Scalable Synthesis of 3-Oxocyclobutanecarboxylic Acid via Safe Mitsunobu Cyclization for Commercial Production
Scalable Synthesis of 3-Oxocyclobutanecarboxylic Acid via Safe Mitsunobu Cyclization for Commercial Production
The pharmaceutical industry continuously seeks robust and safe synthetic routes for complex intermediates, particularly those serving as scaffolds for antiviral and oncology therapeutics. Patent CN113527081B introduces a transformative preparation method for 3-oxocyclobutanecarboxylic acid, a pivotal building block in modern medicinal chemistry. This innovative protocol replaces hazardous high-temperature conditions with a mild, three-step sequence involving ketone protection, Mitsunobu-mediated cyclization, and controlled acidic hydrolysis. By leveraging 1,3-dihydroxyacetone as an inexpensive starting material, the process achieves high purity and yield while eliminating the severe safety hazards associated with traditional sodium hydride methodologies. For R&D directors and supply chain leaders, this represents a significant leap forward in process reliability and industrial scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclobutane derivatives like 3-oxocyclobutanecarboxylic acid has been plagued by dangerous reaction conditions and poor operational safety profiles. Prior art, such as the method disclosed in US2009/233903 A1, relies heavily on the use of sodium hydride (NaH) as a strong base in dimethylformamide (DMF) solvent. This combination necessitates refluxing temperatures as high as 140°C for extended periods, often exceeding 48 hours, to drive the ring-closure reaction to completion. Such conditions create a substantial risk of thermal runaway and explosion due to the pyrophoric nature of sodium hydride and the high boiling point of DMF, which complicates solvent removal and waste treatment. Furthermore, the harsh basic environment can lead to unwanted side reactions and decomposition of sensitive functional groups, resulting in lower yields and difficult purification challenges that hinder commercial scale-up efforts.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113527081B utilizes a sophisticated Mitsunobu reaction strategy that operates under significantly milder and safer conditions. By employing a triphenylphosphine and azodicarboxylate system, the cyclization proceeds efficiently at temperatures ranging from -5°C to 50°C, completely avoiding the need for pyrophoric bases like sodium hydride. The substitution of DMF with greener and more volatile solvents such as tetrahydrofuran (THF) or 1,4-dioxane facilitates easier solvent recovery and reduces the environmental footprint of the manufacturing process. This novel approach not only enhances the safety profile for plant operators but also streamlines the downstream processing, allowing for faster batch turnover and improved overall equipment effectiveness in a commercial setting.
Mechanistic Insights into Mitsunobu-Mediated Cyclization
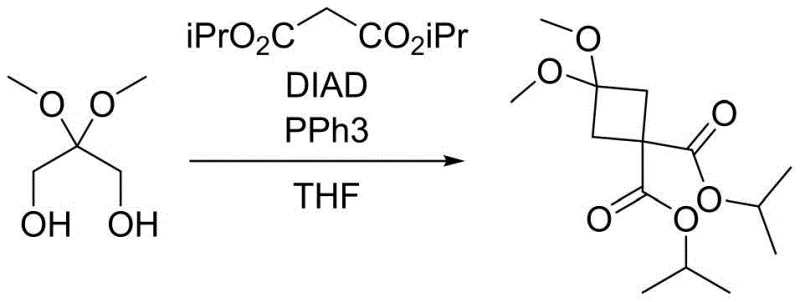
The core of this synthetic innovation lies in the intricate mechanism of the Mitsunobu cyclization, which constructs the strained four-membered cyclobutane ring with high stereochemical control and efficiency. The reaction initiates with the formation of a reactive betaine intermediate between triphenylphosphine and the azodicarboxylate reagent, such as DIAD or DEAD. This activated complex then interacts with the hydroxyl groups of the protected 2,2-dimethoxy-1,3-propanediol, converting them into excellent leaving groups. Simultaneously, the malonic diester, activated by a mild base like triethylamine or potassium tert-butoxide, acts as a nucleophile. The intramolecular nucleophilic attack closes the ring, displacing the triphenylphosphine oxide by-product and forming the stable cyclobutane scaffold. This mechanism is highly advantageous as it avoids the harsh enolate chemistry required in traditional alkylation methods, thereby preserving the integrity of the sensitive ketone protecting group.
Impurity control is meticulously managed through the specific choice of reagents and workup procedures designed to remove the stoichiometric by-product, triphenylphosphine oxide (TPPO). TPPO is notoriously difficult to separate from organic products due to its polarity and solubility characteristics, often requiring extensive chromatography. However, this patent discloses a clever purification tactic where the crude reaction mixture is dissolved in non-polar solvents like n-heptane or petroleum ether. By filtering this solution through a sand core funnel pre-paved with a small amount of silica gel, the polar TPPO and colored impurities are effectively adsorbed and retained, while the desired cyclobutane diester passes through in the filtrate. This simple yet effective solid-phase extraction step dramatically improves the color and purity of the intermediate without the need for expensive column chromatography, ensuring a robust and cost-effective purification workflow suitable for multi-kilogram production.
