Advanced Pd-Catalyzed Synthesis of Sulfur-containing Isoindolinones for Commercial Pharmaceutical Manufacturing
Advanced Pd-Catalyzed Synthesis of Sulfur-containing Isoindolinones for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN109369504B introduces a significant advancement in this domain by detailing a preparation method for sulfur-containing 3-methyleneisoindolin-1-one derivatives. These structures are pivotal in medicinal chemistry, often functioning as core motifs in PDE4 inhibitors and antineoplastic agents. The disclosed technology leverages a palladium(II)-catalyzed tandem alkenylation and cyclization reaction, utilizing aryl formamides and methyl acrylate as primary substrates. This approach represents a strategic shift from traditional multi-step syntheses, offering a streamlined pathway that enhances both atomic economy and operational simplicity. For R&D directors and process chemists, understanding the nuances of this catalytic system is essential for integrating high-purity intermediates into modern drug discovery pipelines efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-methyleneisoindolin-1-one derivatives has relied on methods that often involve harsh reaction conditions, expensive reagents, or cumbersome multi-step sequences. Conventional routes frequently require the pre-functionalization of substrates or the use of stoichiometric amounts of toxic heavy metals, which complicates downstream purification and waste management. Furthermore, the introduction of sulfur-containing moieties adjacent to catalytic centers has traditionally been avoided due to the well-documented phenomenon of catalyst poisoning, where sulfur atoms bind irreversibly to transition metals, halting the catalytic cycle. This limitation has restricted the chemical space available to medicinal chemists, forcing them to explore less efficient alternatives or accept lower yields and higher impurity profiles in the final active pharmaceutical ingredients.
The Novel Approach
The methodology described in the patent overcomes these historical barriers by employing an N,S-bidentate ligand strategy that surprisingly prevents catalyst deactivation while promoting efficient C-H bond activation. By using Pd(OAc)2 as the catalyst and Cu(OAc)2 as the oxidant in a mixed solvent system of 1,2-dichloroethane and glacial acetic acid, the reaction proceeds smoothly at 110°C. This novel approach allows for the direct construction of the isoindolinone core in a single pot, eliminating the need to isolate unstable intermediates. The reaction scheme below illustrates the transformation of N-(2-(alkylthio)phenyl)benzamides into the target sulfur-containing derivatives with remarkable efficiency.
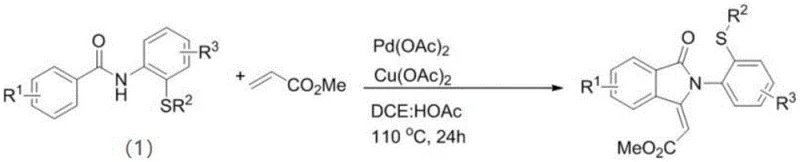
This streamlined process not only reduces the overall number of unit operations but also significantly lowers the environmental footprint by minimizing solvent usage and waste generation. For procurement managers, this translates to a more reliable supply chain for complex intermediates, as the simplified process reduces the risk of batch failures associated with intricate multi-step syntheses. The ability to tolerate a wide range of substituents, including electron-withdrawing groups like halogens and trifluoromethyl groups, further enhances the versatility of this method for generating diverse libraries of pharmacologically active compounds.
Mechanistic Insights into Pd-Catalyzed Tandem Alkenylation/Cyclization
A deep understanding of the catalytic cycle is crucial for R&D teams aiming to optimize this process for commercial scale-up. The proposed mechanism begins with the coordination of the palladium catalyst to the nitrogen and sulfur atoms of the substrate, forming a stable five-membered palladacycle intermediate. This step is critical as it activates the ortho-C-H bond for subsequent functionalization. Unlike typical scenarios where sulfur poisons the catalyst, the specific geometry and electronic properties of this N,S-bidentate system allow for reversible coordination, maintaining the catalytic activity throughout the reaction cycle. The insertion of methyl acrylate into the palladium-carbon bond follows, leading to a seven-membered palladium species.
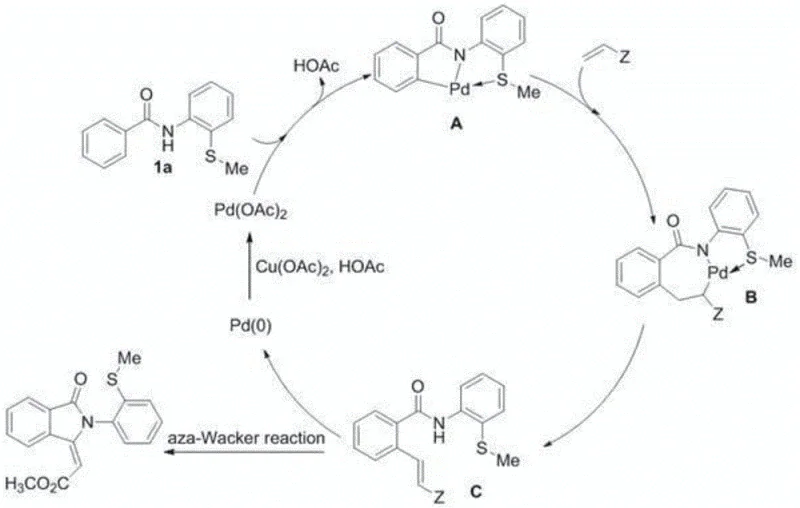
Subsequent beta-hydride elimination generates the ortho-olefinated intermediate and a Pd(0) species. The cycle is completed through an intramolecular aza-Wacker type cyclization, which constructs the isoindolinone ring system, followed by the re-oxidation of Pd(0) to Pd(II) by the copper oxidant. This mechanistic pathway explains the high selectivity and moderate-to-good yields observed across various substrates. For quality control purposes, understanding that the reaction relies on a redox-neutral cycle facilitated by the copper oxidant helps in identifying potential impurities related to incomplete oxidation or side reactions of the olefin. The robustness of this mechanism ensures that the impurity profile remains manageable, a key factor for meeting stringent regulatory standards in pharmaceutical manufacturing.
