Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic routes. These triazole scaffolds are ubiquitous in medicinal chemistry, serving as key structural motifs in numerous bioactive molecules ranging from antifungal agents to kinase inhibitors. The introduction of a trifluoromethyl group at the 5-position is particularly strategic, as it modulates the electronic properties and lipophilicity of the parent molecule, often leading to improved pharmacokinetic profiles. This technical insight report analyzes the novel iodine-promoted cyclization strategy, highlighting its potential for cost reduction in pharmaceutical intermediate manufacturing and its suitability for commercial scale-up of complex heterocycles.
The significance of this technology extends beyond academic interest; it offers a practical solution for reliable pharmaceutical intermediate suppliers aiming to streamline their production pipelines. By utilizing inexpensive and readily available starting materials such as hydrazones and trifluoroethylimidoyl chlorides, the process circumvents the need for hazardous or costly trifluoromethylating reagents. Furthermore, the operational simplicity—eliminating the need for rigorous exclusion of air and moisture—positions this method as a highly attractive candidate for industrial adoption. As we delve deeper into the mechanistic nuances and commercial implications, it becomes evident that this patent represents a substantial leap forward in the efficient synthesis of high-value fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has been fraught with challenges that hinder large-scale production. Traditional literature reports primarily describe two main approaches, both of which suffer from significant drawbacks in a manufacturing context. The first approach involves the direct trifluoromethylation of pre-synthesized 1,2,4-triazole rings. This strategy typically relies on specialized trifluoromethylating reagents, which are often expensive, unstable, and difficult to handle on a multi-kilogram scale. The second mainstream method utilizes synthons bearing a trifluoromethyl group, such as trifluorodiazoethane, to react with coupling substrates. While effective in small-scale laboratory settings, trifluorodiazoethane is inherently unstable and potentially explosive, posing severe safety risks for supply chain heads concerned with plant safety and regulatory compliance. Additionally, many existing protocols require the use of transition metal catalysts, which introduce the risk of heavy metal contamination in the final high-purity pharmaceutical intermediates, necessitating complex and costly purification steps to meet stringent regulatory limits.
The Novel Approach
In stark contrast to these conventional limitations, the methodology outlined in CN110467579B offers a streamlined and safer alternative. This novel approach employs a condensation-cyclization strategy using trifluoroethylimidoyl chloride and hydrazones as the primary building blocks. These starting materials are not only commercially available but can also be synthesized easily from cheap aldehydes and aromatic amines, ensuring a stable and cost-effective supply chain. Crucially, the reaction is promoted by elemental iodine rather than expensive transition metals, effectively eliminating the need for heavy metal scavengers during workup. The process operates under mild thermal conditions (80°C to 100°C) in common organic solvents like dichloroethane, without the stringent requirement for anhydrous or anaerobic environments. This operational robustness significantly lowers the barrier to entry for manufacturing, allowing for easier technology transfer and reducing lead time for high-purity heterocyclic compounds from development to commercial production.
Mechanistic Insights into Iodine-Promoted Cyclization
Understanding the reaction mechanism is vital for R&D directors tasked with optimizing process parameters and controlling impurity profiles. The transformation proceeds through a sophisticated yet efficient cascade of events initiated by base promotion. Initially, sodium acetate facilitates an intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and the hydrazone substrate. This condensation step generates a trifluoroacetamidine intermediate, which subsequently undergoes isomerization to align the reactive centers for cyclization. The addition of elemental iodine then triggers a base-promoted oxidative iodination, creating a reactive iodo-intermediate. This species is pivotal, as it activates the system for the subsequent intramolecular electrophilic substitution. The final step involves aromatization, where the loss of hydrogen iodide restores aromaticity to the triazole ring, yielding the stable 5-trifluoromethyl-1,2,4-triazole product. This mechanistic pathway avoids the formation of unstable diazo species, thereby enhancing the overall safety profile of the reaction.
