Optimizing MDL-100907 Intermediate Production via Novel PCl3 Amidation Technology
The pharmaceutical landscape for central nervous system disorders continues to evolve, with high-selectivity 5-HT2A receptor antagonists like MDL-100907 playing a pivotal role in treating schizophrenia and depression. The efficient production of such complex molecules relies heavily on the availability of high-purity intermediates. Patent CN102491941B introduces a groundbreaking preparation method for N-methoxy-N-methyl-1-p-toluenesulfonylpiperidine-4-amide, a critical Weinreb amide derivative essential for the downstream synthesis of MDL-100907. This technology addresses long-standing bottlenecks in intermediate manufacturing by replacing unstable reagents with a robust, one-pot amidation strategy using phosphorus trichloride. For global procurement teams, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality without the volatility associated with older synthetic routes.
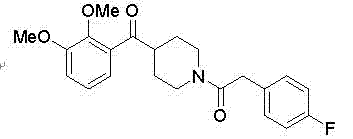
Understanding the molecular architecture of the final API is crucial for appreciating the complexity of the intermediate synthesis. As illustrated in the structural diagram, the target molecule requires precise stereochemical control and functional group tolerance. The intermediate in question serves as the electrophilic partner in a Grignard addition, necessitating exceptional purity to prevent side reactions that could compromise the final drug substance. By optimizing the synthesis of this specific amide, manufacturers can ensure a smoother flow of materials through the value chain, ultimately supporting the production of life-saving medications for neurological conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Weinreb amides has relied on activating carboxylic acids into acid chlorides or utilizing specialized coupling agents that often introduce significant operational hazards. Prior art, such as the methods described in Chinese patent CN101665458, utilized acylation systems involving NBS/PPh3 or trialkyl phosphites like P(NMeOMe)3. While these methods theoretically allow for direct amidation, they are plagued by the inherent instability of the phosphoramidite reagents. These reagents are notoriously difficult to separate and purify, leading to inconsistent batch quality and increased waste generation. Furthermore, the requirement for pre-activation of the carboxyl group or the use of expensive catalysts drives up the cost of goods sold, making cost reduction in API manufacturing difficult to achieve with legacy technologies.
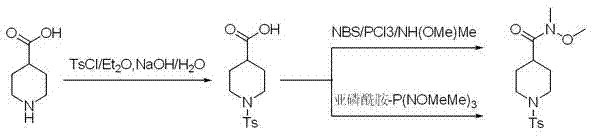
In addition to reagent instability, conventional routes often involve multi-step sequences that increase the overall lead time for production. The isolation of reactive intermediates like acid chlorides requires stringent moisture control and specialized equipment, adding capital expenditure requirements for manufacturing facilities. For supply chain managers, these complexities translate into higher risks of production delays and potential supply interruptions. The accumulation of phosphorous-containing byproducts also complicates wastewater treatment, posing environmental compliance challenges that can halt production in regions with strict regulatory oversight. These factors collectively undermine the economic viability of traditional synthesis paths for high-volume commercial applications.
The Novel Approach
The innovative methodology disclosed in CN102491941B circumvents these issues by employing a direct amidation strategy using industrial-grade PCl3 and N-methyl-N-methoxyamine. This approach eliminates the need to isolate unstable phosphoramidite intermediates, effectively collapsing multiple reaction steps into a single, streamlined operation. By reacting 1-toluenesulfonylpiperidine-4-carboxylic acid directly with the amidating reagents in a one-pot fashion, the process drastically simplifies the workflow. This not only shortens the reaction cycle but also minimizes the handling of hazardous materials, thereby enhancing workplace safety. The result is a robust manufacturing protocol that delivers high yields with minimal purification effort, setting a new standard for efficiency in fine chemical synthesis.
Furthermore, this novel route utilizes raw materials that are commoditized and readily available on the global market, reducing dependency on niche suppliers. The simplicity of the post-treatment process, which involves standard extraction and drying techniques, allows for easy adaptation to existing manufacturing infrastructure without the need for major retrofits. For organizations seeking the commercial scale-up of complex pharmaceutical intermediates, this technology offers a clear pathway to increasing capacity while maintaining rigorous quality standards. The elimination of difficult separation steps ensures that the final product meets stringent purity specifications required for GMP environments, facilitating faster regulatory approval and market entry.
Mechanistic Insights into PCl3-Mediated Direct Amidation
The core of this technological advancement lies in the activation mechanism facilitated by phosphorus trichloride. In this system, PCl3 acts as a potent dehydrating and activating agent, likely generating an acyl chloride species in situ which immediately reacts with the N-methyl-N-methoxyamine. This transient activation avoids the accumulation of free acid chloride, which can be prone to hydrolysis or side reactions if isolated. The reaction proceeds through a nucleophilic attack by the amine on the activated carbonyl carbon, forming the stable Weinreb amide bond. The stoichiometry is carefully balanced, typically employing a molar ratio of 2:1:6 for the acid, PCl3, and amine respectively, to ensure complete conversion while minimizing excess reagent waste.
