Revolutionizing Vinyl Sulfone Production: A Metal-Free Aqueous Protocol for Commercial Scale-Up
Revolutionizing Vinyl Sulfone Production: A Metal-Free Aqueous Protocol for Commercial Scale-Up
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards greener, more sustainable manufacturing processes, driven by both regulatory pressure and economic necessity. Patent CN108623503B introduces a groundbreaking methodology for the synthesis of vinyl sulfone derivatives, a critical structural motif found in numerous bioactive molecules and drug candidates. This innovation addresses the longstanding challenges associated with traditional synthetic routes by utilizing a metal-free catalytic system in an aqueous medium. By leveraging tetrabutylammonium iodide (TBAI) as a promoter and tert-butyl hydroperoxide (TBHP) as an oxidant, this protocol achieves high efficiency at room temperature. For R&D directors and procurement managers seeking a reliable vinyl sulfone supplier, this technology represents a significant leap forward in process intensification and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of vinyl sulfone scaffolds has relied heavily on methodologies that pose significant operational and environmental burdens. Traditional approaches often involve the oxidation of vinyl sulfides, Horner-Wadsworth-Emmons reactions using phosphonates, or Knoevenagel condensations, all of which frequently require harsh reaction conditions and generate substantial chemical waste. More recent advancements have utilized transition metal catalysts, such as copper complexes, to facilitate the direct coupling of alkenes and sulfonyl hydrazides. However, these metal-catalyzed systems necessitate the use of volatile organic solvents and rigorous exclusion of moisture, complicating the manufacturing workflow. Furthermore, the presence of residual heavy metals in the final active pharmaceutical ingredient (API) intermediate requires extensive and costly purification steps to meet stringent regulatory limits, thereby inflating the overall production cost and extending lead times.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN108623503B offers a streamlined, eco-friendly alternative that operates under mild conditions. The core innovation lies in the use of water as the sole solvent, eliminating the need for hazardous organic media and drastically reducing the environmental footprint of the synthesis. The reaction proceeds efficiently at room temperature (25°C) under a standard air atmosphere, removing the energy costs associated with heating and the capital expenditure for inert gas systems. This metal-free strategy not only simplifies the reaction setup but also ensures that the resulting vinyl sulfone derivatives are free from transition metal contamination. For stakeholders focused on cost reduction in pharmaceutical intermediate manufacturing, this approach offers a compelling value proposition by merging high chemical efficiency with operational simplicity.

Mechanistic Insights into TBAI-Promoted Radical Oxidative Coupling
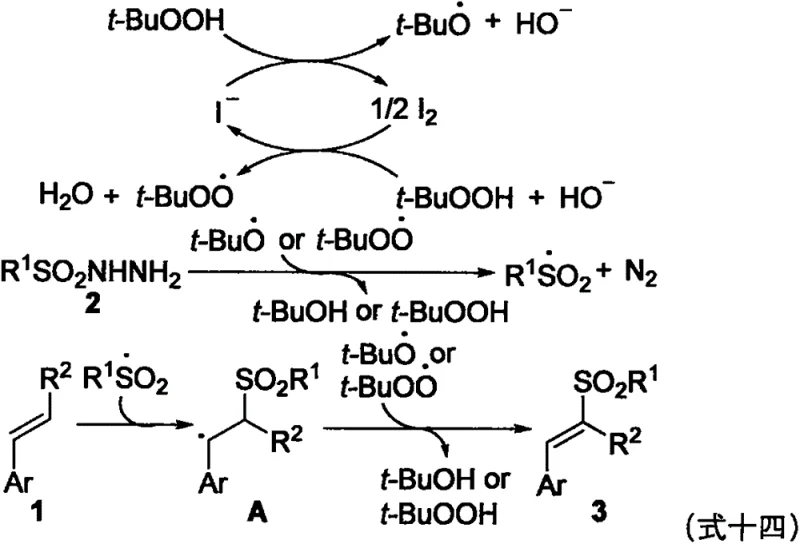
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams evaluating its robustness and scalability. The reaction is initiated by the interaction between the iodide anion from TBAI and the oxidant TBHP, generating reactive iodine species and tert-butoxyl radicals. These radical species abstract a hydrogen atom from the sulfonyl hydrazide, leading to the formation of a sulfonyl radical accompanied by the release of nitrogen gas. This sulfonyl radical then adds regioselectively to the terminal carbon of the styrene double bond, creating a stabilized benzylic radical intermediate. Subsequent oxidation and deprotonation steps yield the final trans-vinyl sulfone product with high stereoselectivity. The use of water as a solvent likely plays a dual role, acting not only as a green medium but also facilitating the stabilization of polar transition states through hydrogen bonding interactions.
