Advanced Synthesis of Chiral Bis-imidazolinylbenzene Compounds for High-Performance Catalysis
Advanced Synthesis of Chiral Bis-imidazolinylbenzene Compounds for High-Performance Catalysis
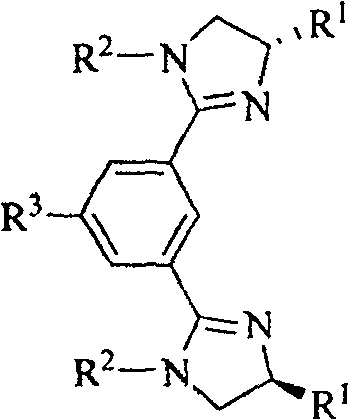
The landscape of asymmetric catalysis has been revolutionized by the development of novel chiral ligands, with bis-imidazolinylbenzene compounds emerging as a critical class of structures for high-value pharmaceutical and fine chemical synthesis. Patent CN100478333C discloses a groundbreaking methodology for producing both chiral and non-chiral variants of these compounds, addressing long-standing challenges in ligand accessibility and structural diversity. This technology leverages a robust synthetic pathway that transforms readily available bis-amido alcohols into complex heterocyclic systems through a streamlined thionyl chloride-mediated cyclization process. For R&D directors and procurement specialists, this represents a significant opportunity to secure reliable pharmaceutical intermediates supplier partnerships that can deliver high-purity ligands without the prohibitive costs associated with traditional diamine-based routes. The versatility of the general formula, allowing for variations in R1, R2, and R3 substituents, ensures that these compounds can be tailored for specific catalytic applications ranging from hydrogenation to carbon-carbon bond formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-imidazoline and related bis-oxazoline ligands has relied heavily on the condensation of dicarboxylic acid derivatives with chiral diamines. This conventional approach presents severe bottlenecks for commercial scale-up of complex polymer additives and fine chemicals, primarily due to the scarcity and exorbitant cost of enantiomerically pure diamines. The preparation of chiral diamines often involves multi-step resolutions or asymmetric syntheses that drastically inflate the overall production cost and extend lead times. Furthermore, the reactivity of diamines can sometimes lead to oligomerization or incomplete cyclization, resulting in difficult-to-remove impurities that compromise the enantioselectivity of the final metal complex catalysts. These factors collectively hinder the widespread industrial adoption of such ligands, forcing process chemists to seek alternative, more economically viable precursors that do not sacrifice stereochemical integrity or catalytic performance.
The Novel Approach
The methodology outlined in the patent data introduces a paradigm shift by utilizing cheap and easy-to-obtain amino alcohols as the foundational building blocks instead of diamines. This strategic substitution fundamentally alters the economic equation of ligand manufacturing, providing a simple and feasible method for synthesizing bis-imidazoline compounds on a kilogram to ton scale. By reacting bis-amido alcohols with thionyl chloride under reflux conditions, the process efficiently activates the hydroxyl groups for subsequent intramolecular cyclization upon treatment with amines. This route not only bypasses the need for expensive diamine starting materials but also offers superior control over the reaction trajectory, minimizing side reactions. The ability to introduce diverse substituents at the R3 position, such as hydroxyl, nitro, or amino groups, further enhances the utility of these compounds by enabling downstream functionalization or immobilization, thereby facilitating the research and wide application of chiral imidazoline compounds in continuous flow processes.
Mechanistic Insights into Thionyl Chloride-Mediated Cyclization
The core of this synthetic innovation lies in the activation of the bis-amido alcohol intermediate using thionyl chloride (SOCl2), which serves as both a chlorinating agent and a dehydrating promoter. In this mechanism, the hydroxyl groups of the starting material are converted into good leaving groups (chlorides or chlorosulfites), which are then displaced by the nitrogen nucleophiles introduced in the second step. The reaction conditions specify a molar ratio of bis-amido alcohol to thionyl chloride between 1:8 and 1:20, ensuring complete conversion and driving the equilibrium towards the activated intermediate. Refluxing for 4 to 10 hours allows for thorough activation, after which the excess thionyl chloride is distilled off to prevent interference with the subsequent amine addition. This careful control of stoichiometry and thermal energy is crucial for maintaining the integrity of the chiral centers, ensuring that the optical purity of the starting amino alcohol is preserved in the final bis-imidazoline product.
Following the activation step, the addition of triethylamine and the chosen amine (aromatic or aliphatic) triggers the ring-closing event that forms the imidazoline heterocycles. The triethylamine acts as an acid scavenger, neutralizing the HCl generated during the cyclization and preventing the protonation of the amine nucleophile, which would render it unreactive. Stirring at room temperature for at least 5 hours provides sufficient time for the nucleophilic attack and ring closure to proceed to completion without the need for harsh heating that could racemize sensitive chiral centers. The resulting crude product is then subjected to a standard aqueous workup involving sodium hydroxide extraction, drying, and concentration. Purification via thin-layer chromatography or column chromatography yields the target bis-imidazolinylbenzene compounds with high purity, ready for coordination with transition metals to form active catalysts for asymmetric synthesis.