How to Synthesize 3-Oxocyclobutanecarboxylic Acid Efficiently
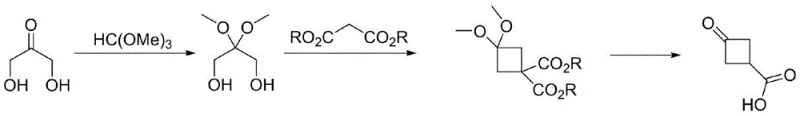
The synthesis of this valuable intermediate is streamlined into three distinct operational stages that maximize yield and minimize hazard. The process begins with the acid-catalyzed protection of 1,3-dihydroxyacetone using trimethyl orthoformate in methanol, followed by the critical Mitsunobu ring-closing step described previously. The final transformation involves the hydrolysis and decarboxylation of the diester intermediate under acidic conditions. To ensure successful implementation, precise control over pH and temperature during the final hydrolysis step is essential to prevent degradation of the cyclobutane ring while ensuring complete removal of the protecting groups. The detailed standardized synthesis steps, including exact molar ratios and specific workup instructions, are provided in the guide below for technical reference.
- Protect 1,3-dihydroxyacetone with trimethyl orthoformate in methanol using p-toluenesulfonic acid catalyst to form 2,2-dimethoxy-1,3-propanediol.
- Perform Mitsunobu cyclization using triphenylphosphine, azodicarboxylate, and malonic diester in THF or dioxane to form the cyclobutane ring.
- Hydrolyze and decarboxylate the diester intermediate under acidic conditions with a phase transfer catalyst to yield the final carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic benefits that extend beyond mere chemical yield. The elimination of hazardous reagents like sodium hydride fundamentally alters the risk profile of the manufacturing facility, potentially lowering insurance premiums and reducing the need for specialized explosion-proof infrastructure. Additionally, the use of common, commodity-grade starting materials such as 1,3-dihydroxyacetone and malonic esters ensures a stable and resilient supply chain, insulating production schedules from the volatility often associated with exotic or custom-synthesized reagents. The simplified purification process further contributes to operational efficiency by reducing solvent consumption and waste generation, aligning with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the removal of expensive and dangerous reagents, leading to substantial cost savings in raw material procurement and handling. By avoiding the use of sodium hydride and high-boiling solvents like DMF, the process eliminates the costly disposal fees and energy-intensive distillation steps required for solvent recovery. Furthermore, the high efficiency of the silica gel filtration trick for removing triphenylphosphine oxide reduces the reliance on preparative HPLC or flash chromatography, which are often bottlenecks in terms of both time and consumable costs. These cumulative efficiencies translate directly into a lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream drug manufacturers. The mild reaction conditions reduce the likelihood of batch failures due to thermal excursions or reagent instability, thereby improving the predictability of delivery timelines. Moreover, the flexibility to use either diethyl or diisopropyl malonate allows for sourcing agility; if one ester becomes scarce or expensive due to market fluctuations, the process can be adapted to use the alternative without compromising the final product quality. This adaptability is a key factor in building a resilient supply chain capable of withstanding global logistical disruptions.
- Scalability and Environmental Compliance: From a scale-up perspective, the transition from laboratory to commercial production is facilitated by the absence of exothermic hazards and the use of standard reactor materials. The process generates less hazardous waste compared to traditional methods, simplifying compliance with environmental protection agencies and reducing the burden on wastewater treatment facilities. The ability to operate at near-atmospheric pressure and moderate temperatures means that existing general-purpose chemical reactors can be utilized without major retrofitting, accelerating the time-to-market for new projects. This ease of scale-up ensures that supply can be rapidly expanded to meet surging demand for antiviral and oncology therapies without compromising safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-oxocyclobutanecarboxylic acid. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and product quality. Understanding these details is crucial for making informed sourcing decisions and integrating this intermediate into complex drug synthesis pipelines.
Q: Why is the Mitsunobu route safer than the conventional NaH method?
A: The conventional method utilizes sodium hydride (NaH) in DMF at high temperatures (140°C), posing significant explosion risks. The patented Mitsunobu route operates at mild temperatures (approx. 50°C) without pyrophoric reagents, drastically improving process safety.
Q: How is triphenylphosphine oxide removed in this process?
A: The process employs a specialized purification technique where the crude product is dissolved in n-heptane and filtered through a sand core funnel paved with a small amount of silica gel. This effectively removes the polar triphenylphosphine oxide by-product, enhancing product color and purity.
Q: What are the primary applications of 3-oxocyclobutanecarboxylic acid?
A: This compound is a critical building block for synthesizing antiviral drugs and kinase inhibitors. The cyclobutane moiety provides unique biological activity, while the carboxyl group allows for condensation with polypeptides and amino acids for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Oxocyclobutanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3-oxocyclobutanecarboxylic acid meets the highest international standards for pharmaceutical applications. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering products that facilitate your R&D success.
We invite you to engage with our technical procurement team to discuss your specific project needs and explore how our optimized manufacturing processes can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of partnering with us. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project starts on a foundation of quality and reliability.
