Scaling High-Performance Pyrrole Dye Sensitizers for Next-Gen Solar Applications
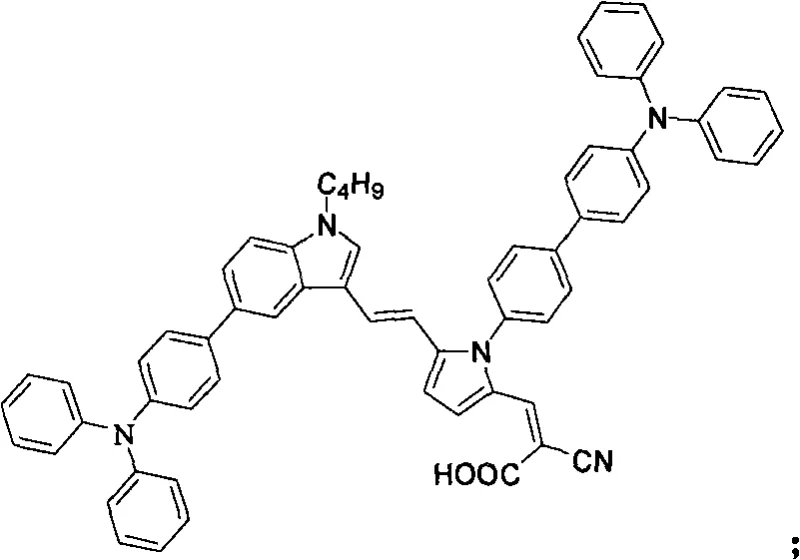
The rapid evolution of renewable energy technologies has placed dye-sensitized solar cells (DSSCs) at the forefront of photovoltaic research, driven by the urgent global need to transition away from fossil fuels. Within this landscape, Patent CN101602759A introduces a groundbreaking class of pyrrole-containing compounds that serve as highly efficient, metal-free organic dye sensitizers. Unlike traditional ruthenium-based complexes which suffer from high costs and limited absorption ranges, these novel pyrrole derivatives exhibit exceptional photoelectric properties and substantial molar absorptivity values, making them ideal candidates for next-generation solar applications. The patent outlines a sophisticated synthetic pathway that leverages the electron-rich nature of the pyrrole ring to enhance intramolecular charge transfer, thereby optimizing the conversion efficiency of solar energy into electricity. For R&D directors and procurement specialists in the electronic chemicals sector, this technology represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of DSSCs has been heavily reliant on metal-organic dye sensitizers, particularly those based on ruthenium polypyridyl complexes. While these materials demonstrated early success, they present significant bottlenecks for large-scale commercialization, primarily due to the scarcity and exorbitant cost of noble metals. Furthermore, metal complexes often exhibit lower molar extinction coefficients compared to their purely organic counterparts, necessitating thicker semiconductor films to capture sufficient light, which can hinder electron transport and increase recombination losses. The synthetic routes for these metal complexes are also frequently complex, requiring stringent anhydrous conditions and expensive ligands, which complicates the supply chain and inflates the overall cost of goods sold. Additionally, the environmental footprint associated with mining and processing heavy metals poses increasing regulatory challenges for manufacturers aiming for green certification.
The Novel Approach
The methodology described in Patent CN101602759A circumvents these issues by utilizing a purely organic framework centered around a pyrrole core, which is inherently electron-rich and chemically versatile. This approach allows for the precise tuning of the HOMO-LUMO energy levels through the strategic attachment of electron-donating groups such as triphenylamine or carbazole via robust carbon-carbon bond-forming reactions. By constructing a D-π-A (Donor-π-Acceptor) architecture, the invention achieves superior light-harvesting capabilities with molar absorptivity values reaching up to 38300 M⁻¹cm¹, significantly outperforming many traditional organic dyes. The synthetic strategy avoids noble metals entirely, relying instead on abundant organic precursors and standard palladium-catalyzed cross-coupling techniques that are well-understood in industrial settings. This shift not only reduces raw material costs but also simplifies the purification process, as there is no need for extensive metal scavenging steps that often plague metal-complex synthesis.
