Advanced Synthesis of High-Purity Chloro 2-Carboxyl Benzophenone for Microelectronics
Advanced Synthesis of High-Purity Chloro 2-Carboxyl Benzophenone for Microelectronics
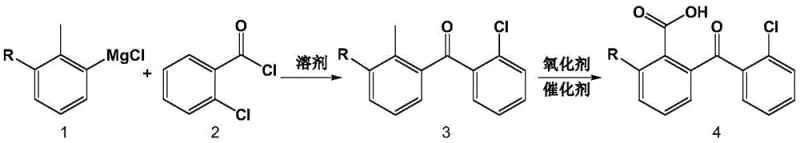
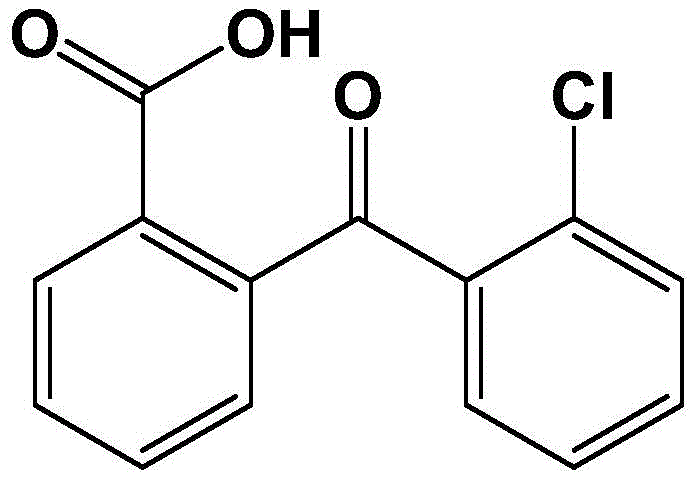
The rapid evolution of the microelectronics and semiconductor industries demands chemical intermediates of exceptional purity and structural precision. Addressing this critical need, the technical disclosure found in patent CN108530293B introduces a robust and environmentally conscious preparation method for high-purity chloro 2-carboxyl benzophenone. This innovative synthetic route departs from traditional, cumbersome methodologies by leveraging a streamlined Grignard reaction followed by a catalytic aerobic oxidation. For R&D directors and procurement specialists seeking reliable sources of advanced electronic chemicals, this process represents a significant leap forward in both quality assurance and process efficiency. By utilizing readily available starting materials like o-chlorotoluene and o-chlorobenzoyl chloride, the method ensures a stable supply chain while delivering a final product with purity specifications that exceed 99.9% under optimized conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzophenone derivatives, particularly those with specific carboxyl substitutions, has relied heavily on Friedel-Crafts acylation reactions involving benzene and phthalic anhydride. While effective for unsubstituted variants, these classical approaches struggle significantly when introducing specific substituents like chlorine atoms onto the aromatic rings without generating complex mixtures of isomers. The lack of regioselectivity in traditional electrophilic aromatic substitution often necessitates difficult and yield-reducing separation processes, which are economically prohibitive for high-value electronic applications. Furthermore, conventional oxidation methods to convert methyl groups to carboxylic acids frequently employ stoichiometric amounts of harsh oxidants such as potassium permanganate or chromic acid, generating substantial quantities of hazardous heavy metal waste that complicate disposal and increase environmental compliance costs.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a highly selective Grignard coupling strategy to construct the benzophenone skeleton with precise control over substitution patterns. By first converting substituted o-chlorotoluene into a Grignard reagent and subsequently reacting it with o-chlorobenzoyl chloride, the process ensures the correct positioning of the carbonyl bridge and the chlorine substituents from the outset. This is followed by a catalytic oxidation step that uses molecular oxygen or air as the terminal oxidant, drastically reducing the chemical waste footprint compared to stoichiometric oxidants. The result is a cleaner reaction profile that simplifies downstream purification, allowing for the isolation of the target chloro 2-carboxyl benzophenone with minimal impurity burden.
Mechanistic Insights into Grignard Coupling and Aerobic Oxidation
The core of this synthetic success lies in the meticulous control of the Grignard formation and the subsequent transition-metal catalyzed oxidation. The initial step involves the insertion of magnesium into the carbon-halogen bond of the o-chlorotoluene derivative in an anhydrous ether or tetrahydrofuran environment. This generates a highly nucleophilic organomagnesium species that attacks the electrophilic carbonyl carbon of the o-chlorobenzoyl chloride. The reaction conditions, typically maintained between 0°C and 150°C depending on the solvent system, allow for complete conversion to the ketone intermediate while minimizing side reactions such as Wurtz-type coupling. The choice of solvent, ranging from petroleum ether to toluene, plays a pivotal role in solubilizing the intermediates and managing the exothermic nature of the Grignard addition, ensuring safety and reproducibility at scale.
