Advanced Pyrene Helicene Synthesis: Enabling Commercial Scale-up of Complex Display Intermediates
The rapid evolution of the organic electronics sector demands increasingly sophisticated molecular architectures capable of delivering superior optoelectronic performance. In this context, Patent CN111233616A introduces a groundbreaking synthetic methodology for a class of pyrene[4]helicene derivatives, which represent a significant advancement in the field of luminescent materials and chemical sensing. These non-planar polycyclic aromatic hydrocarbons possess extended π-conjugated systems that offer exceptional thermal stability and high electron carrying capacity, making them ideal candidates for next-generation display technologies. The patent details a robust, metal-free approach that circumvents the traditional reliance on toxic heavy metal catalysts, thereby aligning with modern green chemistry principles while maintaining high structural fidelity. By leveraging a combination of Wittig olefination and subsequent photochemical cyclization, this technology provides a streamlined pathway to access complex chiral luminescent materials that were previously difficult to manufacture efficiently. For industry leaders seeking a reliable OLED material supplier, understanding the nuances of this synthetic route is critical for securing a competitive edge in the supply of high-purity display intermediates.
![General structural formula of pyrene[4]helicene derivatives showing variable R groups](/insights/img/pyrene-helicene-synthesis-oled-supplier-20260302194036-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of helicenes and related polycyclic aromatic compounds has been heavily dependent on transition metal-catalyzed coupling reactions, which present substantial drawbacks for large-scale industrial application. These conventional methods often necessitate the use of expensive palladium or rhodium catalysts, which not only inflate the raw material costs but also introduce significant challenges regarding residual metal contamination in the final product. For applications in sensitive electronic devices such as OLEDs, even trace amounts of metal impurities can drastically reduce device lifetime and efficiency, requiring rigorous and costly purification steps to meet stringent quality specifications. Furthermore, many traditional synthetic routes involve harsh reaction conditions, including extreme temperatures and pressures, which pose safety risks and complicate the engineering controls required for commercial scale-up of complex polymer additives and electronic chemicals. The environmental burden associated with the disposal of heavy metal waste streams further diminishes the sustainability profile of these legacy processes, creating regulatory hurdles for manufacturers aiming to reduce their carbon footprint.
The Novel Approach
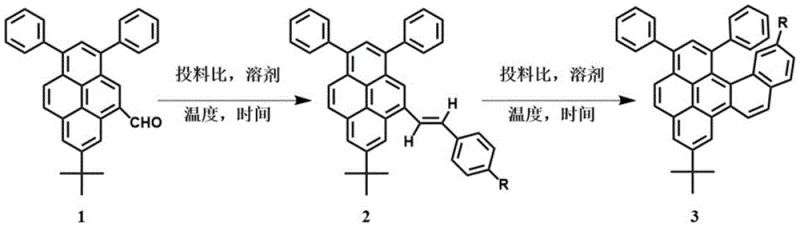
In stark contrast, the methodology disclosed in the patent utilizes a photochemical cyclization strategy that effectively bypasses the need for transition metal catalysts, offering a cleaner and more economically viable alternative for cost reduction in electronic chemical manufacturing. The process begins with a Wittig reaction to install a vinyl bridge, followed by an oxidative photocyclization mediated by elemental iodine and propylene oxide, which serves as an acid scavenger to drive the reaction forward. This metal-free approach not only simplifies the reaction workflow but also ensures that the final helicene products are free from catalytic metal residues, thereby enhancing their suitability for high-performance optoelectronic applications. The use of common commercial reagents such as tetrahydrofuran, benzene, and iodine ensures that the supply chain remains robust and less susceptible to the volatility often seen with specialized organometallic reagents. Additionally, the reaction conditions are remarkably mild, typically proceeding at ambient temperatures around 25°C, which reduces energy consumption and allows for safer operation in standard chemical processing facilities without the need for specialized high-pressure equipment.
Mechanistic Insights into Photochemical Cyclization and Wittig Olefination
The core of this synthetic innovation lies in the precise orchestration of two distinct chemical transformations that build the complex helicene framework with high regioselectivity. The initial step involves the generation of a phosphorus ylide from benzyltriphenylphosphonium chloride using n-butyllithium in anhydrous tetrahydrofunder an inert argon atmosphere, which then attacks the carbonyl group of the 7-tert-butyl-1,3-diphenyl-5-aldehyde pyrene precursor. This Wittig olefination proceeds through a four-membered oxaphosphetane intermediate that collapses to form the desired styryl double bond, establishing the necessary connectivity for the subsequent ring closure. Optimization data indicates that maintaining a molar ratio of 2:2:1 for the phosphonium salt, base, and aldehyde at 25°C for 16 hours yields the best balance between conversion and selectivity, minimizing the formation of side products that could complicate downstream purification. The resulting stilbene-like intermediate possesses the correct geometric alignment to undergo intramolecular cyclization upon exposure to light, a process that is energetically favorable due to the relief of steric strain and the formation of a stable aromatic system.