Mechanistic Insights into Multi-Step Organic Synthesis
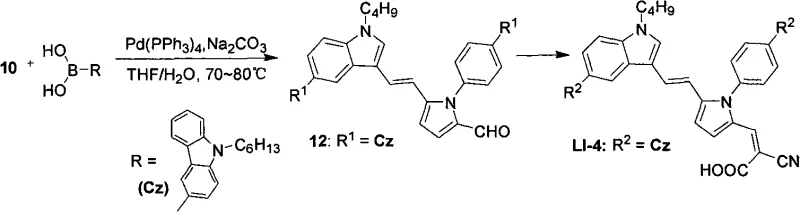
The core of this innovation lies in a meticulously designed five-step synthetic sequence that builds the conjugated system and installs the necessary functional groups with high precision. The process initiates with a Vilsmeier-Haack formylation, where phosphorus oxychloride (POCl₃) and N,N-dimethylformamide (DMF) react under ice-bath conditions to generate a reactive iminium species, which then electrophilically attacks the pyrrole ring to introduce an aldehyde group. This is followed by a Wittig olefination using a phosphonium ylide generated in situ with potassium tert-butoxide (t-BuOK), effectively extending the π-conjugation length crucial for red-shifting the absorption spectrum. A second Vilsmeier formylation is then employed to install a second aldehyde handle, setting the stage for the critical diversification step. The versatility of the platform is realized through a Suzuki-Miyaura cross-coupling reaction, where the brominated intermediate reacts with either triphenylamine monoboronic acid or N-hexylcarbazole-3-boronic acid in the presence of a tetrakis(triphenylphosphine)palladium(0) catalyst. This step is performed at moderate temperatures of 70-80°C in a THF/water biphasic system, ensuring high yields while maintaining functional group tolerance.
The final transformation involves a Knoevenagel condensation between the aldehyde-functionalized intermediate and cyanoacetic acid, catalyzed by piperidine in refluxing acetonitrile. This reaction installs the cyanoacrylic acid anchoring group, which is essential for binding the dye to the titanium dioxide (TiO₂) semiconductor surface. The mechanism proceeds through the formation of an enamine intermediate followed by dehydration to establish the final double bond conjugation. Throughout this sequence, impurity control is managed by leveraging the distinct polarity differences between intermediates, allowing for effective purification via silica gel column chromatography using solvent systems like chloroform or petroleum ether/ethyl acetate mixtures. The absence of sensitive metal-ligand bonds in the final product ensures thermal and photochemical stability, addressing a common failure mode in earlier generations of organic sensitizers.
How to Synthesize Pyrrole-Based Dye Sensitizers Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters outlined in the patent to ensure reproducibility and high purity, particularly regarding the stoichiometry of the Vilsmeier reagents and the inert atmosphere required for the palladium-catalyzed steps. The process is designed to be modular, allowing manufacturers to swap the boronic acid component to tailor the electronic properties of the final dye without altering the core workflow. Detailed standardized operating procedures for each reaction stage, including workup and purification protocols, are essential for maintaining batch-to-batch consistency in a commercial environment.
- Perform Vilsmeier formylation on the pyrrole precursor using POCl3 and DMF under ice-bath conditions to generate the aldehyde intermediate.
- Execute a Wittig reaction between the aldehyde intermediate and a phosphonium salt using t-BuOK in THF to extend the conjugated system.
- Conduct a second Vilsmeier reaction to introduce another aldehyde group, followed by Suzuki coupling with triphenylamine or carbazole boronic acids.
