Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Applications
Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A significant breakthrough in this domain is detailed in patent CN113105402B, which discloses a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These heterocyclic scaffolds are critical structural motifs found in numerous high-value active pharmaceutical ingredients (APIs), including blockbuster drugs like Sitagliptin, Maraviroc, and Deferasirox. The introduction of a trifluoromethyl group into these structures is particularly valuable, as it enhances metabolic stability, lipophilicity, and bioavailability, properties that are essential for modern drug design. This patent presents a robust methodology that addresses long-standing challenges in heterocyclic chemistry, offering a pathway that is not only chemically elegant but also commercially viable for large-scale production.
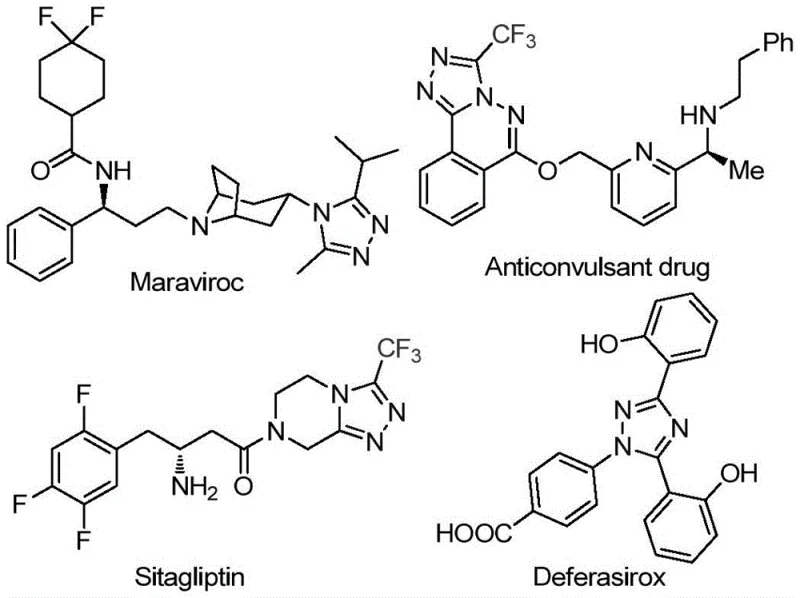
The significance of this technology extends beyond mere academic interest; it represents a tangible opportunity for reliable pharmaceutical intermediate suppliers to optimize their portfolios. By leveraging a metal-free catalytic system, manufacturers can bypass the stringent regulatory hurdles associated with heavy metal residues, a common bottleneck in API synthesis. The process utilizes dimethyl sulfoxide (DMSO) as both a solvent and a reactant promoter, facilitating a tandem reaction sequence that constructs the triazole ring with high efficiency. For procurement managers and supply chain directors, the implication is clear: a reduction in raw material complexity and a streamlining of the purification process, which directly translates to improved margins and supply security.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, especially those bearing both trifluoromethyl and acyl groups, has been fraught with difficulties. Traditional methodologies often rely on the use of expensive transition metal catalysts, such as copper or palladium complexes, which necessitate rigorous exclusion of moisture and oxygen to prevent catalyst deactivation. These苛刻 conditions require specialized equipment, such as gloveboxes or Schlenk lines, driving up capital expenditure and operational costs. Furthermore, the removal of trace heavy metals from the final product to meet pharmacopeial standards involves additional purification steps, such as scavenging resins or extensive chromatography, which drastically reduce overall yield and increase waste generation. The limited substrate scope of many older methods also restricts the ability to introduce diverse functional groups, hindering the rapid exploration of structure-activity relationships (SAR) during drug discovery phases.
The Novel Approach
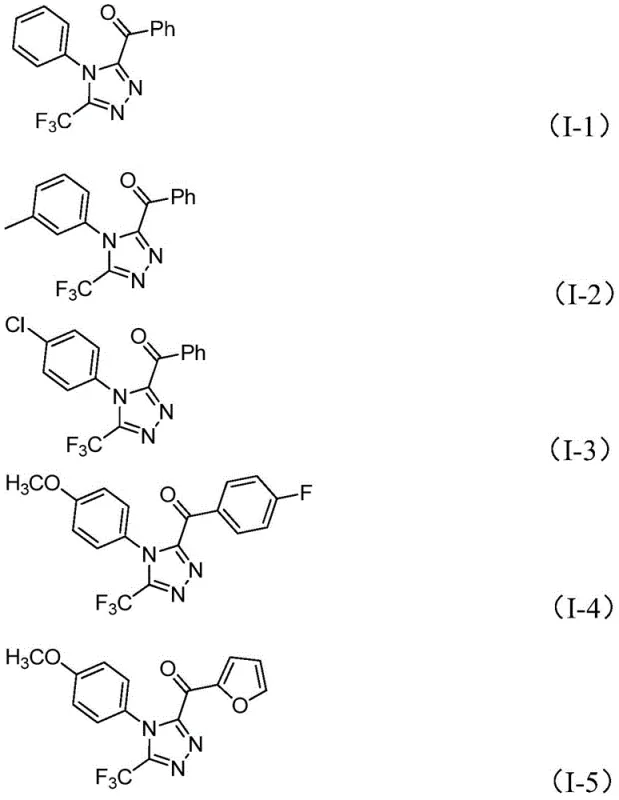
In stark contrast, the method disclosed in patent CN113105402B offers a paradigm shift by employing elemental iodine as a non-metal promoter in a DMSO medium. This approach eliminates the need for anhydrous and oxygen-free environments, allowing reactions to proceed under ambient atmospheric conditions, which dramatically simplifies operational protocols. The reaction tolerates a wide range of functional groups on both the aryl ketone and the hydrazide components, enabling the synthesis of a diverse library of triazole derivatives. As illustrated in the specific examples provided in the patent, substrates with electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like chloro and fluoro, are all compatible, yielding products with respectable efficiency. This versatility makes the process an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing, as it reduces the dependency on exotic reagents and complex engineering controls.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The chemical elegance of this synthesis lies in its tandem mechanism, which effectively combines oxidation and cyclization in a single pot. The process initiates with the iodine-promoted iodination and subsequent Kornblum oxidation of the aryl ethyl ketone in DMSO. This step generates an reactive aryl diketone intermediate in situ, which serves as the electrophilic partner for the subsequent condensation. The trifluoroethylimide hydrazide then attacks this activated carbonyl species, undergoing dehydration to form a hydrazone intermediate. This sequence is crucial because it avoids the isolation of unstable diketone intermediates, thereby improving safety and throughput. The presence of pyridine and sodium dihydrogen phosphate acts as a buffer system, maintaining the optimal pH for the condensation while neutralizing the hydrogen iodide byproduct generated during the oxidation phase.