How to Synthesize Bis-imidazolinylbenzene Efficiently
To implement this synthesis effectively in a pilot or production environment, strict adherence to the moisture-free conditions during the thionyl chloride reflux is paramount to prevent hydrolysis of the activated intermediate. The process begins with the preparation of the bis-amido alcohol precursor, which can be synthesized from chiral or achiral amino acids via reduction and subsequent acylation, ensuring a steady supply of high-quality starting material. Once the bis-amido alcohol is secured, the cyclization protocol described in the patent offers a robust framework for generating diverse libraries of ligands by simply varying the amine component. Detailed standardized synthesis steps see the guide below for precise operational parameters regarding temperature control, addition rates, and workup procedures that maximize yield and minimize waste generation.
- Reflux bis-amido alcohol in thionyl chloride (molar ratio 1: 8-20) for 4-10 hours to activate the hydroxyl groups.
- Remove excess thionyl chloride, dissolve the residue in ether, and filter to remove insolubles before adding triethylamine and the chosen amine.
- Stir the mixture at room temperature for at least 5 hours, followed by aqueous workup with sodium hydroxide and purification via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this amino alcohol-based synthesis route offers profound strategic benefits that extend beyond mere technical feasibility. The primary driver for adoption is the drastic simplification of the raw material supply chain, as amino alcohols are commodity chemicals available from multiple global vendors, unlike specialized chiral diamines which may have single-source dependencies. This diversification of supply sources significantly mitigates the risk of production stoppages due to raw material shortages, ensuring enhanced supply chain reliability for critical catalytic components. Furthermore, the elimination of complex diamine synthesis steps reduces the overall number of unit operations required, leading to lower capital expenditure on equipment and reduced energy consumption per kilogram of product. These efficiencies translate directly into cost reduction in fine chemical manufacturing, allowing end-users to access high-performance chiral ligands at a price point that makes their use in large-scale API production economically justifiable.
- Cost Reduction in Manufacturing: The substitution of expensive chiral diamines with inexpensive amino alcohols fundamentally lowers the bill of materials, which is often the largest cost component in fine chemical synthesis. By removing the need for costly resolution steps or asymmetric syntheses required to produce diamines, the overall production cost is significantly reduced without compromising the quality of the final ligand. Additionally, the use of common solvents like ether and dichloromethore, along with standard reagents like thionyl chloride and triethylamine, ensures that the process remains compatible with existing infrastructure, avoiding the need for specialized reactors or hazardous handling protocols that would drive up operational expenses.
- Enhanced Supply Chain Reliability: Relying on widely available amino alcohols rather than niche diamines creates a more resilient supply chain capable of withstanding market fluctuations and geopolitical disruptions. Since amino alcohols are produced in vast quantities for various industries, their availability is stable, and lead times are typically short, allowing for just-in-time manufacturing strategies. This reliability is crucial for pharmaceutical companies that require consistent quality and timely delivery of intermediates to meet regulatory filing deadlines and commercial launch schedules. The robustness of the synthesis method also means that scaling from gram to kilogram quantities can be achieved with minimal re-optimization, further securing the supply continuity for long-term projects.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, utilizing standard unit operations such as reflux, distillation, and liquid-liquid extraction that are well-understood in chemical engineering. The process avoids the use of heavy metal catalysts in the ligand synthesis itself, reducing the burden of metal removal and waste treatment in the early stages of production. While thionyl chloride requires careful handling, its byproducts (SO2 and HCl) can be effectively scrubbed and managed using standard abatement systems, ensuring compliance with environmental regulations. The ability to introduce functional groups for immobilization also supports green chemistry initiatives by enabling catalyst recycling, which minimizes solvent usage and waste generation in the downstream catalytic applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bis-imidazolinylbenzene compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is essential for evaluating the feasibility of integrating these ligands into your current catalytic processes and for assessing the potential return on investment from switching to this more efficient synthetic methodology.
Q: What is the primary advantage of using amino alcohols over diamines in this synthesis?
A: The primary advantage is cost and availability. Chiral diamines are often expensive and difficult to prepare, whereas chiral amino alcohols are cheap, readily available starting materials that simplify the supply chain for ligand production.
Q: How does the imidazoline structure compare to bisoxazoline ligands?
A: The introduction of the NR group in the imidazoline ring alters the electronic and steric effects compared to the oxygen in oxazolines. This modification is expected to improve cis-selectivity in various asymmetric catalytic reactions.
Q: Can these compounds be immobilized for catalyst recycling?
A: Yes, the R3 substituent can be functional groups like hydroxyl or amino, which facilitates immobilization on polymeric or insoluble carriers, allowing for easy separation and recycling of the catalyst system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-imidazolinylbenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral ligands play in the development of next-generation pharmaceuticals and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early discovery phase or full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of bis-imidazolinylbenzene compounds meets the exacting standards required for asymmetric catalysis. Our commitment to quality assurance means that you can rely on us as a trusted partner for delivering consistent, high-performance intermediates that drive the success of your synthetic campaigns.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our capabilities align with your project goals. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how switching to our optimized synthesis route can improve your bottom line. We are prepared to provide specific COA data and route feasibility assessments to support your decision-making process, ensuring that you have all the necessary information to move forward with confidence. Let us help you accelerate your development timeline and achieve your production targets with our reliable supply of premium chiral ligands.
