Advanced Ruthenium-Catalyzed Synthesis of Benzindole Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance high purity with operational simplicity. A significant breakthrough in this domain is detailed in patent CN109053550B, which discloses a novel method for synthesizing benzindole derivatives through a ruthenium-catalyzed C-H bond activation and cyclization process. This technology represents a paradigm shift from traditional approaches by utilizing a directing group-directed diazocarbene insertion reaction that operates under remarkably mild conditions. Unlike conventional methods that often demand stringent inert atmospheres and complex catalytic systems, this innovation leverages a cymene ruthenium dichloride dimer catalyst in a green ethanol-water solvent system. For R&D directors and procurement specialists alike, this development signals a new era of efficiency in producing high-purity pharmaceutical intermediates, offering a pathway to reduce both technical risk and manufacturing costs while maintaining exceptional yield and substrate versatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzindole compounds, which serve as critical fused heterocyclic skeletons in luminescent materials and medicinal chemistry, has been fraught with technical challenges. Traditional pathways typically rely on the activation and cyclization of naphthylamine with olefins, a process that frequently necessitates harsh reaction conditions to drive the transformation to completion. These legacy methods often require rigorous inert gas protection to prevent catalyst deactivation or substrate oxidation, thereby increasing the capital expenditure for specialized reactor equipment and operational overhead. Furthermore, the catalysts employed in these older protocols are relatively single in variety and often suffer from limited turnover numbers, leading to inconsistent yields and difficult purification profiles. The reliance on non-green solvents and the generation of complex impurity profiles further complicate the downstream processing, making the commercial scale-up of complex pharmaceutical intermediates a costly and environmentally burdensome endeavor that supply chain managers strive to avoid.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach described in the patent introduces a highly efficient ruthenium-catalyzed strategy that fundamentally redefines the synthesis landscape for benzindole derivatives. By employing a directing group-directed diazocarbene insertion mechanism, this method achieves cyclization under mild thermal conditions, specifically at 65°C, without the absolute necessity for inert gas protection. This operational flexibility is a game-changer for manufacturing facilities, as it drastically simplifies the reactor setup and reduces the dependency on expensive nitrogen or argon supplies. The use of a mixed ethanol and water solvent system not only aligns with modern green chemistry principles but also facilitates easier product isolation and solvent recovery. As illustrated in the core reaction scheme below, the transformation of naphthylamine and diazo esters into the target benzindole structure is achieved with remarkable precision and yield.
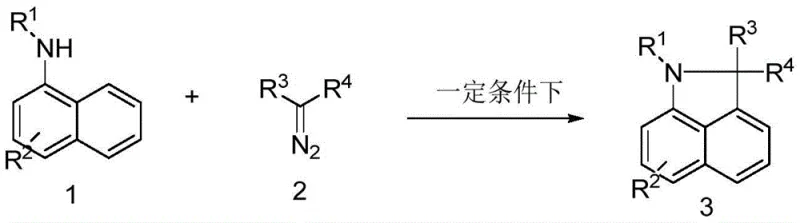
The structural diversity achievable through this method is exemplified by the successful synthesis of various derivatives, such as the ethyl 2-acetyl-1,2-dihydrobenzol[c,d]indole-2-carboxylate shown below, which serves as a foundational scaffold for further functionalization in drug discovery pipelines.
![Chemical structure of Ethyl 2-acetyl-1,2-dihydrobenzol[c,d]indole-2-carboxylate from Example 1](/insights/img/benzindole-synthesis-ruthenium-pharma-supplier-20260303020029-03.png)
Mechanistic Insights into Ru-Catalyzed C-H Activation and Cyclization
The mechanistic elegance of this synthesis lies in the unique role of the cymene ruthenium dichloride dimer catalyst in facilitating the C-H bond activation functionalization. In this catalytic cycle, the ruthenium center coordinates with the directing group on the naphthylamine substrate, effectively lowering the activation energy required for the cleavage of the proximal C-H bond. This activation allows for the subsequent insertion of the diazocarbene species generated from the diazo ester, a step that is traditionally difficult to control with high regioselectivity. The presence of cesium acetate as an additive plays a crucial role in stabilizing the catalytic species and promoting the deprotonation steps necessary for the cyclization to proceed smoothly. This synergistic interaction between the metal catalyst and the additive ensures that the reaction proceeds with high fidelity, minimizing the formation of side products that typically plague carbene insertion reactions. For technical teams, understanding this mechanism is vital for optimizing reaction parameters and ensuring that the process remains robust even when scaling from gram to kilogram quantities.
