Advanced Rhodium-Catalyzed Synthesis of Trifluoromethyl-Substituted Enamine Compounds for Pharmaceutical Applications
Advanced Rhodium-Catalyzed Synthesis of Trifluoromethyl-Substituted Enamine Compounds for Pharmaceutical Applications
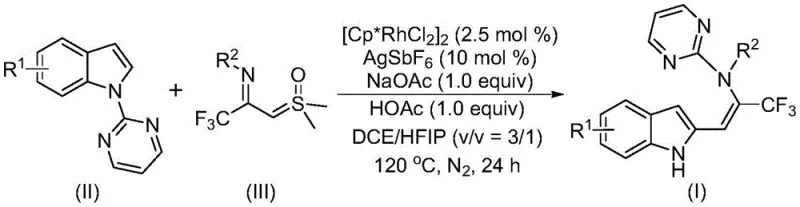
The pharmaceutical industry constantly seeks efficient pathways to construct complex molecular scaffolds that possess high biological activity. A significant breakthrough in this domain is detailed in Chinese Patent CN115925692A, which discloses a novel preparation method for trifluoromethyl-substituted enamine compounds. This technology leverages a sophisticated rhodium-catalyzed carbon-hydrogen (C-H) activation strategy to couple readily available indole derivatives with trifluoroacetimide sulfur ylides. The resulting multifunctionalized enamine compounds are critical building blocks in medicinal chemistry, particularly because the incorporation of a trifluoromethyl group can drastically improve the metabolic stability, lipophilicity, and bioavailability of drug candidates. By enabling the direct functionalization of the indole core without extensive pre-modification, this patent offers a streamlined route for generating diverse libraries of bioactive molecules, addressing a key bottleneck in early-stage drug discovery and process development for reliable pharmaceutical intermediates supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of enamine functional groups onto indole skeletons has relied heavily on transition metal-catalyzed C-H activation reactions that require pre-installed directing groups on the indole substrate. While effective in specific contexts, these conventional methodologies suffer from significant limitations regarding structural diversity and synthetic efficiency. The necessity of installing and subsequently removing directing groups adds multiple synthetic steps, increasing both the cost of goods sold and the environmental footprint of the manufacturing process. Furthermore, traditional routes often utilize ketene imines or other specialized substrates that limit the scope of accessible chemical space, restricting the ability of chemists to explore novel structure-activity relationships. These constraints make it difficult to achieve the high throughput required for modern drug discovery programs, where rapid access to diverse analogues is paramount for identifying lead compounds with optimal pharmacological profiles.
The Novel Approach
In stark contrast to legacy methods, the technology described in CN115925692A introduces a highly efficient tandem reaction sequence involving C-H activation followed by a directing group migration. This innovative approach utilizes cheap and readily available indole compounds and trifluoroacetimide sulfur ylides as starting materials, bypassing the need for complex substrate pre-functionalization. The reaction proceeds through a rhodium-catalyzed mechanism where the pyrimidine nitrogen acts as a transient directing group, facilitating C-C bond formation before migrating to its final position via a 1,5-shift. This strategy not only simplifies the synthetic route but also exhibits exceptional functional group tolerance, allowing for the incorporation of various substituents such as halogens, alkyl groups, and alkoxy groups on both the indole and the aryl rings. The versatility of this method is evidenced by the successful synthesis of numerous derivatives with yields frequently exceeding 80%, establishing it as a superior platform for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Migration
The core of this technological advancement lies in the precise orchestration of organometallic steps mediated by the dichlorocyclopentyl rhodium(III) dimer catalyst. The mechanism initiates with the coordination of the rhodium species to the pyrimidine nitrogen of the sulfur ylide reagent, which directs the activation of the C-H bond at the C2 or C3 position of the indole ring depending on the specific substrate geometry. Following C-H cleavage, the rhodium-carbene species generated from the sulfur ylide undergoes insertion to form a new carbon-carbon bond. Crucially, the reaction does not stop at this stage; instead, it proceeds through an isomerization to form an initial enamine intermediate. The defining feature of this chemistry is the subsequent intramolecular migration of the pyrimidine group. This 1,5-migration reaction is thermodynamically driven and results in the final stable trifluoromethyl-substituted enamine product. Understanding this mechanistic pathway is vital for R&D teams aiming to optimize reaction parameters, as the balance between C-H activation kinetics and migration rates determines the overall selectivity and purity of the final API intermediate.
Impurity control in this synthesis is managed through careful selection of the solvent system and additives. The patent highlights the use of a mixed solvent system comprising dichloroethane (DCE) and hexafluoroisopropanol (HFIP), which plays a dual role in solubilizing the reactants and stabilizing the cationic rhodium intermediates. Additionally, the inclusion of sodium acetate and acetic acid as additives helps to buffer the reaction medium and facilitate the proton transfer steps essential for the catalytic cycle. By maintaining strict control over the molar ratios of the catalyst, promoter (silver hexafluoroantimonate), and additives, the process minimizes side reactions such as homocoupling or decomposition of the sensitive sulfur ylide. This rigorous control over the reaction environment ensures that the resulting high-purity OLED material or pharmaceutical precursor meets the stringent quality standards required for downstream applications, reducing the burden on purification processes.
