Revolutionizing Indole Carboglycoside Production: Room-Temperature Synthesis for Commercial API Manufacturing
The Chinese patent CN115433170A introduces a groundbreaking synthetic methodology for indole carboglycoside compounds that addresses critical limitations in traditional approaches while offering significant advantages for commercial pharmaceutical manufacturing. This innovative process utilizes commercially available sugar alkenes as starting materials, enabling a streamlined pathway to these biologically important compounds through a palladium-catalyzed cyclization reaction that proceeds efficiently at room temperature without requiring strict anhydrous or oxygen-free conditions. The method represents a substantial advancement over conventional techniques that typically demand elevated temperatures above 100°C and complex pre-functionalized glycosyl donors, thereby establishing a more practical and scalable route for producing these valuable pharmaceutical intermediates with enhanced operational safety and environmental profile.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for constructing indole carboglycoside skeletons have relied on lithium-indole reactions with sugar lactones or epoxy sugars, which require difficult-to-prepare organolithium reagents and stoichiometric promoters under harsh conditions that create environmental concerns while resulting in lengthy synthetic sequences with limited practical utility. More recent transition metal-catalyzed approaches have employed iodoglucosene as glycosyl donors under palladium catalysis at elevated temperatures around 100°C, but these methods still depend on pre-functionalized sugar derivatives that are not readily available and necessitate strict anhydrous and oxygen-free operational environments that complicate scale-up procedures. The requirement for directing groups in certain methodologies further impacts step economy and atom efficiency, creating additional purification challenges that increase production costs and reduce overall yield consistency across different substrate types.
The Novel Approach
The patented methodology overcomes these limitations by employing commercially accessible sugar alkenes (glucosene, galactosene, or rhamnosene) in combination with o-alkynyl anilines under palladium catalysis at ambient temperature conditions. This innovative approach eliminates the need for pre-functionalized glycosyl donors while operating effectively without stringent anhydrous or oxygen-free requirements, significantly simplifying process implementation across diverse manufacturing environments. The reaction demonstrates excellent substrate universality across various functional groups and maintains strong stereoselectivity without requiring additional resolution steps, making it particularly suitable for industrial-scale production of pharmaceutical intermediates where consistent quality and operational simplicity are paramount considerations for both R&D and manufacturing teams.
Mechanistic Insights into Palladium-Catalyzed Cyclization
The reaction mechanism begins with coordination between o-alkynyl aniline and active divalent palladium iodide to form intermediate A, followed by intramolecular nucleophilic cyclization generating intermediate B. This intermediate then undergoes stereoselective addition with the double bond in sugar alkenes to produce intermediate C, which subsequently eliminates through ortho heteroatom elimination to yield the final 2,3-position unsaturated indole carboglycoside products. This catalytic cycle operates efficiently at room temperature due to the optimized combination of palladium catalyst (5%-15% molar amount), base (molar ratio 1-3:1), additive (molar ratio 1-3:1), and DMF solvent system that facilitates smooth progression through each mechanistic step without requiring thermal activation that would otherwise be necessary for similar transformations.
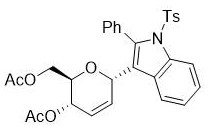
The process demonstrates exceptional control over stereochemistry through the stereoselective addition step between intermediate B and the sugar alkene double bond, ensuring consistent configuration in the final product without requiring additional chiral auxiliaries or resolution techniques. This inherent stereocontrol is particularly valuable for pharmaceutical applications where specific stereoisomers are required for biological activity, as it eliminates costly separation steps while maintaining high purity standards essential for API intermediate production. The mild reaction conditions also prevent common side reactions such as epimerization or decomposition that frequently occur in high-temperature processes, resulting in cleaner product profiles with fewer impurities that would otherwise require extensive purification.
How to Synthesize Indole Carboglycosides Efficiently
This patented methodology represents a significant advancement in indole carboglycoside synthesis by enabling room-temperature operation with commercially available starting materials while maintaining excellent yield and selectivity across diverse substrates. The following standardized procedure details the optimized conditions developed through extensive experimentation that ensure reliable production of high-purity indole carboglycoside intermediates suitable for pharmaceutical applications.
