Revolutionizing Quinolinyl Ketone Synthesis: Scalable Pd-Catalyzed Process for Pharmaceutical Intermediates Manufacturing
The recently granted Chinese patent CN113214149B introduces a groundbreaking synthetic methodology for phenyl(quinolin-8-yl)methanone derivatives, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovation addresses longstanding challenges in quinoline chemistry by establishing a direct C-H activation pathway that circumvents the need for pre-functionalized substrates. The process achieves remarkable efficiency through a carefully engineered palladium-catalyzed system operating under precisely controlled conditions, delivering yields up to 87% while maintaining exceptional selectivity. This development holds particular relevance for the pharmaceutical industry where these structural motifs serve as essential building blocks for bioactive compounds targeting cancer, bone metabolism disorders, and neurological conditions. The patent's emphasis on operational simplicity and raw material accessibility positions this technology as a strategic solution for manufacturers seeking to enhance both process economics and supply chain resilience in the production of complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 8-benzoylquinoline derivatives have been severely constrained by their reliance on expensive rhodium or ruthenium catalysts coupled with specialized starting materials such as 8-formylquinoline or quinoline carboxylates. These approaches suffer from multiple critical limitations including high catalyst costs that significantly impact commercial viability, limited substrate availability that creates supply chain vulnerabilities, and complex multi-step sequences that reduce overall process efficiency. The requirement for pre-functionalized quinoline derivatives introduces additional purification challenges and increases the risk of impurity formation, particularly problematic for pharmaceutical applications where stringent purity specifications must be met. Furthermore, these conventional methods often operate under harsh conditions that complicate scale-up and increase energy consumption, making them economically unattractive for large-scale manufacturing despite their scientific merit. The cumulative effect of these constraints has created a persistent bottleneck in the production of these valuable intermediates.
The Novel Approach
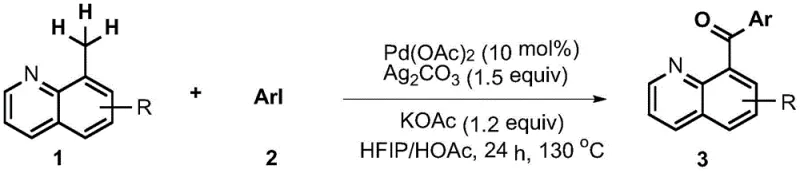
The patented methodology overcomes these limitations through an elegant palladium-catalyzed C-H activation strategy that directly transforms readily available 8-methylquinoline derivatives and aryl iodides into target products. By employing cost-effective palladium acetate as the catalyst in combination with silver carbonate as an additive, the process eliminates dependence on precious metal catalysts while maintaining high reaction efficiency. The carefully optimized solvent system comprising hexafluoroisopropanol and glacial acetic acid at a precise 3:7 volume ratio creates an ideal reaction environment that enhances catalyst stability and substrate solubility. Operating at a controlled temperature of 130°C for 24 hours in sealed tubes ensures consistent product formation without requiring specialized equipment, making this approach exceptionally well-suited for industrial implementation. The simplicity of the workup procedure involving standard extraction and chromatography further enhances its practical utility for commercial manufacturing operations.
Mechanistic Insights into Palladium-Catalyzed C-H Activation
The reaction proceeds through a sophisticated catalytic cycle initiated by oxidative addition of aryl iodide to palladium(0), generated in situ from palladium(II) acetate reduction. This key step forms an aryl-palladium intermediate that subsequently undergoes regioselective C-H activation at the methyl group position of the quinoline substrate, facilitated by the nitrogen atom's coordination to palladium. The resulting organopalladium species then participates in a reductive elimination process that forms the new carbon-carbon bond while regenerating the active catalyst species. Silver carbonate plays a crucial role as an oxidant to maintain the catalytic cycle by reoxidizing palladium(0) back to palladium(II), while potassium acetate serves as a base to facilitate deprotonation during the C-H activation step. The unique solvent mixture provides optimal polarity to stabilize all intermediate species while preventing catalyst decomposition throughout the reaction sequence.
Impurity control is achieved through the precise regulation of reaction parameters that minimize side reactions such as homocoupling or over-oxidation. The solvent system's ability to dissolve both organic substrates and inorganic additives ensures homogeneous reaction conditions that prevent localized concentration gradients which could lead to impurity formation. The controlled temperature profile at 130°C avoids thermal decomposition pathways while maintaining sufficient energy for the C-H activation step, resulting in cleaner product profiles as evidenced by the high yields reported across diverse substrates. The workup procedure involving aqueous sodium carbonate wash effectively removes acidic byproducts without affecting product integrity, while column chromatography provides final purification to meet pharmaceutical-grade purity requirements without requiring specialized techniques.
