Advanced Asymmetric Synthesis of Dapoxetine Hydrochloride for Commercial Scale-up
Advanced Asymmetric Synthesis of Dapoxetine Hydrochloride for Commercial Scale-up
The pharmaceutical landscape for treating premature ejaculation has been significantly shaped by the introduction of Dapoxetine hydrochloride, a short-acting selective serotonin reuptake inhibitor (SSRI). As demand for this active pharmaceutical ingredient (API) continues to grow globally, the efficiency and sustainability of its manufacturing process have become critical focal points for R&D directors and supply chain managers alike. Patent CN103304434A discloses a novel synthesizing method that represents a paradigm shift from traditional racemic resolution to direct asymmetric catalysis. This technology utilizes a self-prepared chiral catalyst, specifically (S)-alpha-alpha-diisopropyl dimethyl tert-butyl silicon oxygroup prolinol, to transform trans-cinnamaldehyde and N-carbobenzoxy hydroxylamine into the crucial chiral intermediate (S)-3-amino-3-phenyl propyl alcohol. By bypassing the inherent inefficiencies of resolution chemistry, this route offers a robust pathway for producing high-purity Dapoxetine intermediates suitable for rigorous commercial applications.
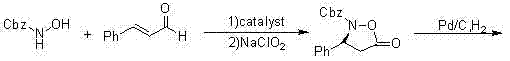
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Dapoxetine hydrochloride has relied heavily on two primary strategies, both of which suffer from significant economic and environmental drawbacks. The first category involves the synthesis of racemic Dapoxetine followed by chiral resolution, often utilizing L-tartaric acid as a resolving agent. As detailed in prior art such as Route One and Route Two, these methods necessitate repeated recrystallization steps to achieve acceptable enantiomeric excess (e.e.) values. This approach is fundamentally flawed from a mass balance perspective, as resolution theoretically discards at least 50% of the synthesized material (the unwanted enantiomer), leading to substantial raw material waste and increased disposal costs. Furthermore, the reliance on late-stage resolution means that expensive reagents and processing time are invested in material that is ultimately discarded, creating a bottleneck for cost reduction in pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN103304434A employs an asymmetric catalytic strategy that establishes chirality at the very beginning of the synthesis. By reacting trans-cinnamaldehyde with N-benzyloxycarbonyl hydroxylamine in the presence of the specialized organocatalyst, the process directly generates the chiral framework with exceptional stereocontrol. This eliminates the need for resolution entirely, effectively doubling the theoretical yield compared to racemic routes. The subsequent steps involve a streamlined sequence of oxidation, hydrogenation, and reduction to convert the initial adduct into the key intermediate (S)-3-amino-3-phenylpropyl alcohol. This intermediate is then methylated and coupled with 1-naphthol via a tosylate displacement, avoiding the more hazardous and expensive Mitsunobu reaction conditions often cited in older literature. The result is a process that is not only chemically elegant but also operationally simpler, with a reported total yield of 51.43%, which is significantly higher than many conventional alternatives.
Mechanistic Insights into Organocatalytic Asymmetric Addition
The core innovation of this synthesis lies in the design and application of the chiral organocatalyst. The catalyst, derived from L-proline, features a bulky silyl protecting group and isopropyl substituents that create a highly defined chiral environment. During the initial reaction with trans-cinnamaldehyde, the catalyst likely forms a chiral enamine or iminium ion intermediate, directing the nucleophilic attack of the hydroxylamine derivative to one specific face of the aldehyde. This precise spatial control is what enables the achievement of e.e. values exceeding 99% for the chiral intermediate. Unlike transition metal-catalyzed asymmetric hydrogenation which requires expensive noble metals like rhodium or ruthenium and stringent oxygen-free conditions, this organocatalytic system operates under relatively mild conditions, typically ranging from 5°C to room temperature. The robustness of the catalyst allows for efficient turnover without the risk of heavy metal contamination, a critical quality attribute for API intermediates intended for human consumption.
Furthermore, the impurity profile of this route is inherently cleaner due to the high stereoselectivity of the initial step. In resolution-based processes, the presence of the opposite enantiomer can lead to complex impurity profiles that are difficult to separate in downstream processing. By establishing high optical purity early, the subsequent chemical transformations—such as the oxidation using sodium chlorite and the reduction using sodium borohydride—proceed with minimal formation of diastereomeric impurities. The oxidation step converts the initial adduct into a carboxylic acid derivative (Compound I), which is then subjected to catalytic hydrogenation over Pd/C to yield the amino acid (Compound II). This sequence ensures that the chiral center remains intact while functional groups are manipulated to prepare for the final coupling. The use of standard reagents like boron trifluoride etherate for reduction and paraformaldehyde for methylation further underscores the practicality of this method for large-scale operations where reagent availability and cost are paramount.