Following the formation of the vinyl intermediate, the second critical stage involves an oxidative photocyclization where elemental iodine acts as both an oxidant and a radical initiator under light irradiation. The presence of propylene oxide is mechanistically crucial as it scavenges the hydrogen iodide byproduct generated during the aromatization step, preventing acid-catalyzed degradation of the sensitive helicene structure and driving the equilibrium towards the fully aromatic product. This step effectively locks the molecule into its characteristic helical conformation, imparting the unique chiroptical properties that make these compounds valuable for circularly polarized luminescence applications. Impurity control is managed through a straightforward workup procedure involving extraction with dichloromethane and washing with saturated brine, followed by drying over anhydrous magnesium sulfate to remove residual water. Final purification is achieved via column chromatography on silica gel using a hexane and chloroform mixture, which effectively separates the target helicene from unreacted starting materials and iodine byproducts, ensuring the high-purity standards required for electronic grade materials.
How to Synthesize Pyrene[4]helicene Efficiently
To successfully implement this synthesis in a laboratory or pilot plant setting, operators must adhere to strict anhydrous conditions during the Wittig step to prevent quenching of the reactive ylide species by moisture. The subsequent photocyclization requires careful control of light intensity and reaction time, with experimental data suggesting that an 8-hour irradiation period provides optimal conversion without excessive decomposition of the product. Detailed standardized operating procedures for scaling this route from gram to kilogram quantities are essential to maintain consistency in yield and purity across different production batches.
- Perform a Wittig reaction between 7-tert-butyl-1,3-diphenyl-5-aldehyde pyrene and benzyltriphenylphosphonium chloride using n-butyllithium in THF at 25°C for 16 hours.
- Conduct oxidative photocyclization on the resulting styryl intermediate using elemental iodine and propylene oxide in benzene under light irradiation for 8 hours.
- Purify the crude reaction mixture through extraction, drying, and column chromatography using a hexane/chloroform eluent system to isolate the target helicene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility, directly impacting the bottom line through operational efficiencies. The elimination of precious metal catalysts removes a major cost driver and supply risk, as the market for palladium and similar metals is often subject to geopolitical instability and price fluctuations that can disrupt production schedules. By relying on abundant and inexpensive reagents like iodine and phosphonium salts, manufacturers can achieve significant cost reduction in manufacturing while insulating their operations from raw material volatility. Furthermore, the stability of the starting materials, which do not require cryogenic storage or special handling precautions, simplifies warehouse management and reduces the overhead costs associated with hazardous material compliance.
- Cost Reduction in Manufacturing: The absence of expensive transition metal catalysts fundamentally alters the cost structure of producing these advanced intermediates, allowing for more competitive pricing models in the global market. Without the need for specialized ligands or metal recovery systems, the capital expenditure required for setting up production lines is substantially lowered, making the technology accessible to a broader range of chemical manufacturers. The simplified purification process also reduces solvent consumption and waste disposal fees, contributing to a leaner and more profitable operational model that maximizes resource utilization.
- Enhanced Supply Chain Reliability: Sourcing common organic solvents and reagents ensures a resilient supply chain that is less vulnerable to the bottlenecks often experienced with niche specialty chemicals. The robustness of the synthetic route means that production can be ramped up quickly to meet surging demand from the display and lighting sectors without the long lead times associated with custom catalyst synthesis. This reliability is crucial for maintaining continuous production schedules for downstream clients who depend on just-in-time delivery of high-quality electronic materials to keep their own assembly lines running smoothly.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of toxic heavy metals make this process inherently easier to scale from benchtop to industrial reactors while meeting increasingly strict environmental regulations. Facilities can operate with reduced ventilation requirements and simpler effluent treatment systems, lowering the barrier to entry for new production sites and facilitating geographic diversification of the supply base. This environmental compatibility not only mitigates regulatory risk but also enhances the brand reputation of suppliers who can market their products as sustainably manufactured, a key differentiator in today's eco-conscious marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrene helicene synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their existing portfolios. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this photochemical synthesis method over traditional metal-catalyzed routes?
A: The photochemical method eliminates the need for expensive and toxic transition metal catalysts, significantly reducing environmental impact and purification costs while utilizing common commercial reagents.
Q: What specific reaction conditions optimize the yield of the Wittig intermediate?
A: Optimal conditions involve a molar ratio of 2:2:1 for benzyltriphenylphosphonium chloride, n-butyllithium, and the aldehyde precursor in THF solvent at 25°C for 16 hours.
Q: Is this synthesis route scalable for industrial production of OLED materials?
A: Yes, the process utilizes stable raw materials that do not require special storage conditions and employs standard unit operations like extraction and column chromatography, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrene[4]helicene Supplier
As the demand for high-performance organic semiconductors continues to accelerate, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic capabilities tailored for the electronic materials sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging deep expertise in photochemistry and organometallic transformations to deliver consistent results. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and optical properties of every batch before shipment. This commitment to quality assurance guarantees that our clients receive materials that perform reliably in their final applications, whether in OLED displays, chemical sensors, or biological imaging probes.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be adapted to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the economic benefits of switching to this metal-free process for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of integrating these advanced pyrene helicene derivatives into your product development pipeline.