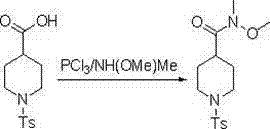
Impurity control is inherently built into this mechanistic design. By avoiding the use of phosphoramidites, the process prevents the formation of complex phosphorous oxide byproducts that are notoriously difficult to remove via crystallization or chromatography. The byproducts generated from PCl3, primarily phosphorous acid derivatives, are water-soluble and are effectively removed during the aqueous workup phase involving saturated sodium bicarbonate quenching. This results in a crude product of high purity, reducing the burden on downstream purification units. For R&D directors focused on impurity profiles, this mechanism offers a cleaner reaction trajectory that simplifies analytical validation and ensures the high-purity pharmaceutical intermediate meets all necessary identity and assay criteria.
How to Synthesize N-Methoxy-N-Methyl-1-P-Toluenesulfonyl Piperidine-4-Amide Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control and solvent selection. The process begins with the preparation of the starting acid, followed by the critical amidation step where reagents are mixed under anhydrous conditions. The choice of solvent, such as toluene or dichloromethane, influences the reaction kinetics and heat dissipation. Detailed standard operating procedures regarding mixing rates, addition sequences, and quenching protocols are essential to replicate the high yields observed in the patent examples. The following guide outlines the standardized synthesis steps derived from the patented methodology to ensure reproducible results in a pilot or production setting.
- Prepare the starting material 1-toluenesulfonylpiperidine-4-carboxylic acid by reacting piperidine-4-carboxylic acid with TsCl in an alkaline aqueous solution, followed by acidification and drying.
- In an anhydrous aprotic solvent such as toluene or dichloromethane, mix the carboxylic acid with N-methyl-N-methoxyamine at 0°C to form the reaction mixture.
- Slowly add PCl3 solution dropwise, stir at 0°C to 60°C for 1 to 5 hours, then quench with saturated sodium bicarbonate, extract with ethyl acetate, and dry to obtain the pure amide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this PCl3-mediated synthesis offers profound advantages for procurement strategies and supply chain resilience. The shift away from exotic, unstable reagents to commodity chemicals fundamentally alters the cost structure of the intermediate. By utilizing industrial-grade PCl3 and simple amine salts, manufacturers can decouple their production costs from the volatile pricing of specialized coupling agents. This stability in raw material sourcing translates directly into more predictable budgeting and reduced exposure to supply shocks. Additionally, the simplified workflow reduces labor hours and utility consumption per kilogram of product, driving down the overall manufacturing overhead without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the reduction in processing steps lead to substantial cost savings. Since the process avoids the isolation of intermediates, there is a significant decrease in solvent usage and energy consumption associated with multiple distillation or crystallization cycles. The high atom economy of the direct amidation ensures that a larger proportion of raw materials end up in the final product, minimizing waste disposal costs. These efficiencies compound to offer a highly competitive price point for bulk purchasers seeking long-term supply agreements.
- Enhanced Supply Chain Reliability: Relying on widely available reagents like PCl3 and toluene mitigates the risk of single-source supplier dependency. Unlike specialized phosphoramidites which may have limited global production capacity, the inputs for this process are produced by numerous chemical manufacturers worldwide. This diversity in the supply base ensures continuity of supply even during regional disruptions. Furthermore, the robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites with minimal technology transfer friction, enhancing overall network flexibility.
- Scalability and Environmental Compliance: The process is inherently green, generating fewer hazardous byproducts and utilizing less toxic reagents compared to traditional methods. The aqueous workup effectively removes inorganic salts, resulting in an organic phase that is easier to concentrate and dry. This simplicity facilitates seamless scale-up from laboratory grams to multi-tonne commercial batches. The reduced environmental footprint aligns with modern sustainability goals, helping companies meet their corporate social responsibility targets while maintaining regulatory compliance in strict jurisdictions.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and application of this intermediate. These responses are grounded in the experimental data and technical disclosures found within the patent literature. Understanding these details is vital for assessing the feasibility of integrating this route into your existing supply chain. The following section addresses key concerns regarding reagent stability, reaction optimization, and scalability potential.
Q: Why is the PCl3 method superior to phosphoramidite-based synthesis?
A: Traditional methods using phosphoramidites like P(NMeOMe)3 suffer from reagent instability and difficult separation processes. The PCl3 method avoids isolating unstable intermediates, simplifying purification and significantly improving operational safety and yield consistency.
Q: What are the optimal reaction conditions for this amidation?
A: The reaction proceeds efficiently in anhydrous aprotic solvents like toluene or dichloromethane. The optimal temperature range is between 0°C and 60°C, with a reaction time of 1 to 5 hours depending on the specific solvent and thermal profile chosen.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes cheap, readily available raw materials and involves a simple post-treatment workflow involving standard extraction and drying. The high yields reported (over 90%) and lack of toxic byproducts make it highly scalable and environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methoxy-N-Methyl-1-P-Toluenesulfonyl Piperidine-4-Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable intermediates in the development of novel therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-methoxy-N-methyl-1-p-toluenesulfonylpiperidine-4-amide meets the highest industry standards. Our commitment to quality and consistency makes us an ideal partner for pharmaceutical companies aiming to optimize their MDL-100907 supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this PCl3-mediated process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence. Let us collaborate to drive efficiency and innovation in your pharmaceutical manufacturing operations.