- Combine o-alkynyl aniline (0.2 mmol), sugar alkene (0.4 mmol), palladium catalyst (0.02 mmol), base (0.4 mmol), and additive (0.4 mmol) in a clean reactor under ambient conditions without requiring strict anhydrous or oxygen-free environments.
- Add organic solvent (2 mL DMF) to the reaction mixture and stir at room temperature for 5-10 hours, monitoring reaction progress through standard analytical techniques without requiring specialized equipment.
- After completion, extract the reaction mixture three times with ethyl acetate and water, dry the combined organic phases over anhydrous sodium sulfate, filter, concentrate under reduced pressure, and purify by column chromatography using petroleum ether/ethyl acetate (5: 1) as eluent.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals in the pharmaceutical industry by transforming what was previously a complex multi-step process requiring specialized equipment into a streamlined room-temperature operation using readily available materials. The elimination of high-temperature requirements removes a major bottleneck in production scheduling while significantly reducing energy consumption and associated operational costs across manufacturing facilities.
- Cost Reduction in Manufacturing: The utilization of commercially available sugar alkenes instead of pre-functionalized glycosyl donors eliminates multiple synthetic steps required for precursor preparation, substantially reducing raw material costs while avoiding expensive metal scavenging procedures previously needed with transition metal catalysts. This streamlined approach creates significant cost savings through reduced processing time, lower energy consumption from room-temperature operation, and minimized waste generation compared to conventional high-temperature methods.
- Enhanced Supply Chain Reliability: The use of readily available starting materials with no specialized storage requirements ensures consistent supply availability while eliminating dependencies on niche chemical suppliers that often create production bottlenecks. The simplified process with minimal equipment requirements enables rapid technology transfer between manufacturing sites and facilitates quick scale-up from laboratory to commercial production without requiring specialized infrastructure investments.
- Scalability and Environmental Compliance: The room-temperature operation significantly reduces energy consumption while eliminating hazardous high-temperature processing steps that require special safety protocols. The simplified waste stream from this process contains fewer toxic byproducts compared to traditional methods, making waste treatment more straightforward and environmentally friendly while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement specialists, R&D scientists, and supply chain managers regarding implementation of this innovative synthesis methodology for indole carboglycoside production. These answers are based on extensive experimental validation documented in the patent literature.
Q: How does this room-temperature synthesis method improve supply chain reliability compared to conventional approaches?
A: The elimination of high-temperature requirements (previously needing >100°C) removes a critical bottleneck in production scheduling and equipment availability. This room-temperature process can be implemented across diverse manufacturing facilities without specialized heating systems, significantly reducing lead times for high-purity pharmaceutical intermediates while maintaining consistent quality through simplified process control parameters.
Q: What specific cost reduction benefits does this method offer for API intermediate manufacturing?
A: By utilizing commercially available sugar alkenes instead of pre-functionalized glycosyl donors requiring complex preparation, this method eliminates multiple synthetic steps and associated purification costs. The room-temperature operation reduces energy consumption while avoiding expensive metal scavenging procedures previously needed with transition metal catalysts, creating substantial cost savings in commercial scale-up of complex pharmaceutical intermediates.
Q: How does this synthesis method address purity concerns for pharmaceutical applications?
A: The mild reaction conditions prevent thermal degradation pathways common in high-temperature processes, resulting in cleaner reaction profiles with fewer side products. The stereoselective nature of the palladium-catalyzed cyclization produces consistent stereochemistry without requiring additional resolution steps, ensuring stringent purity specifications required for pharmaceutical intermediates while maintaining excellent functional group compatibility across diverse substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Carboglycoside Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical capabilities. As a CDMO expert specializing in challenging heterocyclic syntheses, we have successfully implemented this patented room-temperature methodology across multiple client projects with consistent quality outcomes that meet or exceed regulatory requirements for pharmaceutical applications.
For immediate technical support, please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your production needs along with detailed COA data and route feasibility assessments demonstrating how this innovative methodology can optimize your supply chain for indole carboglycoside intermediates.
