Scalable Production of FPTX Energetic Materials via Novel Three-Step Nitration Strategy
The landscape of high-energy-density materials is undergoing a significant transformation with the emergence of advanced nitrogen-rich heterocycles that promise to surpass current benchmarks like CL-20. A pivotal development in this sector is detailed in patent CN116284010A, which discloses a revolutionary synthetic methodology for 1,2,9,10-tetranitropyrazolo[1,5-d:5',1'-f][1,2,3,4]tetrazine, commonly known as FPTX. This compound has garnered intense interest due to its exceptional detonation velocity of 9631 m/s and detonation pressure of 44 GPa, properties that rival or exceed those of existing state-of-the-art explosives. Furthermore, FPTX exhibits superior thermal stability with a decomposition temperature of 233°C and lower mechanical sensitivity compared to CL-20, making it an ideal candidate for next-generation propulsion and warhead applications. The patent introduces a streamlined three-step protocol involving direct nitration, amination, and oxidative coupling, which fundamentally addresses the historical challenges of low yields and hazardous multi-step sequences associated with this class of energetic materials.
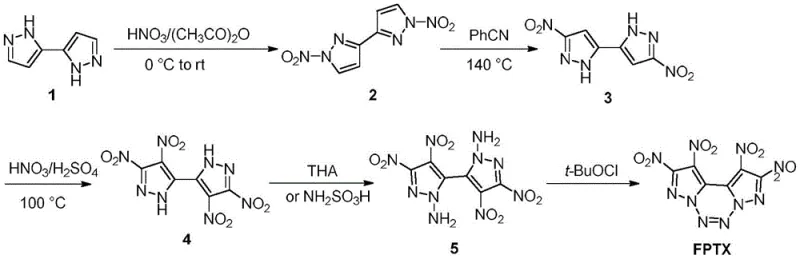
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN116284010A, the synthesis of FPTX was constrained by a convoluted and perilous pathway first reported in literature such as J. Am. Chem. Soc. 2017. As illustrated in the reaction scheme above, the conventional route required a tedious five-step sequence to transform the simple bipyrazole starting material into the final target. A major bottleneck in this legacy process was the construction of the tetranitro intermediate, which necessitated three distinct nitration and nitro-transfer operations. Each of these steps involved the handling of highly energetic and unstable intermediates, requiring rigorous isolation and purification protocols that drastically increased the risk of accidental detonation. Furthermore, the cumulative yield loss across five separate chemical transformations rendered the process economically unviable for large-scale manufacturing, effectively limiting FPTX to small-batch laboratory research rather than practical industrial deployment.
The Novel Approach
In stark contrast to the fragmented legacy workflows, the patented methodology collapses the synthesis into a concise three-step trajectory that maximizes atom economy and operational safety. The cornerstone of this innovation is the ability to achieve direct tetranitration of the bipyrazole scaffold in a single pot, bypassing the need for intermediate nitro-group migrations. By utilizing a robust potassium nitrate and sulfuric acid system, the process efficiently installs four nitro groups simultaneously, creating the critical precursor in one go. This drastic simplification not only accelerates the overall production timeline but also minimizes the number of times hazardous materials must be isolated and transferred between reactors. The subsequent amination and oxidative cyclization steps utilize standard reagents under controlled conditions, ensuring that the entire pathway remains amenable to scale-up while maintaining the high purity required for sensitive energetic applications.
Mechanistic Insights into Direct Tetranitration and Oxidative Coupling
The chemical ingenuity of this process lies primarily in the first step, where the electron-deficient bipyrazole ring undergoes aggressive electrophilic substitution. The use of a KNO3/H2SO4 mixture generates a high concentration of nitronium ions in situ, which are capable of overcoming the deactivating effects of the initial nitro groups to achieve full tetranitration. Maintaining the reaction at 100°C for an extended period (at least 72 hours) ensures complete conversion without the degradation often seen with harsher nitrating agents like fuming nitric acid alone. This thermal activation strategy allows for a cleaner reaction profile with fewer byproducts, directly contributing to a higher quality crude intermediate that requires less downstream purification. The mechanistic efficiency here is critical for R&D teams aiming to replicate the process, as it demonstrates that thermodynamic control can be leveraged to drive difficult substitutions that kinetic control might fail to achieve.
Following the formation of the tetranitro intermediate, the mechanism shifts to nucleophilic aromatic substitution and oxidative dimerization. The amination step replaces specific nitro groups with amino functionalities using reagents like hydroxylamine-O-sulfonic acid, which acts as a potent electrophilic aminating agent in the presence of a base. This transformation sets the stage for the final ring-closing event, where tert-butyl hypochlorite serves as the oxidant. This reagent facilitates the formation of the central tetrazine ring by coupling the amino groups, a process that releases the strain of the precursor and locks the molecule into its final high-energy fused architecture. Understanding these mechanistic nuances is essential for optimizing reaction parameters, such as solvent choice (acetonitrile is preferred) and stoichiometry, to ensure consistent batch-to-batch reproducibility in a commercial setting.
