Scalable Silver-Catalyzed Synthesis of 1,3-Diarylpropynones for Advanced Pharmaceutical Manufacturing
Scalable Silver-Catalyzed Synthesis of 1,3-Diarylpropynones for Advanced Pharmaceutical Manufacturing
The landscape of organic synthesis for complex ketone intermediates is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective methodologies in the pharmaceutical sector. Patent CN108276268B introduces a groundbreaking preparation method for 1,3-diarylpropynones, utilizing a silver-catalyzed decarboxylative coupling strategy that fundamentally alters the economic and operational parameters of production. This innovative approach leverages alpha-keto acids and aryl alkynoic acids as stable, inexpensive building blocks, replacing hazardous and costly traditional reagents. By employing a catalytic amount of silver salt alongside a cheap inorganic oxidant, the process achieves high yields under remarkably mild conditions, typically around 50°C. This technological leap not only enhances safety profiles by eliminating toxic gases but also streamlines the supply chain for critical pharmaceutical intermediates. For R&D directors and procurement managers alike, this represents a pivotal shift towards sustainable and economically viable manufacturing protocols.
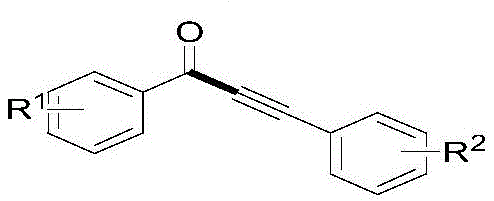
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-diarylpropynones has relied heavily on transition metal-catalyzed coupling reactions involving acyl halides and terminal alkynes, or the carbonylation of aryl halides. These conventional pathways are fraught with significant logistical and safety challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. The use of acyl halides is particularly problematic due to their inherent instability and sensitivity to moisture, necessitating stringent storage conditions and specialized handling equipment that drive up operational costs. Furthermore, traditional Sonogashira-type carbonylation reactions often require the use of gaseous carbon monoxide, a colorless, odorless, and highly toxic substance that poses severe safety risks in a manufacturing environment. The reliance on palladium-copper bimetallic catalytic systems further exacerbates cost issues, as palladium salts are expensive and require rigorous removal processes to meet the strict heavy metal limits imposed by regulatory bodies for high-purity API intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN108276268B offers a robust alternative that circumvents these historical bottlenecks through a radical decarboxylative mechanism. By utilizing free carboxylic acids as radical precursors, the process capitalizes on their low toxicity, high stability, and commercial availability, effectively reducing raw material costs and supply chain volatility. The reaction proceeds smoothly in a mixed solvent system of DMSO and water, utilizing silver acetate as a cost-effective catalyst and ammonium persulfate as a benign oxidant. This shift eliminates the need for toxic carbon monoxide and unstable acyl halides, thereby simplifying the engineering controls required for production. The mild reaction temperature of 50°C significantly reduces energy consumption compared to high-temperature alternatives, while the excellent functional group tolerance ensures that diverse substrates can be processed without extensive protection-deprotection sequences. This results in a streamlined workflow that is ideally suited for a reliable pharmaceutical intermediate supplier aiming to optimize production efficiency.
Mechanistic Insights into Silver-Catalyzed Decarboxylative Coupling
The core of this synthetic breakthrough lies in the intricate radical mechanism facilitated by the silver catalyst and the persulfate oxidant. The reaction initiates with the oxidative decarboxylation of the alpha-keto acid, where the silver species interacts with the carboxylate group to generate an acyl radical intermediate. This step is critical as it bypasses the need for pre-functionalized acylating agents, directly accessing the reactive acyl species from stable precursors. Simultaneously, the aryl alkynoic acid undergoes a similar decarboxylative activation or acts as a radical acceptor, depending on the specific pathway nuances, ultimately leading to the formation of the carbon-carbon triple bond linkage. The use of ammonium persulfate serves as a potent single-electron oxidant, regenerating the active silver species and driving the catalytic cycle forward without the accumulation of inhibitory byproducts. This mechanistic elegance allows for the construction of the 1,3-diarylpropynone scaffold with high atom economy and minimal waste generation.
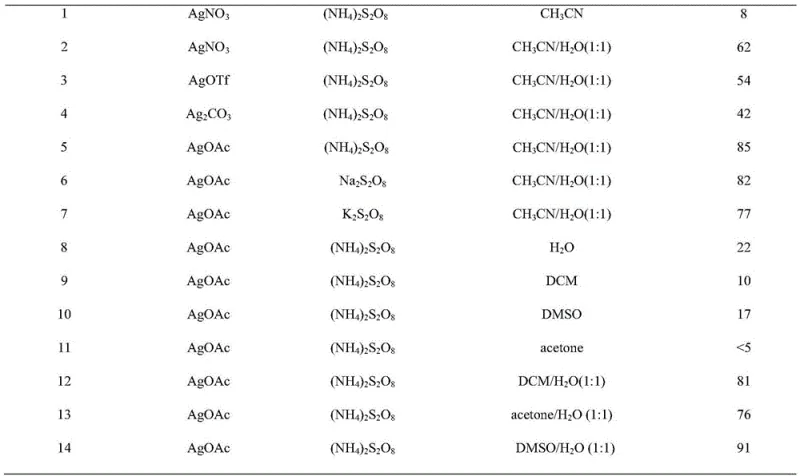
From an impurity control perspective, this radical pathway offers distinct advantages over ionic mechanisms often seen in traditional coupling reactions. The mild conditions prevent the degradation of sensitive functional groups that might otherwise decompose under harsh acidic or basic environments. For instance, electron-withdrawing groups such as nitro and cyano substituents, as well as electron-donating groups like methoxy and alkyl chains, are well-tolerated, as evidenced by the broad substrate scope demonstrated in the patent data. The selectivity of the silver catalyst minimizes side reactions such as homocoupling of the alkyne or over-oxidation of the ketone moiety. This high level of chemoselectivity translates directly to a cleaner crude reaction profile, reducing the burden on downstream purification processes. For quality assurance teams, this means a more consistent impurity profile and a higher likelihood of meeting stringent purity specifications without requiring multiple recrystallization steps, thereby enhancing the overall yield and throughput of the manufacturing process.