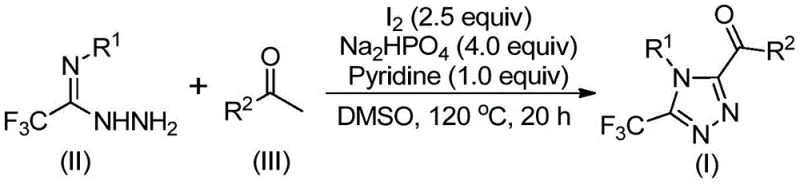
Following the formation of the hydrazone, the reaction mixture is heated to higher temperatures (110-130°C) to drive the intramolecular cyclization. Under these thermal conditions, and promoted by the continued presence of iodine, the hydrazone undergoes an oxidative cyclization to close the 1,2,4-triazole ring. The mechanism likely involves the formation of an iodonium species that facilitates the nucleophilic attack of the nitrogen atom onto the adjacent carbon, followed by elimination to restore aromaticity. This metal-free oxidative cyclization is particularly advantageous for impurity control, as it minimizes the formation of metal-complexed side products that are difficult to separate. The result is a cleaner reaction profile, which simplifies downstream processing and ensures that the final high-purity 1,2,4-triazole derivatives meet the stringent quality specifications required for clinical applications.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
Implementing this synthesis route requires careful attention to the two-stage heating protocol to maximize conversion and minimize byproduct formation. The initial stage focuses on the activation of the ketone, while the second stage drives the ring closure. Operators must ensure that the stoichiometry of iodine and base is maintained within the optimized ranges specified in the patent to prevent over-oxidation or incomplete cyclization. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to guide process development teams in replicating these results.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate oxidation.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iodine-promoted synthesis offers profound advantages for supply chain resilience and cost management. The primary driver of value is the substitution of expensive, supply-constrained transition metal catalysts with elemental iodine, a commodity chemical that is abundant and inexpensive. This shift not only lowers the direct material cost but also mitigates the risk of supply disruptions associated with precious metals. Furthermore, the elimination of heavy metals removes the necessity for specialized metal scavenging resins and the associated validation testing, leading to substantial cost savings in the purification stage. The simplified operational requirements, specifically the lack of need for inert atmosphere techniques, allow for the utilization of standard glass-lined reactors, reducing capital investment barriers for scale-up.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the drastic simplification of the reaction setup and workup. By avoiding the use of noble metal catalysts, manufacturers eliminate the high cost of catalyst procurement and the complex recovery processes often required to make their use economically feasible. Additionally, the ability to run the reaction without strict anhydrous conditions reduces the energy consumption associated with solvent drying and nitrogen purging. The high atom economy of the tandem reaction means that fewer raw materials are wasted, and the overall yield per batch is optimized, directly impacting the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available starting materials such as aryl ethyl ketones and hydrazides, which are produced by numerous global chemical suppliers. This commoditization of raw materials reduces dependency on single-source vendors and provides procurement teams with greater negotiating leverage. The robustness of the reaction conditions also means that the process is less susceptible to variations in environmental factors, ensuring consistent batch-to-bquality. This reliability is critical for maintaining steady production schedules and meeting the just-in-time delivery demands of downstream API manufacturers.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is significantly lower than traditional methods, aligning with the growing industry emphasis on green chemistry. The absence of toxic heavy metals simplifies waste treatment and disposal, reducing the regulatory burden and costs associated with hazardous waste management. The process has been demonstrated to be scalable to the gram level with ease, suggesting a smooth path to kilogram and ton-scale production. This scalability ensures that the technology can support the commercial scale-up of complex pharmaceutical intermediates without requiring fundamental process redesigns, facilitating a faster time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their production pipelines.
Q: Does this synthesis method require expensive heavy metal catalysts?
A: No, the patented process utilizes elemental iodine as a non-metal promoter, eliminating the need for costly transition metal catalysts and the subsequent complex removal steps required for pharmaceutical grade purity.
Q: What are the reaction conditions regarding moisture and oxygen sensitivity?
A: Unlike many traditional heterocyclic syntheses, this method does not require strict anhydrous or oxygen-free conditions, significantly simplifying operational requirements and reducing equipment costs for large-scale production.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly states that the method can be easily expanded to the gram level and beyond, utilizing cheap and readily available starting materials like aryl ketones, making it highly viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our team of expert chemists has extensively evaluated the technology described in patent CN113105402B and possesses the capability to translate this laboratory-scale innovation into robust commercial processes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of triazole intermediate meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical team to explore how this cost-effective synthesis route can be integrated into your specific project requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, helping you identify opportunities for margin improvement. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, taking the first step towards a more efficient and reliable supply partnership.