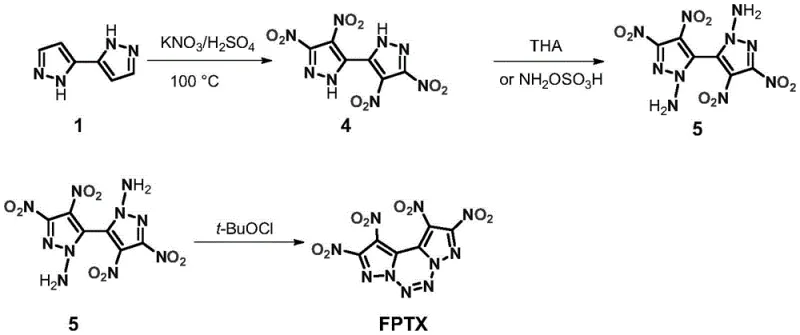
How to Synthesize FPTX Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure both safety and yield. The process begins with the careful preparation of the nitrating mixture at low temperatures before heating to drive the reaction to completion. Following the isolation of the tetranitro intermediate, the subsequent functionalization steps must be performed under anhydrous conditions to prevent hydrolysis of the sensitive nitro-amino species. For a comprehensive breakdown of the specific reagent quantities, addition rates, and workup procedures required to execute this chemistry safely, please refer to the standardized guide below.
- Perform direct tetranitration of 2H,2'H-3,3'-bipyrazole using a KNO3/H2SO4 system at 100°C for over 72 hours to yield 4,4',5,5'-tetranitro-2H,2'H-3,3'-bipyrazole.
- Conduct an amination reaction on the tetranitro intermediate using hydroxylamine-O-sulfonic acid or O-tosylhydroxylamine in acetonitrile with a base.
- Execute the final oxidative coupling using tert-butyl hypochlorite in acetonitrile to cyclize the structure into the target FPTX molecule.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from a five-step to a three-step synthesis represents a substantial opportunity for cost optimization and risk mitigation. The reduction in unit operations directly correlates to lower capital expenditure on processing equipment and reduced consumption of solvents and utilities per kilogram of product. By eliminating two entire synthetic stages, the facility footprint required for production is minimized, and the throughput capacity is inherently increased without the need for additional reactor trains. This efficiency gain is particularly valuable in the energetic materials sector, where regulatory compliance and safety overheads can often dwarf the raw material costs. The streamlined nature of this route ensures that supply chains are less vulnerable to disruptions caused by complex multi-vendor dependencies for specialized intermediates.
- Cost Reduction in Manufacturing: The consolidation of the nitration phase into a single step eliminates the need for expensive nitro-transfer reagents and the associated waste disposal costs. By removing the requirement for multiple isolation and purification cycles for unstable intermediates, the process significantly lowers the labor and energy intensity of the manufacturing campaign. This structural simplification translates to a more favorable cost of goods sold (COGS), making high-performance energetic materials more accessible for broader defense and aerospace applications without compromising on quality specifications.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as potassium nitrate, sulfuric acid, and tert-butyl hypochlorite ensures a robust and resilient supply chain. Unlike legacy methods that may depend on bespoke or hard-to-source catalysts, this protocol utilizes reagents that are readily available in the global chemical market. This accessibility reduces lead times for raw material procurement and mitigates the risk of production stoppages due to supply shortages. Furthermore, the improved stability of the intermediates generated in this route allows for safer storage and transport if semi-finished goods need to be moved between facilities.
- Scalability and Environmental Compliance: The simplified workflow inherently generates less chemical waste, aligning with increasingly stringent environmental regulations governing the production of energetic materials. Fewer reaction steps mean fewer aqueous and organic waste streams to treat, reducing the burden on effluent treatment plants and lowering the overall environmental footprint of the operation. The high safety profile of the direct nitration method also facilitates easier permitting and regulatory approval for scale-up, allowing manufacturers to ramp up production volumes from pilot plant to commercial tonnage with greater confidence and speed.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating FPTX production into their existing portfolios.
Q: How does the new synthesis method improve safety compared to previous routes?
A: The novel method consolidates the synthesis of the key tetranitro intermediate from three hazardous nitration steps into a single direct nitration process. This significantly reduces the number of isolation procedures involving unstable nitro-compounds, thereby minimizing exposure risks and improving overall operational safety during manufacturing.
Q: What are the key advantages of using KNO3/H2SO4 for the nitration step?
A: Utilizing a potassium nitrate and sulfuric acid system allows for a highly efficient direct tetranitration at elevated temperatures (100°C). This approach eliminates the need for complex nitro-transfer reagents and multiple protection/deprotection sequences found in older literature, resulting in a more robust and scalable reaction profile suitable for industrial application.
Q: Is this synthesis route suitable for large-scale production of energetic materials?
A: Yes, the reduction of the synthetic route from five steps to three steps, combined with the use of commercially available reagents like tert-butyl hypochlorite and standard mineral acids, greatly enhances the feasibility of commercial scale-up. The simplified workflow reduces processing time and equipment requirements, addressing previous bottlenecks in FPTX manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable FPTX Supplier
As the demand for high-energy-density materials continues to grow, having a partner with deep technical expertise in energetic material synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced process chemistry to deliver high-purity FPTX and related compounds. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global defense and aerospace contractors. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for sensitive energetic applications, providing our clients with unwavering confidence in our supply.
We invite potential partners to engage with our technical procurement team to discuss how this novel synthesis route can be adapted to your specific needs. By collaborating with us, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this streamlined three-step process. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can secure your supply chain for next-generation energetic materials.