How to Synthesize Sulfur-containing 3-methyleneisoindolin-1-one Efficiently
The practical implementation of this synthesis requires careful attention to reaction parameters to maximize yield and purity. The patent outlines a generalized procedure that is amenable to standard laboratory and pilot plant equipment. Operators should ensure that the molar ratios of catalyst and oxidant are strictly maintained, as deviations can impact the turnover number of the palladium species. The use of anhydrous conditions and high-purity solvents is recommended to prevent hydrolysis of the ester functionality or deactivation of the catalyst. Detailed standardized operating procedures for this specific transformation are provided below to guide technical teams in replicating the results consistently.
- Mix N-(2-(alkylthio)phenyl)benzamide substrate with methyl acrylate, Pd(OAc)2 catalyst (10 mol%), and Cu(OAc)2 oxidant (1.2 equivalents) in a solvent mixture of 1,2-dichloroethane and glacial acetic acid (3: 1 ratio).
- Heat the reaction mixture under an oil bath at 110°C for 24 hours to facilitate the tandem alkenylation and cyclization reaction.
- Neutralize the mixture with saturated sodium bicarbonate, extract with ethyl acetate, dry over anhydrous magnesium sulfate, and purify the residue via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers substantial benefits for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. The elimination of intermediate isolation steps drastically reduces the total processing time and labor costs associated with the production of these valuable heterocycles. By consolidating multiple synthetic transformations into a single tandem reaction, manufacturers can achieve significant throughput improvements without requiring additional reactor capacity. This efficiency gain is particularly valuable for supply chain heads who must manage tight delivery schedules and minimize inventory holding costs for volatile intermediates.
- Cost Reduction in Manufacturing: The process utilizes readily available starting materials such as substituted benzamides and methyl acrylate, which are commodity chemicals with stable pricing. The avoidance of exotic ligands or specialized reagents further drives down the raw material costs. Additionally, the simplified workup procedure, which involves basic neutralization and extraction, reduces the consumption of auxiliary chemicals and solvents. This lean manufacturing approach ensures that the final cost of goods sold is optimized, providing a competitive edge in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance to air and moisture relative to other sensitive organometallic processes, enhances the reliability of supply. The broad substrate scope means that a single production line can be adapted to produce a variety of derivatives by simply changing the starting benzamide, offering flexibility to meet diverse customer demands. This adaptability reduces the lead time for high-purity pharmaceutical intermediates, allowing partners to respond quickly to changes in drug development pipelines without the need for extensive process re-validation.
- Scalability and Environmental Compliance: The reaction operates at a moderate temperature of 110°C, which is easily achievable with standard heating systems and does not require cryogenic conditions or high-pressure equipment. This facilitates straightforward scale-up from gram to kilogram and ton scales. Furthermore, the reduced generation of waste solution and the absence of hazardous gas emissions align with modern green chemistry principles. This environmental compliance minimizes the regulatory burden and disposal costs, ensuring long-term sustainability for the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation. They are intended to clarify the operational feasibility and strategic value of adopting this method for large-scale production. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios.
Q: Why is the use of N,S-bidentate ligands significant in this synthesis?
A: Traditionally, sulfur atoms are known to poison palladium catalysts, limiting their use in C-H activation. This patent demonstrates a breakthrough where the N,S-bidentate directing group successfully coordinates with palladium without deactivating the catalyst, enabling efficient tandem cyclization.
Q: What are the primary advantages of this method over conventional routes?
A: The method eliminates the need for isolating intermediate products, utilizing a one-pot tandem reaction. It operates under relatively mild conditions (110°C) with commercially available substrates, significantly simplifying the workflow and reducing energy consumption compared to multi-step traditional syntheses.
Q: What is the substrate scope for this reaction?
A: The process exhibits strong substrate universality, tolerating various electron-donating and electron-withdrawing groups on the benzamide ring, including methyl, methoxy, fluoro, chloro, bromo, and trifluoromethyl substituents, as well as different thio-groups like methyl, ethyl, and phenyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-containing Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the Pd-catalyzed tandem alkenylation/cyclization route described in CN109369504B and confirmed its viability for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this streamlined process. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the synthesis of complex organic molecules.