Following the formation of the chloro-2-methylbenzophenone intermediate, the transformation of the benzylic methyl group into a carboxylic acid is achieved through a radical-mediated autoxidation mechanism. Catalysts such as cobalt acetate or manganese acetate facilitate the generation of benzylic radicals which react with dissolved oxygen to form hydroperoxides. These unstable intermediates decompose to yield the corresponding aldehyde and finally the carboxylic acid. The use of acetic acid or propionic acid as the reaction solvent not only solubilizes the organic substrate but also stabilizes the metal catalyst species. This catalytic cycle is highly efficient, requiring only minute amounts of metal catalyst (0.001-5% molar ratio), and proceeds smoothly at moderate temperatures of 20-150°C, preserving the integrity of the sensitive chloro-substituents on the aromatic rings.
How to Synthesize Chloro 2-Carboxyl Benzophenone Efficiently
Implementing this synthesis requires careful attention to moisture exclusion during the Grignard phase and precise temperature control during the oxidation phase. The process begins with the activation of magnesium turnings, often aided by iodine, to ensure rapid initiation of the organometallic formation. Once the Grignard reagent is established, the controlled addition of the acid chloride is critical to manage heat release. The subsequent oxidation step benefits from efficient gas-liquid mass transfer to ensure adequate oxygen supply for the catalytic cycle. Finally, the purification strategy relies on a specific recrystallization protocol using mixed solvents to achieve the ultra-high purity demanded by the electronics sector.
- Prepare the Grignard reagent by reacting chlorine-substituted or non-substituted o-chlorotoluene with magnesium powder in anhydrous ether or tetrahydrofuran under nitrogen protection.
- React the prepared Grignard reagent with o-chlorobenzoyl chloride in a suitable solvent at temperatures between 0-150°C to form the chloro-2-methylbenzophenone intermediate.
- Oxidize the methyl group of the intermediate to a carboxyl group using air or oxygen in the presence of a cobalt or manganese acetate catalyst in acetic acid solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers distinct strategic advantages over legacy manufacturing routes. The shift towards catalytic aerobic oxidation eliminates the need for purchasing and disposing of expensive, hazardous stoichiometric oxidants, leading to a drastic simplification of the waste management infrastructure. This reduction in hazardous waste handling translates directly into lower operational expenditures and reduced regulatory burden, making the supply of these critical intermediates more resilient against environmental policy changes. Additionally, the use of commodity chemicals like o-chlorotoluene and magnesium ensures that raw material costs remain stable and predictable, shielding buyers from the volatility often associated with specialized reagents.
- Cost Reduction in Manufacturing: The elimination of stoichiometric heavy metal oxidants and the use of low-loading transition metal catalysts significantly reduce the raw material cost per kilogram of the final product. Furthermore, the simplified work-up procedure, which involves hot filtration and direct recrystallization rather than complex chromatographic separations, lowers energy consumption and labor costs. The ability to recycle solvents like toluene and acetic acid further enhances the economic viability of the process, ensuring that the final price point remains competitive for high-volume applications in the microelectronics sector.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals such as o-chlorotoluene and o-chlorobenzoyl chloride, the manufacturing process is decoupled from the supply risks associated with exotic or single-source reagents. The robustness of the Grignard and oxidation steps allows for flexible production scheduling, as the reaction windows are broad (1-36 hours for coupling, 2-48 hours for oxidation), accommodating various batch sizes without compromising quality. This flexibility ensures consistent delivery timelines, a critical factor for downstream manufacturers who operate on tight just-in-time production schedules for semiconductor fabrication.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations common in fine chemical plants. The absence of highly toxic reagents like chromium or manganese in stoichiometric quantities simplifies the environmental permitting process and reduces the long-term liability associated with site contamination. The final recrystallization step using a toluene-ethanol mixture is easily scalable and effective at removing trace metal catalysts and organic by-products, ensuring that the final product meets the rigorous purity standards required for electronic grade materials without the need for additional polishing steps.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and capabilities of this synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this intermediate into their own formulation or synthesis pipelines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and reliability for decision-making purposes.
Q: What purity levels can be achieved with this synthesis method?
A: The patented process utilizes a specific toluene-ethanol recrystallization system that consistently achieves product purity exceeding 99.5%, with optimized ratios reaching greater than 99.9%, meeting the stringent requirements for microelectronic applications.
Q: Which catalysts are suitable for the oxidation step?
A: The process supports a variety of transition metal catalysts including cobalt acetate, manganese acetate, nickel acetate, and copper acetate, often used in conjunction with bromide promoters to facilitate efficient aerobic oxidation.
Q: Is this method scalable for industrial production?
A: Yes, the method employs standard unit operations such as Grignard formation, reflux, and hot filtration followed by recrystallization, making it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloro 2-Carboxyl Benzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the advancement of microelectronic and pharmaceutical technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of chloro 2-carboxyl benzophenone meets the exacting standards of the global electronics industry. Our commitment to quality assurance means that you receive a product that is ready for immediate use in sensitive applications without the need for further purification.
We invite you to collaborate with us to optimize your supply chain for these essential chemical building blocks. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient manufacturing processes can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can support your next generation of electronic materials.