The radical nature of this mechanism was rigorously confirmed through control experiments using radical scavengers. When inhibitors such as BHT or hydroquinone were introduced into the reaction system, the formation of the target product was almost completely suppressed, indicating that a free-radical pathway is indeed operative. This mechanistic clarity provides confidence in the predictability of the reaction across different substrate classes. The system tolerates a wide array of functional groups, including halogens, nitro groups, and ethers, without interfering with the radical propagation cycle. For technical teams, this implies that the process can be adapted for the synthesis of complex, multifunctionalized intermediates without requiring extensive re-optimization of reaction parameters for each new derivative.
How to Synthesize Vinyl Sulfone Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution, making it highly attractive for both laboratory-scale discovery and pilot-plant production. The procedure involves simply combining the alkene substrate, sulfonyl hydrazide, catalytic amount of TBAI, and stoichiometric TBHP in water within a standard reaction vessel. The mixture is stirred at ambient temperature until conversion is complete, typically monitored by thin-layer chromatography or gas chromatography. Following the reaction, standard workup procedures involving extraction with ethyl acetate and drying over anhydrous sodium sulfate afford the crude product, which can be purified via column chromatography. The detailed standardized synthesis steps are outlined in the guide below.
- Charge a Schlenk flask with alkene substrate, sulfonyl hydrazide, TBAI promoter, and TBHP oxidant in water.
- Stir the mixture at 25°C under air atmosphere for approximately 36 hours until TLC indicates completion.
- Extract with ethyl acetate, dry over sodium sulfate, and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this aqueous, metal-free protocol offers transformative benefits for supply chain stability and cost management. The elimination of transition metal catalysts removes a major bottleneck in the purification process, as there is no longer a need for specialized scavenging resins or repeated recrystallizations to remove trace metals. This simplification directly translates to reduced processing time and lower consumption of auxiliary materials. Furthermore, the use of water as a solvent significantly lowers raw material costs compared to high-purity organic solvents and mitigates the risks associated with solvent flammability and toxicity. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity vinyl sulfone derivatives.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the removal of expensive metal catalysts and the substitution of organic solvents with water. By avoiding the procurement of noble metals and the disposal costs associated with hazardous organic waste, manufacturers can achieve substantial cost savings. Additionally, the ambient temperature operation reduces energy consumption for heating and cooling, further optimizing the utility costs associated with large-scale production runs. The simplified downstream processing also reduces labor hours and equipment occupancy time, enhancing overall plant throughput.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like TBAI and TBHP, which are readily available in bulk quantities, ensures a stable supply of reagents compared to specialized ligand-metal complexes that may face sourcing volatility. The robustness of the reaction under air atmosphere means that production is less susceptible to interruptions caused by inert gas supply failures or stringent moisture control requirements. This reliability is critical for maintaining consistent delivery schedules for API intermediates, allowing procurement managers to forecast inventory needs with greater confidence and reduce safety stock levels.
- Scalability and Environmental Compliance: The green chemistry credentials of this method align perfectly with increasingly strict environmental regulations regarding solvent emissions and heavy metal discharge. Scaling up an aqueous process is inherently safer due to the non-flammable nature of the reaction medium, reducing the risk of thermal runaway incidents. The absence of toxic metal residues simplifies the regulatory filing process for new drug applications, as the impurity profile is cleaner and easier to characterize. This facilitates faster time-to-market for new pharmaceutical products relying on these vinyl sulfone scaffolds.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the primary advantages of this aqueous synthesis method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts like copper and avoids toxic organic solvents, significantly reducing environmental impact and downstream purification costs associated with heavy metal removal.
Q: Does this protocol tolerate diverse functional groups on the styrene substrate?
A: Yes, the reaction demonstrates broad substrate scope, successfully accommodating electron-donating groups such as methoxy and methyl, as well as electron-withdrawing groups like chloro, bromo, and nitro substituents.
Q: Is the reaction sensitive to oxygen or moisture?
A: The process is remarkably robust, operating efficiently under standard air atmosphere conditions without the need for inert gas protection, which simplifies operational requirements for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinyl Sulfone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic technologies to maintain a competitive edge in the global market. Our team of expert chemists has thoroughly evaluated the metal-free aqueous protocol described in CN108623503B and possesses the technical capability to implement this route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of vinyl sulfone derivative delivered meets the highest quality standards required by the pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced manufacturing capability for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to green chemistry and operational excellence can drive value for your organization.