How to Synthesize Trifluoromethyl-Substituted Enamine Efficiently
To implement this synthesis effectively, operators must adhere to the specific stoichiometric ratios and thermal conditions outlined in the patent data. The process begins by charging a reaction vessel with the rhodium catalyst, silver salt promoter, and acetate additives under an inert atmosphere. Subsequently, the indole substrate and the trifluoroacetimide sulfur ylide are introduced into the DCE/HFIP solvent mixture. The reaction mixture is then heated to a temperature range of 110-130°C, typically maintained at 120°C, for a duration of 18 to 30 hours. Monitoring the reaction progress is essential to ensure complete consumption of the starting materials while avoiding prolonged heating that could degrade the product. Upon completion, the workup involves simple filtration to remove metal salts, followed by silica gel treatment and column chromatography to isolate the pure enamine compound. For a detailed step-by-step guide on optimizing these parameters for your specific substrate, please refer to the standardized protocol below.
- Combine dichlorocyclopentyl rhodium(III) dimer catalyst, silver hexafluoroantimonate promoter, and sodium acetate/acetic acid additives in a reaction vessel.
- Add the indole compound substrate and trifluoroacetimide sulfur ylide reagent into an organic solvent mixture of dichloroethane and hexafluoroisopropanol.
- Heat the reaction mixture to 110-130°C for 18-30 hours under nitrogen atmosphere, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthesis method offers transformative advantages that directly impact the bottom line and operational resilience. The primary driver of value is the utilization of commodity chemicals as starting materials. Indoles and aromatic amines, which are precursors to the sulfur ylide, are produced on a massive global scale, ensuring a stable and competitive supply base. This reliance on bulk chemicals rather than bespoke, multi-step intermediates significantly de-risks the supply chain against shortages and price volatility. Furthermore, the high atom economy of the tandem reaction means that a larger proportion of the raw material mass is incorporated into the final product, reducing waste disposal costs and improving the overall sustainability profile of the manufacturing operation. For supply chain heads, this translates to a more predictable and reliable sourcing strategy for complex organic building blocks.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps for the indole substrate represents a substantial reduction in processing time and reagent costs. Traditional methods often require protecting group chemistry and separate installation of directing groups, each adding labor, solvent, and purification expenses. By consolidating these steps into a single catalytic pot, the new method drastically simplifies the process flow. Additionally, the high yields reported (often above 80%) mean that less raw material is required to produce a given amount of product, further driving down the unit cost. This efficiency allows for significant cost savings in the production of high-value pharmaceutical intermediates without compromising on quality or purity specifications.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes directly to supply continuity. The reaction tolerates a wide range of functional groups, meaning that variations in raw material quality or slight changes in substrate structure do not necessitate a complete re-optimization of the process. This flexibility is crucial for maintaining production schedules when facing supply fluctuations. Moreover, the reagents used, such as the rhodium catalyst and silver salts, are standard items in the fine chemical industry, ensuring that replenishment is straightforward. This reliability reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and accelerating the timeline from clinical trials to commercial launch.
- Scalability and Environmental Compliance: The patent explicitly notes that the reaction can be expanded to gram-level scales with high efficiency, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates. The use of standard organic solvents like dichloroethane and established purification techniques like column chromatography means that existing manufacturing infrastructure can be adapted with minimal capital expenditure. From an environmental standpoint, the high efficiency and reduced step count lower the overall E-factor (mass of waste per mass of product), aligning with green chemistry principles. This makes the process not only economically attractive but also compliant with increasingly stringent environmental regulations governing chemical manufacturing facilities globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of this Rhodium-catalyzed method over traditional enamine synthesis?
A: This method utilizes a tandem C-H activation and directing group migration strategy, eliminating the need for pre-functionalized indole substrates. It offers superior structural diversity, high functional group tolerance, and achieves yields often exceeding 80%, significantly streamlining the production of complex pharmaceutical intermediates.
Q: Is this synthesis protocol scalable for commercial manufacturing?
A: Yes, the patent explicitly demonstrates that the reaction can be effectively expanded to gram-level scales with consistent efficiency. The use of commercially available starting materials and standard purification techniques like column chromatography indicates strong potential for kilogram-to-ton scale-up in industrial settings.
Q: What specific solvents and conditions are critical for optimal yield?
A: Optimal results are achieved using a mixed solvent system of dichloroethane (DCE) and hexafluoroisopropanol (HFIP) at a volume ratio of roughly 3:1. The reaction requires heating to 120°C for approximately 24 hours under a nitrogen atmosphere to ensure complete conversion and high purity of the trifluoromethyl-substituted product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl-Substituted Enamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in CN115925692A for the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into robust manufacturing processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific enamine derivatives or large-scale supply of key intermediates, our infrastructure is designed to meet the exacting demands of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug discovery programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, helping you identify opportunities for efficiency and budget optimization. We encourage you to contact our technical procurement team today to request specific COA data for our available catalog items or to discuss route feasibility assessments for your proprietary targets. Let us be your trusted partner in navigating the complexities of modern chemical synthesis and bringing life-saving medicines to market faster.
