Revolutionizing Naphthalene Derivative Synthesis: Scalable Metal-Free Catalysis for Pharmaceutical Intermediates
The patent CN115611695A introduces a groundbreaking metal-free synthesis methodology for substituted naphthalene derivatives, addressing critical limitations in traditional pharmaceutical intermediate production. This innovative process utilizes methyl trifluoromethanesulfonate (MeOTf) and potassium bromide (KBr) as synergistic organic catalysts to construct complex naphthalene frameworks under remarkably mild conditions. Unlike conventional metal-catalyzed approaches that introduce trace metal contamination risks, this method operates entirely without transition metals while achieving high yields through a streamlined one-pot procedure. The technology specifically targets the pharmaceutical industry's urgent need for pure, structurally diverse naphthalene-based intermediates used in bioactive compounds such as carbamate insecticides and natural product derivatives. By eliminating metal residues that compromise drug efficacy and require expensive post-processing, this patent delivers a fundamentally cleaner synthetic pathway with significant implications for both product quality and manufacturing economics in regulated environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for substituted naphthalene derivatives predominantly rely on transition metal catalysts, creating persistent challenges for pharmaceutical manufacturers. These methods necessitate stringent inert atmosphere conditions that increase operational complexity and equipment costs while introducing unavoidable trace metal residues into the final product. Such contamination poses significant risks to drug efficacy and safety profiles, requiring extensive and costly purification protocols to meet regulatory standards for pharmaceutical intermediates. The multi-step nature of conventional approaches often involves harsh reaction conditions exceeding 150°C, prolonged reaction times, and complex workup procedures including quenching and multiple extraction stages. These limitations collectively result in higher production costs, extended lead times, and compromised supply chain reliability due to the specialized infrastructure required. Furthermore, the dependence on scarce or expensive metal catalysts creates vulnerability to raw material price volatility and supply constraints that directly impact commercial viability.
The Novel Approach
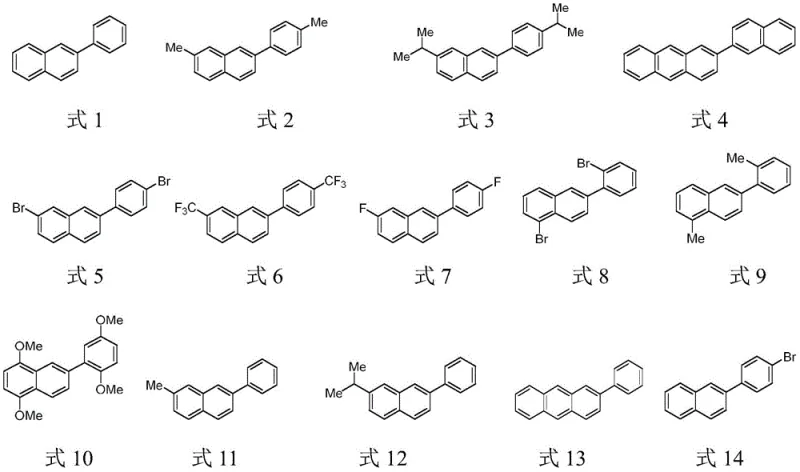
The patented methodology overcomes these limitations through an elegant organic catalyst system operating under ambient air conditions without any metal participation. By utilizing MeOTf and KBr in precise stoichiometric ratios (0.05:0.1:1.0:1.0), the process achieves efficient cyclization at moderate temperatures (120–130°C) within short reaction times (10–30 minutes). The reaction proceeds through a concerted catalytic mechanism that activates both epoxide substrates simultaneously, enabling direct formation of the naphthalene core structure with exceptional regioselectivity. Crucially, the methodology eliminates all inert gas requirements while maintaining high yields across diverse substrate combinations, as demonstrated by the successful synthesis of fourteen distinct derivatives including brominated, fluorinated, and alkyl-substituted variants. This approach represents a paradigm shift by transforming a traditionally complex multi-step process into a simple one-pot operation with minimal purification needs.
Mechanistic Insights into MeOTf/KBr Concerted Catalysis
The catalytic cycle operates through a sophisticated dual activation mechanism where MeOTf protonates the epoxide oxygen while KBr provides bromide nucleophiles to facilitate ring opening. This concerted action generates highly reactive carbocation intermediates that undergo intramolecular Friedel-Crafts alkylation with the adjacent aromatic system, forming the naphthalene backbone with precise regiocontrol. The absence of metal catalysts prevents unwanted side reactions such as homocoupling or over-reduction that commonly occur in transition metal-mediated processes, thereby enhancing product purity and reducing impurity formation pathways. The mild reaction conditions (120–130°C in ethanol) prevent thermal decomposition of sensitive functional groups while maintaining sufficient energy for the cyclization step, demonstrating an optimal balance between reactivity and selectivity that is unattainable with conventional methods.
Impurity control is inherently achieved through the reaction's self-limiting nature and precise stoichiometric control of catalysts. The one-pot design minimizes intermediate handling that typically introduces contaminants, while the direct solvent removal after reaction completion eliminates aqueous workup steps where impurities often form. The patent demonstrates consistent high purity across all synthesized derivatives through rigorous analytical validation including NMR and GC-MS characterization, with no detectable metal residues due to the complete absence of metal catalysts in the entire process. This inherent purity advantage significantly reduces downstream purification burden compared to metal-catalyzed routes where extensive metal removal protocols are required to meet pharmaceutical quality standards.
