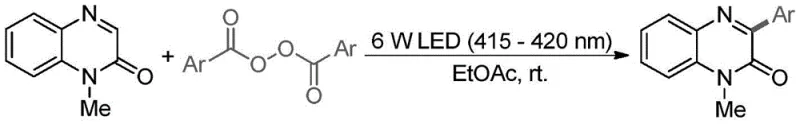
Advanced Photocatalytic Route for High-Purity 3-Aryl-N-Methylquinoxalin-2(1H)-one Manufacturing
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as privileged structures in drug discovery. Patent CN112028841A introduces a groundbreaking photocatalytic synthesis method for 3-aryl-N-methylquinoxalin-2(1H)-one compounds, a class of molecules renowned for their anticancer and antibacterial properties. This technology represents a paradigm shift from traditional thermal or metal-catalyzed processes to a mild, visible-light-driven protocol that eliminates the need for external photocatalysts. By leveraging the intrinsic photosensitivity of the quinoxalinone substrate itself under 415-420 nm violet light irradiation, this method achieves high selectivity and excellent yields in an environmentally benign ethyl acetate solvent system. For global procurement teams and R&D directors, this innovation signals a move towards greener chemistry that does not compromise on output quality or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the C3-functionalization of quinoxalin-2(1H)-ones has been fraught with significant operational challenges that hinder large-scale manufacturing. Prior art methods often rely on harsh conditions or exotic reagents that complicate supply chains and increase production costs. For instance, earlier approaches utilized specialized phase transfer catalysts like 2D-COF-1, which are difficult to synthesize and procure in bulk quantities, creating a bottleneck for industrial application. Other reported strategies employed strong oxidants like potassium persulfate under heating conditions, which not only extend reaction times but also pose safety risks due to thermal instability. Furthermore, these conventional routes frequently suffer from moderate to low yields and poor atom economy, generating substantial waste streams that require costly disposal protocols. The reliance on transition metals in some variants also introduces the risk of heavy metal contamination, necessitating rigorous and expensive purification steps to meet stringent pharmaceutical purity standards.
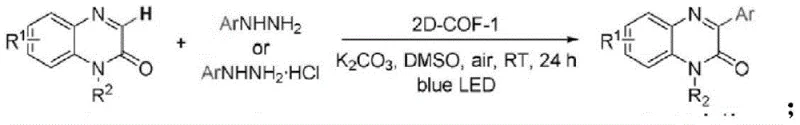
The Novel Approach
In stark contrast, the novel photocatalytic method disclosed in the patent offers a streamlined, one-pot solution that operates under exceptionally mild conditions. By utilizing readily available aryl peroxides as the aryl source and simple LED lights as the energy input, this process bypasses the need for expensive catalysts entirely. The reaction proceeds efficiently at room temperature in ethyl acetate, a green solvent preferred for its low toxicity and ease of recovery. This approach not only simplifies the operational workflow but also dramatically improves the impurity profile of the final product. The elimination of external additives means fewer downstream purification steps, directly translating to reduced processing time and lower utility consumption. Moreover, the method exhibits remarkable robustness across a wide range of substrates, maintaining high performance even with sterically hindered or electronically diverse aryl groups, making it a versatile platform for generating diverse libraries of bioactive intermediates.
Mechanistic Insights into Visible-Light Induced Radical Decarboxylation
Understanding the underlying mechanism is crucial for R&D teams aiming to optimize this process for specific analogues. The reaction operates through a fascinating radical pathway initiated by the absorption of violet light (415-420 nm) by the N-methylquinoxalin-2(1H)-one substrate. Upon excitation, the substrate enters a highly reactive excited state that facilitates single-electron transfer with the aryl peroxide reagent. This interaction triggers the homolytic cleavage of the peroxide bond, generating a benzoyloxy radical which rapidly undergoes decarboxylation to release carbon dioxide and form a reactive phenyl radical. This phenyl radical then adds selectively to the C3 position of the quinoxalinone ring, forming a key nitrogen-centered radical intermediate. Subsequent single-electron transfer and deprotonation steps restore aromaticity, yielding the desired 3-aryl product. Remarkably, the product itself possesses photosensitive properties similar to the starting material, suggesting a potential autocatalytic effect that sustains the reaction efficiency as conversion progresses.
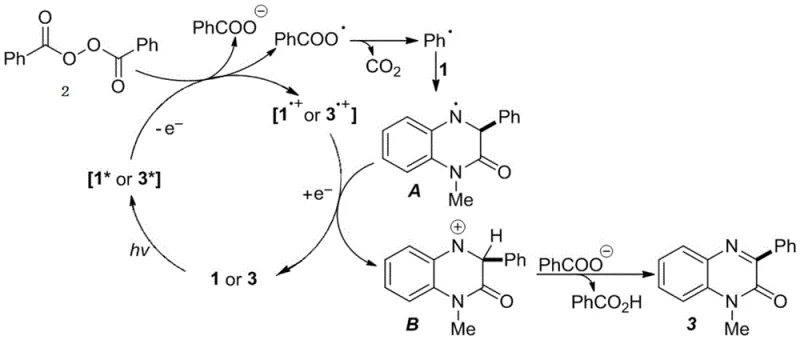
From an impurity control perspective, this mechanism offers distinct advantages over thermal radical initiators. The precise wavelength control ensures that radical generation is spatially and temporally confined to the photon flux, minimizing uncontrolled side reactions such as polymerization or over-oxidation that are common in thermal processes. The use of ethyl acetate as the solvent further stabilizes the radical intermediates, preventing unwanted solvent participation. Additionally, the absence of transition metals eliminates the formation of metal-complexed byproducts, which are notoriously difficult to remove and can catalyze degradation during storage. The high regioselectivity observed at the C3 position is driven by the electronic distribution of the excited quinoxalinone, ensuring that the phenyl radical attacks the most nucleophilic site. This inherent selectivity reduces the burden on chromatographic purification, allowing for simpler crystallization or extraction protocols to achieve high-purity specifications required for clinical grade materials.