How to Synthesize 1,3-Diarylpropynone Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and reproducibility, leveraging the robust conditions outlined in the patent. The process begins by dispersing the alpha-keto acid and aryl alkynoic acid in a specific ratio, typically slightly excess keto acid to ensure complete conversion of the alkyne source. The addition of the silver catalyst and oxidant must be managed carefully to maintain the catalytic cycle without inducing rapid exotherms, although the reaction is generally温和 (mild). The choice of solvent, a 1:1 mixture of DMSO and water, is crucial for solubilizing both the organic substrates and the inorganic oxidant, creating a homogeneous reaction environment that facilitates efficient mass transfer. Detailed standardized synthesis steps follow below, ensuring that technical teams can replicate the high yields reported in the literature.
- Disperse alpha-keto acid, aryl alkynoic acid, AgOAc catalyst, and ammonium persulfate oxidant in a DMSO and water mixed solvent system.
- Stir the reaction mixture at 50°C for 3 hours under an air atmosphere to facilitate the decarboxylative coupling.
- Extract the product with diethyl ether, concentrate under reduced pressure, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver-catalyzed decarboxylative coupling method presents a compelling value proposition centered on cost stability and operational simplicity. The elimination of expensive palladium catalysts and toxic carbon monoxide gas removes two of the most significant cost drivers and safety liabilities associated with traditional synthesis routes. By switching to stable carboxylic acid feedstocks, the supply chain becomes more resilient, as these materials are commodity chemicals with reliable global availability, unlike specialized acyl halides which may have limited suppliers and longer lead times. This transition effectively de-risks the sourcing strategy for key intermediates, ensuring continuity of supply even in volatile market conditions. Furthermore, the simplified workup procedure, involving basic extraction and chromatography, reduces the demand for specialized waste treatment facilities required for heavy metal removal, contributing to substantial cost savings in environmental compliance.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the substitution of high-cost reagents with commodity chemicals. Replacing palladium salts with silver acetate drastically lowers the catalyst cost, while the use of ammonium persulfate as an oxidant is significantly cheaper than specialized organic oxidants. Additionally, the avoidance of acyl halides eliminates the need for their synthesis or purchase, which often involves additional processing steps and hazardous reagents like thionyl chloride. The mild reaction conditions also translate to lower energy costs, as heating to 50°C is far less energy-intensive than the high temperatures required for many traditional coupling reactions. These factors combine to create a manufacturing process with a significantly reduced cost of goods sold (COGS), allowing for more competitive pricing in the final pharmaceutical intermediates market.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous starting materials greatly enhances the reliability of the supply chain. Carboxylic acids are generally solids with long shelf lives and do not require the specialized containment or transport regulations associated with corrosive acyl halides or toxic gases. This simplifies logistics and reduces the risk of shipment delays due to regulatory compliance issues. Moreover, the robustness of the reaction means that slight variations in raw material quality are less likely to result in batch failures, providing a buffer against supply fluctuations. For supply chain planners, this predictability is invaluable, enabling more accurate forecasting and inventory management, ultimately reducing lead time for high-purity intermediates reaching the production line.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by its homogeneous nature and lack of hazardous gas evolution. The absence of carbon monoxide removes the need for complex gas handling infrastructure, making the technology transfer to large-scale reactors straightforward and safe. The aqueous solvent component aligns with green chemistry principles, reducing the volume of organic solvents required and simplifying wastewater treatment. The byproduct of the decarboxylation is carbon dioxide, which is easily vented, minimizing solid waste generation. This environmental friendliness not only reduces disposal costs but also aligns with the increasing regulatory pressure on pharmaceutical manufacturers to adopt sustainable practices, ensuring long-term viability and compliance with global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the feasibility of this method for their specific applications. Understanding these nuances is essential for making informed decisions about process adoption and optimization.
Q: What are the advantages of this silver-catalyzed method over traditional Sonogashira coupling?
A: This method avoids the use of toxic carbon monoxide gas and expensive palladium-copper bimetallic systems. It utilizes stable carboxylic acid precursors instead of unstable acyl halides, operating under mild conditions with excellent functional group tolerance.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses cheap and readily available reagents like silver acetate and ammonium persulfate. The reaction conditions are mild (50°C) and the workup involves simple extraction and chromatography, making it highly amenable to scale-up.
Q: What is the functional group compatibility of this reaction?
A: The reaction demonstrates broad compatibility, tolerating substituents such as halogens (F, Cl, Br), nitro groups, esters, ethers, and heterocycles like quinoline and thiophene without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Diarylpropynone Supplier
The technological potential of this silver-catalyzed decarboxylative coupling route is immense, offering a pathway to high-quality intermediates that meet the rigorous demands of modern drug discovery and development. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that consistency is key in pharmaceutical manufacturing, and our processes are designed to deliver high-purity 1,3-diarylpropynones with minimal batch-to-batch variation, supporting your critical timelines.
We invite you to explore how this innovative synthesis method can optimize your supply chain and reduce overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable supply of advanced intermediates backed by deep chemical expertise and a dedication to operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