- Finalize the synthesis via Knoevenagel condensation with cyanoacetic acid in acetonitrile using piperidine as a catalyst to yield the target dye.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this pyrrole-based synthesis model offers profound strategic benefits that extend beyond simple unit cost savings. The elimination of ruthenium and other precious metals from the bill of materials immediately de-risks the supply chain from the volatility associated with mining outputs and geopolitical tensions affecting rare earth markets. Moreover, the reliance on commodity chemicals such as DMF, THF, and acetonitrile, alongside readily available heterocyclic building blocks, ensures a stable and diversified supplier base that can support large-volume production without bottlenecking. The mild reaction conditions, particularly the Suzuki coupling which operates at atmospheric pressure and moderate temperatures, reduce the energy intensity of the manufacturing process, contributing to lower utility costs and a smaller carbon footprint per kilogram of product.
- Cost Reduction in Manufacturing: The most significant economic driver is the complete removal of noble metal catalysts from the final product structure, which eliminates the need for expensive and technically challenging metal scavenging resins or activated carbon treatments typically required to meet ppm-level metal specifications in electronic materials. By utilizing organic donors like triphenylamine and carbazole, which are produced at scale for other industries, the raw material costs are substantially lower compared to specialized organometallic ligands. Furthermore, the high yields reported in the patent examples, such as the 74.3% yield in the final Knoevenagel step, minimize waste generation and maximize the throughput of existing reactor infrastructure, leading to a more favorable cost-per-gram metric.
- Enhanced Supply Chain Reliability: The synthetic route relies on robust chemistry that is less sensitive to trace moisture or oxygen compared to organolithium or Grignard reagents, reducing the risk of batch failures due to environmental excursions. The intermediates generated, such as the brominated pyrrole derivatives, are stable solids that can be stockpiled, providing a buffer against supply disruptions for downstream coupling partners. This stability simplifies logistics and warehousing requirements, allowing for just-in-time manufacturing models that reduce inventory carrying costs while ensuring continuous availability for cell assembly lines.
- Scalability and Environmental Compliance: The process utilizes solvents and reagents that are well-established in the fine chemical industry, facilitating straightforward technology transfer from laboratory to pilot and commercial scales without the need for exotic equipment. The aqueous workup steps and the ability to recycle solvents like chloroform and acetonitrile align with modern green chemistry principles, easing the burden on wastewater treatment facilities. Additionally, the absence of heavy metal waste streams simplifies regulatory compliance and disposal costs, making the facility more attractive to investors focused on ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrrole-based dye technology, derived directly from the patent specifications and practical manufacturing considerations. Understanding these nuances is critical for stakeholders evaluating the feasibility of integrating these materials into their existing DSSC production lines.
Q: What are the key advantages of pyrrole-based dyes over traditional metal-complex sensitizers?
A: Pyrrole-based organic dyes offer significantly higher molar absorptivity coefficients compared to metal complexes, allowing for better light harvesting. Furthermore, they eliminate the reliance on expensive noble metals like ruthenium, drastically reducing raw material costs and simplifying the supply chain.
Q: How does the Suzuki coupling step impact the scalability of this process?
A: The Suzuki coupling utilizes robust palladium catalysis under relatively mild conditions (70-80°C), which is highly amenable to scale-up. The use of stable boronic acid intermediates ensures consistent quality and reduces the formation of difficult-to-remove impurities often associated with harsher cross-coupling methods.
Q: What is the expected purity profile for these dye sensitizers?
A: The patented process employs multiple purification stages, including silica gel column chromatography and recrystallization, ensuring high purity. The final products (LI-3 and LI-4) are characterized by rigorous NMR and MS analysis, confirming structural integrity essential for consistent DSSC performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrole Dye Sensitizer Supplier
As the demand for high-efficiency, low-cost photovoltaic materials accelerates, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures that your transition to advanced organic sensitizers is seamless and scalable. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to handle the specific requirements of air-sensitive Suzuki couplings and multi-step purifications. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced HPLC and NMR techniques to verify the structural integrity and optical properties of every batch, ensuring they meet the exacting standards required for electronic applications.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential ROI of switching to our pyrrole-based platform. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your R&D and supply chain teams to make informed decisions for the future of your solar energy projects.