Furthermore, the impurity control mechanism inherent in this route is superior to many transition-metal catalyzed processes. Because the reaction does not involve redox steps that can generate unpredictable oxidative byproducts, the resulting crude mixture is significantly cleaner. The mild conditions prevent the decomposition of sensitive functional groups on the substrate, such as the amino or halo substituents seen in various examples within the patent data. This stability translates directly into a simplified purification workflow, where standard silica gel column chromatography is sufficient to isolate the target benzindole derivative in high purity. The ability to tolerate a wide range of substituents, including electron-withdrawing and electron-donating groups, without compromising the integrity of the catalytic cycle demonstrates the versatility of this ruthenium system. This level of control is essential for producing reliable pharmaceutical intermediates where strict impurity specifications are mandatory for regulatory compliance.
How to Synthesize Benzindole Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward sequence of operations that leverages the mild nature of the chemistry. The process begins with the dissolution of the naphthylamine starting material in a mixture of water and ethanol, followed by the addition of the diazo ester, the ruthenium catalyst, and the cesium acetate additive. The reaction mixture is then heated to a moderate temperature of 65°C and stirred for approximately 16 hours, during which time the C-H activation and cyclization occur spontaneously without the need for specialized atmospheric controls. Detailed standardized synthesis steps for specific derivatives are provided in the guide below, ensuring reproducibility and consistency across different batches.
- Dissolve naphthylamine and diazo ester in a mixed solvent of ethanol and water, then add the ruthenium catalyst and cesium acetate additive.
- Heat the reaction mixture to 65°C and stir for 16 hours without the need for inert gas protection to facilitate C-H bond activation and cyclization.
- Upon completion, extract the product with dichloromethane, concentrate the organic phase, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly address the pain points of procurement managers and supply chain heads. The elimination of inert gas protection and the use of common, low-cost solvents like ethanol and water significantly lower the operational expenditure associated with the manufacturing process. This reduction in complexity allows for faster turnaround times and reduces the risk of production delays caused by equipment failures related to high-pressure or high-vacuum systems. Moreover, the high yield and broad substrate scope mean that a single production line can be adapted to manufacture a wide variety of benzindole derivatives, enhancing the flexibility of the supply chain and reducing the need for dedicated infrastructure for each specific product. These factors combine to create a more resilient and cost-effective supply model for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the reaction conditions and the choice of reagents. By removing the requirement for expensive inert gases and specialized anhydrous solvents, the direct material costs are significantly lowered. Additionally, the use of a robust ruthenium catalyst that functions effectively in aqueous media reduces the burden on solvent recovery systems and waste treatment facilities. The high conversion rates observed in the patent examples imply that less raw material is wasted, further driving down the cost per kilogram of the final active pharmaceutical ingredient precursor. These cumulative savings make the process highly competitive for large-scale commercial production.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on exotic reagents or fragile reaction conditions that are prone to failure. This method mitigates those risks by utilizing stable raw materials that are not easily oxidized and can be stored under standard conditions. The tolerance for ambient atmosphere during the reaction means that minor breaches in sealing do not necessarily result in batch failure, providing a safety net for manufacturing operations. This robustness ensures that delivery schedules can be met consistently, reducing the lead time for high-purity intermediates and allowing downstream drug manufacturers to plan their production cycles with greater confidence and accuracy.
- Scalability and Environmental Compliance: As regulatory pressure mounts on the chemical industry to reduce its environmental footprint, the adoption of green solvents becomes a strategic imperative. The ethanol-water solvent system used in this synthesis is inherently safer and more environmentally friendly than traditional chlorinated or aromatic solvents. This alignment with green chemistry principles simplifies the permitting process for new manufacturing lines and reduces the costs associated with hazardous waste disposal. Furthermore, the mild thermal conditions reduce energy consumption, contributing to a lower carbon footprint for the overall manufacturing process. These attributes make the technology not only commercially viable but also sustainable for long-term industrial application.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed synthesis. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of this ruthenium-catalyzed method over traditional benzindole synthesis?
A: This method eliminates the need for harsh inert gas protection and utilizes a green ethanol/water solvent system, significantly simplifying operational complexity and reducing environmental impact compared to traditional olefin activation routes.
Q: Is this synthesis route scalable for industrial production of pharmaceutical intermediates?
A: Yes, the reaction operates under mild conditions (65°C) with stable raw materials that are not prone to oxidation, making it highly suitable for commercial scale-up and consistent batch-to-batch quality control.
Q: What is the substrate scope for the naphthylamine and diazo components in this reaction?
A: The process demonstrates strong substrate applicability, tolerating various substituents such as halogens, amino groups, alkyls, and aryl groups on the naphthylamine, as well as diverse acyl groups on the diazo compound.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzindole Derivative Supplier
The technological potential of this ruthenium-catalyzed C-H activation route is immense, offering a clear path to high-quality benzindole scaffolds essential for modern drug development. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to handle the complex analysis required for these fused heterocyclic systems, guaranteeing that every batch delivered adheres to the highest international standards for pharmaceutical intermediates. We understand the critical nature of supply chain reliability and are committed to being a partner that supports your long-term growth.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be integrated into your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless transition from laboratory discovery to commercial success.