How to Synthesize (S)-3-Amino-3-phenylpropyl Alcohol Efficiently
The synthesis of the key chiral intermediate is the linchpin of the entire Dapoxetine production process. The patent details a specific protocol starting with the preparation of the catalyst itself from Boc-L-proline methyl ester, followed by the asymmetric addition reaction. The process requires careful temperature control, initially cooling to 5°C during the addition of reagents to manage exotherms and maintain selectivity, before warming to room temperature for the oxidation phase. The subsequent hydrogenation and reduction steps are designed to be telescoped where possible, minimizing isolation losses. For a comprehensive understanding of the specific stoichiometric ratios, solvent choices, and workup procedures required to replicate this high-yielding pathway, please refer to the standardized synthesis guide below.
- Perform asymmetric catalytic reaction between trans-cinnamaldehyde and N-Cbz-hydroxylamine using a chiral prolinol catalyst to form the chiral intermediate.
- Oxidize the intermediate using sodium chlorite to obtain the carboxylic acid derivative, followed by catalytic hydrogenation to yield the chiral amino acid.
- Reduce the amino acid to the corresponding amino alcohol, methylate the amine, and couple with 1-naphthol via tosylation to finalize the Dapoxetine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this asymmetric catalytic route offers tangible strategic benefits beyond mere chemical elegance. The most significant advantage is the drastic simplification of the supply chain for raw materials. Traditional routes often rely on specialized chiral pool starting materials or expensive resolving agents that can be subject to market volatility. In contrast, this novel method utilizes trans-cinnamaldehyde and N-Cbz-hydroxylamine, which are commodity chemicals available from multiple global suppliers, thereby enhancing supply chain reliability and reducing the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of the resolution step provides a direct and substantial impact on the cost of goods sold (COGS). By avoiding the loss of 50% of the material inherent in resolution processes, the effective cost per kilogram of the chiral intermediate is nearly halved, even before accounting for the savings in solvents and energy associated with repeated recrystallizations. Additionally, the replacement of the Mitsunobu reaction—which typically requires expensive azodicarboxylates and phosphines—with a tosylation-substitution sequence further drives down reagent costs. The use of a non-metallic organocatalyst also removes the need for costly and regulated heavy metal scavenging steps, simplifying the purification workflow and reducing waste disposal expenses.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply continuity. The process operates at near-ambient temperatures and pressures (with the exception of the hydrogenation step which is standard industry practice), reducing the need for specialized cryogenic or high-pressure equipment that can be maintenance-intensive. The high yield and selectivity mean that batch failure rates are minimized, ensuring consistent output volumes. Furthermore, the scalability of organocatalysis is well-documented, allowing for seamless transfer from pilot plant to multi-ton commercial production without the nonlinear scale-up issues often encountered with sensitive transition metal catalysts.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The avoidance of heavy metals aligns with increasingly stringent regulatory guidelines regarding residual metals in pharmaceuticals. The higher overall yield translates to less waste generation per unit of product, supporting green chemistry initiatives. The solvents used, such as ethyl acetate, methanol, and dichloromethane, are standard industrial solvents with established recovery and recycling protocols, facilitating compliance with environmental regulations and reducing the carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the feasibility and advantages of adopting this route for commercial production.
Q: What is the primary advantage of this new synthesis method over traditional resolution routes?
A: The primary advantage is the elimination of the racemic resolution step, which typically wastes 50% of the material. This asymmetric catalytic route achieves high stereoselectivity (>99% ee) directly from achiral starting materials, significantly improving overall yield and reducing waste.
Q: Is the chiral catalyst used in this process commercially viable for large-scale production?
A: Yes, the catalyst is a proline derivative synthesized from readily available precursors like Boc-L-proline methyl ester. It avoids the use of expensive noble metals or complex chiral ligands found in other methods, making it highly suitable for industrial scale-up.
Q: What is the overall yield and purity profile of the final Dapoxetine Hydrochloride?
A: The patent reports a total yield of approximately 51.43% calculated from trans-cinnamaldehyde. The key chiral intermediate achieves an e.e. value of over 99%, ensuring high optical purity for the final API without the need for extensive recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dapoxetine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is smooth and efficient. We understand the critical nature of stringent purity specifications and operate rigorous QC labs equipped to verify enantiomeric excess and impurity profiles at every stage of the synthesis, guaranteeing that our Dapoxetine intermediates meet the highest global standards.
We invite potential partners to engage with our technical procurement team to discuss how this advanced asymmetric synthesis can be integrated into your supply chain. By leveraging our manufacturing capabilities, you can secure a reliable source of high-quality intermediates while achieving significant operational efficiencies. Please contact us to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your project requirements.
