Advanced Synthesis of Star-Shaped Photoactive Compounds for Organic Electronics and Solar Cells
Advanced Synthesis of Star-Shaped Photoactive Compounds for Organic Electronics and Solar Cells
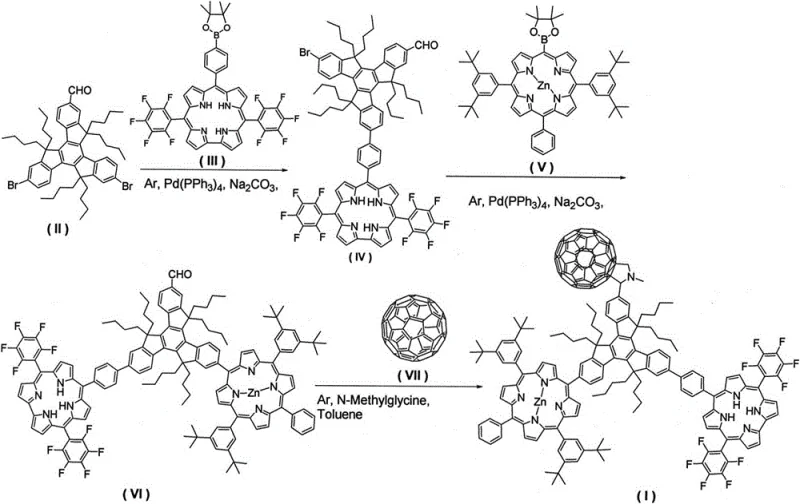
The rapid evolution of organic photovoltaic materials demands increasingly sophisticated molecular architectures capable of efficient energy conversion. Patent CN109096313B introduces a groundbreaking preparation method for a trimeric indenyl corrole-porphyrin-fullerene star-shaped compound, representing a significant leap forward in the design of donor-acceptor systems. This innovative structure integrates three distinct photoactive groups—corrole, porphyrin, and fullerene—onto a rigid truxene core, creating a robust platform for photoinduced energy and electron transfer. For R&D directors and procurement specialists seeking reliable electronic chemical suppliers, this technology offers a pathway to high-performance materials with streamlined manufacturing protocols. The patent details a robust synthetic strategy that overcomes historical challenges in synthesizing complex star-shaped molecules, ensuring both high purity and structural integrity essential for next-generation optoelectronic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of star-shaped compounds with donor-acceptor structures has been plagued by significant technical hurdles that impede commercial viability. Traditional routes often involve excessive synthesis steps, leading to cumulative yield losses and increased production costs. A critical bottleneck identified in prior art is the difficulty in separating the target product from by-products due to their similar polarity profiles, which frequently results in low overall yields and compromised purity. Furthermore, conventional methods may require harsh reaction conditions that limit the functional group tolerance and scalability of the process. These inefficiencies create substantial supply chain risks for manufacturers of organic solar cells and artificial photosynthesis systems, where material consistency is paramount. The inability to efficiently purify these complex architectures has long been a barrier to their widespread adoption in high-end electronic applications.
The Novel Approach
The methodology disclosed in CN109096313B revolutionizes this landscape by employing a modular, step-wise construction strategy centered on Suzuki coupling and 1,3-dipolar cycloaddition. This approach allows for the precise grafting of different photoactive groups onto the 2, 7, and 12 positions of the truxene core under mild reaction conditions. By utilizing tetrakis(triphenylphosphine)palladium as a catalyst, the process achieves high selectivity and avoids the formation of intractable by-product mixtures. The reaction conditions are notably gentle, typically ranging from 65-85°C for the coupling steps and 110-120°C for the final cycloaddition, which preserves the integrity of sensitive functional groups. This streamlined protocol not only simplifies the operational workflow but also enhances the feasibility of cost reduction in organic photovoltaic material manufacturing by minimizing waste and energy consumption.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Cycloaddition
The core of this synthesis relies on the versatility of palladium-catalyzed cross-coupling reactions to build the molecular scaffold. In the initial stages, the 7,12-dibromo-truxene aldehyde derivative reacts with meso-phenyl borate carbazole and porphyrin derivatives via Suzuki coupling. This mechanism involves the oxidative addition of the aryl bromide to the Pd(0) center, followed by transmetallation with the organoboron species and reductive elimination to form the new carbon-carbon bond. The use of a biphasic solvent system comprising tetrahydrofuran, methanol, and water facilitates the solubility of both organic substrates and the inorganic base, sodium carbonate, ensuring efficient catalytic turnover. This precise control over bond formation is critical for maintaining the symmetry and electronic properties of the final star-shaped architecture, which directly influences its performance in light-harvesting applications.