How to Synthesize Quinolinyl Ketone Derivatives Efficiently
This patented synthetic route represents a significant advancement in the manufacturing of quinolinyl ketone derivatives, offering both scientific elegance and practical manufacturing advantages over conventional approaches. The methodology provides a direct pathway from commercially accessible starting materials to high-value pharmaceutical intermediates through a single catalytic transformation that eliminates multiple synthetic steps required by traditional methods. Detailed standardized synthesis procedures have been developed based on this patent technology to ensure consistent product quality and yield across different manufacturing scales. The following step-by-step guide outlines the critical operational parameters necessary for successful implementation in industrial settings, with specific attention to process control points that ensure optimal performance and product quality.
- Combine 0.1 mmol of substituted 8-methylquinoline with 0.15 mmol aryl iodide and catalyst system comprising Pd(OAc)₂ (0.01 mmol) and Ag₂CO₃ (0.15 mmol) in a pressure-resistant tube with magnetic stirrer.
- Add potassium acetate (0.12 mmol) and mixed solvent system of hexafluoroisopropanol and glacial acetic acid at volume ratio 3: 7 to achieve homogeneous reaction medium.
- Seal reaction tube and heat at precisely controlled 130°C for 24 hours under inert atmosphere, followed by standard workup including ethyl acetate dilution and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits specifically tailored to address procurement and supply chain challenges in pharmaceutical intermediate manufacturing. By replacing expensive transition metal catalysts with cost-effective palladium systems and utilizing readily available starting materials, the process fundamentally transforms the economic landscape for producing these critical intermediates. The elimination of complex multi-step sequences reduces both capital investment requirements and operational complexity, creating significant opportunities for cost optimization throughout the manufacturing value chain. These advantages translate directly into enhanced competitiveness for manufacturers while providing procurement teams with greater flexibility in supplier selection and negotiation leverage.
- Cost Reduction in Manufacturing: The substitution of rhodium or ruthenium catalysts with palladium acetate creates substantial cost savings through reduced catalyst expenditure and elimination of expensive metal removal processes from downstream purification. The use of commercially available 8-methylquinoline derivatives instead of specialized aldehydes or esters significantly lowers raw material costs while simplifying supply chain management. The single-step transformation reduces solvent consumption, energy requirements, and labor costs compared to traditional multi-stage syntheses, collectively contributing to meaningful operational cost reductions without compromising product quality or yield.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials eliminates critical supply chain vulnerabilities associated with specialized precursors required by conventional methods. This approach provides procurement teams with multiple sourcing options for key raw materials, reducing dependency on single suppliers and mitigating risk from potential supply disruptions. The simplified process design enables faster response times to changing demand patterns while maintaining consistent product quality, allowing manufacturers to better meet customer requirements without inventory overstocking or production delays.
- Scalability and Environmental Compliance: The robust reaction conditions and straightforward workup procedure facilitate seamless scale-up from laboratory to commercial production volumes without requiring specialized equipment modifications. The elimination of hazardous reagents and reduction in solvent usage compared to traditional methods significantly lowers environmental impact while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices. This environmentally responsible approach supports corporate sustainability initiatives while maintaining economic viability through reduced waste treatment costs and improved resource efficiency.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding the implementation of this patented synthesis methodology for quinolinyl ketone derivatives. These responses are based on detailed analysis of the patent documentation and practical manufacturing experience with similar catalytic systems, providing actionable insights for procurement, R&D, and supply chain decision-makers evaluating this technology for commercial adoption.
Q: How does this method overcome limitations of conventional Rh/Ru-catalyzed approaches?
A: The novel Pd(OAc)₂-catalyzed system eliminates reliance on expensive rhodium or ruthenium catalysts while maintaining high regioselectivity. By utilizing readily available 8-methylquinoline derivatives instead of specialized aldehydes or esters, it addresses the critical supply chain vulnerability of precursor scarcity that plagues traditional methods, as documented in the patent background.
Q: What cost reduction mechanisms are enabled by the simplified reaction pathway?
A: The elimination of transition metal purification steps through the use of palladium acetate instead of precious metal catalysts significantly reduces downstream processing costs. The single-step transformation from commercially accessible starting materials avoids multi-stage syntheses, thereby lowering both raw material expenses and operational complexity in manufacturing environments.
Q: How does the solvent system enhance scalability for commercial production?
A: The hexafluoroisopropanol/acetic acid (3:7 v/v) mixture provides optimal solubility for both organic substrates and inorganic additives while maintaining thermal stability at 130°C. This homogeneous reaction medium prevents catalyst deactivation and facilitates consistent heat transfer during scale-up, directly supporting reliable transition from laboratory to industrial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl(quinolin-8-yl)methanone Derivatives Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates like phenyl(quinolin-8-yl)methanone derivatives. Our technical expertise in palladium-catalyzed transformations ensures consistent delivery of products meeting stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. We have successfully implemented this patented methodology across multiple production scales while maintaining exceptional product quality and regulatory compliance, providing pharmaceutical manufacturers with reliable access to these critical building blocks for drug development pipelines.
Engage with our technical procurement team today to request a Customized Cost-Saving Analysis demonstrating how this innovative synthesis can optimize your supply chain economics. We provide comprehensive support including specific COA data, route feasibility assessments, and dedicated technical consultation to ensure seamless integration into your manufacturing operations while maximizing value creation throughout your procurement lifecycle.
