl-4,5-Dihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 4-aryl-4,5-dihydrofuran intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this green synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency into your supply chain.