Following the assembly of the truxene-porphyrin framework, the final step employs a 1,3-dipolar cycloaddition to attach the fullerene acceptor. In this reaction, sarcosine reacts with the aldehyde group on the truxene core to generate an azomethine ylide intermediate in situ. This dipole then undergoes a concerted cycloaddition with the electron-deficient double bonds of the fullerene C60 cage. This specific functionalization strategy is advantageous because it occurs under relatively mild thermal conditions in toluene, avoiding the degradation of the delicate porphyrin and corrole macrocycles. The resulting covalent linkage ensures stable electronic communication between the donor (corrole/porphyrin) and acceptor (fullerene) units, enabling the long-life charge separation states necessary for efficient solar energy conversion.
How to Synthesize Trimeric Indenyl Corrole-Porphyrin-Fullerene Efficiently
The synthesis of this high-purity star-shaped compound is achieved through a logical sequence of three distinct chemical transformations, each optimized for yield and purity. The process begins with the construction of the carbazole-functionalized truxene intermediate, followed by the attachment of the porphyrin unit, and concludes with the fullerene grafting. Each step utilizes standard laboratory techniques such as refluxing under inert atmosphere and silica gel column chromatography for purification, making the protocol accessible for scale-up. The detailed standardized synthesis steps below outline the specific reagents, stoichiometry, and conditions required to replicate this advanced material successfully.
- Perform Suzuki coupling between 7,12-dibromo-truxene aldehyde derivative and meso-phenyl borate carbazole derivative using Pd(PPh3)4 catalyst at 65-85°C to obtain the trimeric indenyl carbazole intermediate.
- React the trimeric indenyl carbazole intermediate with meso-phenyl borate porphyrin derivative under similar Suzuki coupling conditions to form the carbazole-truxene-porphyrin ternary system compound.
- Conduct a 1,3-dipolar cycloaddition reaction between the ternary system compound, fullerene, and sarcosine in toluene at 110-120°C to finalize the star-shaped compound structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical performance. The simplification of the reaction sequence directly translates to reduced operational complexity, lowering the barrier for entry into the production of high-value optoelectronic materials. By eliminating the need for extreme reaction conditions and complex purification workflows associated with older methods, manufacturers can achieve significant cost savings in organic photovoltaic material manufacturing. The robustness of the Suzuki coupling and cycloaddition steps ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with downstream device fabricators. Furthermore, the use of commercially available starting materials mitigates supply chain risks, ensuring a steady flow of raw materials for continuous production.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates the need for expensive and hazardous reagents often required in traditional star-shaped compound synthesis. By utilizing a universal palladium catalyst system and common solvents like THF and toluene, the process minimizes raw material expenditures. Additionally, the high selectivity of the reactions reduces the burden on downstream purification, leading to substantial reductions in solvent usage and waste disposal costs. This efficiency allows for a more competitive pricing structure for the final high-purity star-shaped compound without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, well-characterized building blocks such as truxene derivatives and fullerene ensures a resilient supply chain. Unlike proprietary intermediates that may be sourced from single vendors, the precursors used in this method are widely available in the global chemical market. This diversity of supply sources reduces lead time for high-purity electronic chemicals and protects against disruptions caused by geopolitical or logistical issues. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites, further enhancing supply continuity.
- Scalability and Environmental Compliance: The commercial scale-up of complex star-shaped compounds is often hindered by safety concerns regarding high temperatures or pressures. This method operates at moderate temperatures (65-120°C) and atmospheric pressure, significantly reducing the engineering controls required for large-scale reactors. The simplified workup procedures, involving standard extraction and chromatography, are readily adaptable to industrial continuous flow or batch processes. Moreover, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, facilitating smoother regulatory approvals for new production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this trimeric indenyl corrole-porphyrin-fullerene star-shaped compound. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and potential applications for industry stakeholders. Understanding these details is crucial for R&D teams evaluating the material for integration into new device architectures.
Q: What are the primary advantages of this star-shaped molecular structure?
A: The unique three-dimensional star-emitter structure facilitates better regulation of photoelectric properties and morphological characteristics. The outwardly extending support arms significantly improve charge transfer and energy transfer efficiency compared to linear analogues.
Q: How does this method improve upon conventional synthesis routes?
A: Conventional methods often suffer from multiple synthesis steps, complex reactions, and low yields due to difficult separation of products with similar polarity. This novel approach utilizes mild reaction conditions and simple separation methods, drastically simplifying the process.
Q: What are the key applications for this trimeric indenyl compound?
A: Due to its strong light absorption capacity and efficient electron/energy transfer processes, this compound is ideal for use in light absorption antennas, solar cells, artificial simulation of photosynthesis, and liquid crystal materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimeric Indenyl Corrole-Porphyrin-Fullerene Supplier
As the demand for advanced organic electronic materials continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of commercial production. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for high-performance solar cells and optical devices. We understand the critical nature of impurity profiles in photoactive materials and employ advanced analytical techniques to guarantee the quality of every batch delivered.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can be optimized for your specific application requirements. By engaging with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to further enhance process efficiency and reduce overall expenditure. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project timelines. Let us help you accelerate the development of next-generation photoelectric functional materials with our proven expertise in complex organic synthesis.