How to Synthesize Substituted Naphthalene Derivatives Efficiently
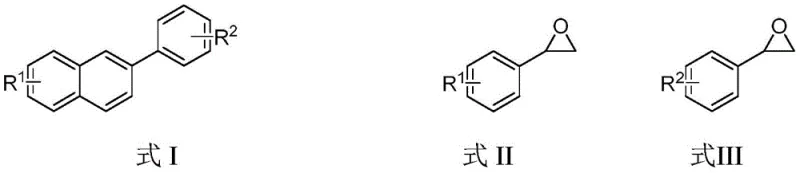
This patent provides a robust framework for synthesizing structurally diverse naphthalene-based intermediates through a precisely engineered catalytic system that eliminates traditional process limitations. The methodology represents a significant advancement over conventional approaches by enabling high-yield production under ambient conditions without requiring specialized equipment or hazardous reagents. Detailed standardized synthesis steps for commercial implementation are outlined below to ensure consistent quality and scalability across manufacturing environments.
- Combine potassium bromide (KBr), formula II compound, and formula III compound in activated ethanol solvent under ambient air conditions, ensuring complete dissolution before catalyst addition.
- Introduce methyl trifluoromethanesulfonate (MeOTf) to the reaction mixture, maintaining precise molar ratios of 0.05: 0.1:1.0:1.0 for MeOTf:KBr:formula II:formula III.
- Heat the sealed reaction vessel to 120–130°C for 10–30 minutes, followed by direct solvent removal via rotary evaporation without quenching or extraction steps.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by transforming traditionally complex manufacturing processes into streamlined operations with enhanced reliability. The elimination of metal catalysts resolves persistent supply chain vulnerabilities associated with precious metal dependencies while creating substantial cost optimization opportunities through process simplification. These advantages collectively strengthen procurement strategies by improving supplier flexibility, reducing operational risks, and enhancing overall value delivery for pharmaceutical manufacturers seeking reliable intermediate sources.
- Cost Reduction in Manufacturing: The complete elimination of transition metal catalysts removes significant expenses associated with metal procurement, specialized handling protocols, and extensive post-reaction purification required to eliminate trace metal residues from final products. This organic catalyst system utilizes commercially available reagents that avoid price volatility inherent in precious metals, while the simplified one-pot procedure reduces solvent consumption and eliminates multiple intermediate isolation steps. The ambient air operation further decreases infrastructure costs by removing requirements for inert gas systems, collectively delivering substantial cost savings without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: The use of readily available commercial substrates and standard laboratory equipment creates exceptional supply chain resilience by eliminating dependencies on specialized materials or custom manufacturing setups. The robustness of the reaction under ambient conditions ensures consistent performance across different production environments without requiring precise atmospheric control, significantly reducing batch failure risks. This methodology enables flexible production scaling from laboratory to commercial volumes using existing infrastructure, providing procurement teams with greater supplier options and reduced lead time variability compared to traditional metal-catalyzed processes that require dedicated facilities.
- Scalability and Environmental Compliance: The straightforward process design facilitates seamless scale-up from laboratory to commercial production volumes while maintaining high yields and purity standards essential for pharmaceutical applications. The elimination of hazardous metal waste streams simplifies environmental compliance by removing complex waste treatment requirements associated with heavy metal disposal. The reduced number of processing steps lowers overall energy consumption and solvent waste generation, aligning with green chemistry principles while supporting corporate sustainability initiatives without sacrificing manufacturing efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations for implementing this patented technology in pharmaceutical intermediate manufacturing operations. These responses are derived directly from the patent's experimental data and technical disclosures to provide accurate implementation guidance for procurement and R&D teams evaluating this methodology.
Q: How does this metal-free process eliminate trace metal contamination risks in pharmaceutical intermediates?
A: The exclusive use of organic catalysts (MeOTf/KBr) completely avoids transition metals, eliminating residual metal contamination that would otherwise require costly purification steps and compromise drug efficacy in final pharmaceutical products.
Q: What supply chain advantages does the ambient air reaction condition provide?
A: The elimination of inert gas requirements simplifies reactor setup and reduces dependency on specialized equipment, enabling faster production ramp-up and enhanced reliability in standard manufacturing facilities without additional infrastructure investments.
Q: How does the one-pot methodology improve cost efficiency compared to traditional routes?
A: The integrated reaction and simplified purification (direct rotary evaporation followed by single-column chromatography) significantly reduces processing time and eliminates multiple intermediate isolation steps, substantially lowering operational costs while maintaining high purity standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Naphthalene Derivative Supplier
Our company leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver high-purity substituted naphthalene derivatives meeting stringent purity specifications through rigorously validated processes. As a specialized CDMO partner, we combine deep expertise in organic catalysis with state-of-the-art manufacturing capabilities and rigorous QC labs to ensure consistent product quality across all scales. Our technical team has successfully implemented this patented MeOTf/KBr methodology in commercial settings, optimizing parameters for maximum yield while maintaining the critical metal-free advantage essential for pharmaceutical applications.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can enhance your specific supply chain requirements. Contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs.