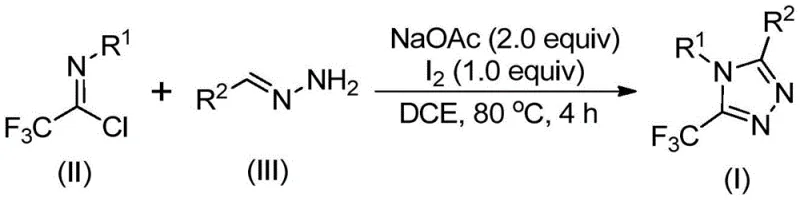
From an impurity control perspective, the use of iodine as a promoter offers distinct advantages over radical-based trifluoromethylation methods. Radical processes often lead to non-selective functionalization and complex byproduct mixtures that are difficult to separate. In this iodine-mediated ionic pathway, the reaction exhibits high regioselectivity, predominantly forming the desired 5-substituted isomer. The patent data indicates that various substituents on the aryl rings of both the imidoyl chloride and the hydrazone are well-tolerated, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like bromine and nitro. This broad functional group tolerance suggests that the electronic nature of the substrates does not drastically alter the reaction pathway, providing R&D teams with a versatile platform for generating diverse libraries of triazole derivatives without needing to re-optimize conditions for each new analog.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazoles Efficiently
The practical execution of this synthesis is designed for simplicity and reproducibility, making it ideal for both laboratory discovery and pilot plant operations. The protocol involves a straightforward one-pot procedure where the reactants are mixed in a solvent such as dichloroethane, heated to facilitate the initial condensation, and then treated with iodine to drive the cyclization to completion. The molar ratios are optimized to ensure high conversion, typically employing a slight excess of the hydrazone relative to the imidoyl chloride to drive the equilibrium forward. Post-reaction workup is equally uncomplicated, involving filtration and standard silica gel chromatography if high purity is required, though crystallization may be feasible for certain derivatives. For a detailed breakdown of the specific stoichiometric ratios, temperature profiles, and isolation techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane.
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to allow initial condensation and isomerization.
- Add elemental iodine to the system and continue heating for 1 to 2 hours to promote oxidative cyclization and aromatization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the economic implications of this synthetic route are profound. The shift away from precious metal catalysis and hazardous reagents translates directly into tangible operational efficiencies. By removing the dependency on palladium, copper, or rhodium catalysts, manufacturers can avoid the volatile pricing associated with these commodities and eliminate the costly downstream processing steps required to reduce metal residues to ppm levels. Furthermore, the ability to run the reaction without strict inert atmosphere protection (nitrogen or argon blanketing) reduces the complexity of the reactor setup and lowers utility consumption. This simplification of the process infrastructure allows for faster batch turnover and reduced capital expenditure on specialized equipment, contributing to substantial cost savings in API intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts is a primary driver for cost optimization. In traditional cross-coupling or C-H activation strategies, the cost of the catalyst and the ligands can be prohibitive, especially when scaling to tonnage quantities. Additionally, the removal of heavy metals often requires specialized scavenger resins or multiple recrystallizations, which decrease overall yield and increase waste disposal costs. By utilizing elemental iodine and sodium acetate, both of which are commodity chemicals with stable pricing, this method ensures a predictable and low cost of goods sold (COGS). The use of cheap aldehydes and amines as precursors for the hydrazone and imidoyl chloride further anchors the raw material costs at a minimum, ensuring long-term economic viability for high-volume production.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability of starting materials. The precursors for this reaction—aromatic amines, aldehydes, and hydrazine hydrate—are produced on a massive global scale for various industries, ensuring a robust and diversified supply base. Unlike specialized trifluoromethylating reagents that may have single-source suppliers or long lead times, the inputs for this process are readily accessible from multiple vendors. This redundancy mitigates the risk of supply disruptions and allows procurement teams to negotiate better terms. Moreover, the stability of the intermediates means they can be stockpiled if necessary, providing a buffer against market fluctuations and ensuring continuous production schedules for critical pharmaceutical programs.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden challenges related to heat transfer and safety, but this methodology is inherently scalable. The reaction exotherms are manageable, and the absence of explosive diazo compounds removes a major safety hurdle for plant engineers. From an environmental standpoint, the process generates less hazardous waste compared to methods using toxic heavy metals or strong oxidants. The solvent systems employed, such as dichloroethane, are well-understood and can be efficiently recovered and recycled in modern facilities. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing site, a key metric for modern ESG-compliant supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl-triazole synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical decision-makers evaluating this technology for their pipeline.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in patent CN110467579B utilizes elemental iodine as a promoter instead of expensive heavy metal catalysts like palladium or copper, significantly simplifying purification and reducing costs.
Q: What are the reaction conditions regarding moisture and oxygen?
A: Unlike many traditional heterocycle syntheses, this process does not require strict anhydrous or anaerobic conditions, making it much easier to operate and scale up in standard manufacturing facilities.
Q: Is the substrate scope limited for this triazole synthesis?
A: The method demonstrates broad substrate tolerance, accommodating various substituted aryl groups, alkenyl groups, and heteroaryl groups on both the imidoyl chloride and hydrazone components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and commercialization. Our team of expert chemists has extensively evaluated the technology disclosed in CN110467579B and is fully prepared to leverage this iodine-promoted cyclization for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from gram-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate meets the highest quality standards.
We invite you to collaborate with us to unlock the full potential of this cost-effective synthesis. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to contact our technical procurement team today to request specific COA data for our existing triazole library or to discuss route feasibility assessments for your novel analogs. Let us help you optimize your supply chain and reduce your time-to-market with our reliable and scalable manufacturing solutions.