How to Synthesize 3-Aryl-N-Methylquinoxalin-2(1H)-one Efficiently
Implementing this photocatalytic protocol requires careful attention to light source specifications and reagent stoichiometry to maximize throughput. The process is designed to be operationally simple, requiring standard glassware and commercially available LEDs, making it accessible for both laboratory scale-up and pilot plant operations. The key to success lies in maintaining the optimal molar ratio of 1:1.5 between the quinoxalinone and the peroxide, along with ensuring the light intensity remains at the sweet spot of 6W to balance reaction rate and energy efficiency. Detailed standard operating procedures regarding mixing speeds, light distance, and workup techniques are essential for reproducibility. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized protocol outlined below.
- Prepare the reaction mixture by dissolving N-methylquinoxalin-2(1H)-one and aryl peroxide in ethyl acetate at a molar ratio of 1: 1.5.
- Irradiate the solution with a 6W LED light source emitting at 415-420 nm wavelength while stirring at room temperature.
- Monitor reaction progress via TLC, then purify the crude product using silica gel column chromatography with petroleum ether/ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology presents a compelling value proposition centered on cost stability and operational simplicity. By removing the dependency on scarce and volatile transition metal catalysts, manufacturers can insulate their production costs from the fluctuations of the precious metals market. The switch to common organic peroxides and standard LED lighting infrastructure significantly lowers the barrier to entry for production, allowing for decentralized manufacturing capabilities if needed. Furthermore, the use of ethyl acetate aligns with increasingly strict environmental regulations regarding solvent emissions, reducing the compliance burden and associated fees. The mild reaction conditions also extend the lifespan of reactor equipment by minimizing corrosion and thermal stress, leading to lower capital expenditure on maintenance and replacement over the long term.
- Cost Reduction in Manufacturing: The elimination of expensive photocatalysts such as Iridium or Ruthenium complexes, which can cost thousands of dollars per gram, results in substantial direct material savings. Additionally, the removal of post-reaction metal scavenging steps reduces the consumption of specialized resins and filtration media. The energy profile of the reaction is also favorable, as low-power LEDs consume significantly less electricity compared to the heating mantles or high-pressure lamps required for traditional thermal or UV methods. These cumulative efficiencies drive down the overall cost of goods sold (COGS), enhancing margin potential for high-volume API intermediates.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically benzoyl peroxide derivatives and N-methylquinoxalinones, are commodity chemicals with robust global supply networks. Unlike specialized ligands or custom-synthesized catalysts that may have long lead times and single-source risks, these reagents are available from multiple vendors worldwide. This diversification mitigates the risk of supply disruptions and allows for more flexible inventory management strategies. The simplicity of the reaction setup also means that production can be easily transferred between different manufacturing sites without the need for highly specialized equipment, ensuring business continuity.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration issues, but the use of flow chemistry or thin-film reactors can effectively address this, allowing for seamless transition from grams to tons. The process generates minimal hazardous waste, primarily consisting of benign organic byproducts and carbon dioxide, simplifying effluent treatment. The absence of heavy metals in the waste stream further reduces the classification of waste as hazardous, lowering disposal costs. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These insights are derived directly from the experimental data and optimization studies detailed in the patent documentation. Understanding these nuances is vital for process engineers and quality assurance teams evaluating the feasibility of this technology for their specific product pipelines. The answers provided reflect the current state of the art as described in the intellectual property.
Q: Does this photocatalytic method require expensive transition metal catalysts?
A: No, the process described in patent CN112028841A is unique because it operates without external photocatalysts or transition metals. The substrate itself acts as the photosensitizer under specific violet light irradiation.
Q: What is the optimal light wavelength for this decarboxylative coupling?
A: Experimental data indicates that a narrow wavelength range of 415-420 nm is critical. Yields drop significantly outside this range, with 6W power being the optimal intensity for balancing efficiency and energy consumption.
Q: Can this method tolerate diverse functional groups on the aryl ring?
A: Yes, the method demonstrates excellent functional group compatibility, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as bromo and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-N-Methylquinoxalin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with advanced photoreactors capable of precisely controlling wavelength and intensity, guaranteeing consistent batch-to-batch quality. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every kilogram of 3-aryl-N-methylquinoxalin-2(1H)-one delivered meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific drug candidates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project's volume and timeline requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the future of green pharmaceutical manufacturing.
