Advanced Palladium-Catalyzed Synthesis of Pyrazino-fused Quinazolinones for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of Pyrazino-fused Quinazolinones for Scalable Pharmaceutical Manufacturing
The landscape of heterocyclic chemistry is constantly evolving, driven by the need for more sustainable and efficient synthetic routes for complex pharmaceutical intermediates. Patent CN112209935B introduces a groundbreaking methodology for the preparation of pyrazino-fused quinazolinone substances, a class of compounds with significant potential in drug discovery and material science. This innovation addresses critical bottlenecks in traditional synthesis by utilizing a palladium-catalyzed cyclization strategy that operates effectively in aqueous media. For R&D directors and process chemists, this represents a paradigm shift away from hazardous organic solvents towards greener, cost-effective manufacturing protocols. The technology enables the direct construction of the tricyclic core from readily available 2-(2-benzoyl-4-oxoquinazolin-3(4H)-yl)acetonitrile derivatives and arylboronic acids, achieving yields as high as 94% under mild conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused quinazolinone scaffolds has been plagued by significant operational and safety challenges that hinder large-scale production. Prior art methods often rely on toxic reagents such as phenylhydrazine or require the use of hazardous solvents like 1,4-dioxane, which pose severe health risks to operators and complicate waste disposal protocols. Furthermore, conventional routes frequently necessitate multi-step sequences involving the pre-synthesis of specific skeletons, leading to accumulated yield losses and increased processing time. The reliance on expensive transition metal catalysts in non-aqueous environments also demands rigorous purification steps to remove residual heavy metals, a critical requirement for pharmaceutical grade intermediates that drastically inflates production costs. These legacy processes lack the atom economy and environmental compatibility required by modern green chemistry standards, making them increasingly obsolete for industrial applications.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a streamlined one-pot reaction that directly couples acetonitrile derivatives with arylboronic acids to form the target pyrazino-fused structure. This approach eliminates the need for intermediate isolation, thereby reducing unit operations and minimizing material loss during transfer. A key differentiator is the use of water as the preferred solvent, which not only enhances the economic profile by replacing expensive organic solvents but also facilitates easier product separation and catalyst recovery. The reaction proceeds smoothly under normal pressure and moderate temperatures ranging from 70 to 90°C, ensuring energy efficiency and operational safety. By employing a palladium catalyst system promoted by methanesulfonic acid, the process achieves exceptional conversion rates and substrate tolerance, allowing for the rapid generation of diverse chemical libraries.
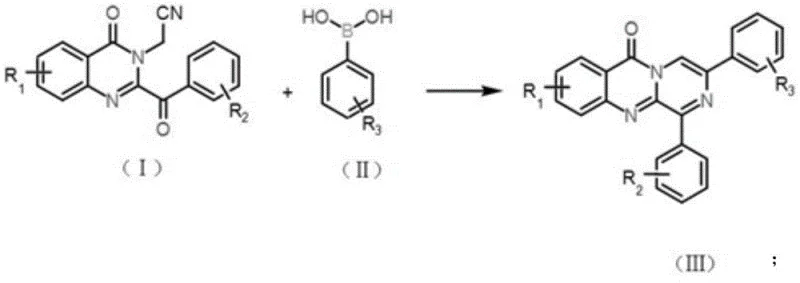
The versatility of this synthetic route is further demonstrated by its ability to accommodate a wide range of substituents on both the quinazolinone and the boronic acid components. As illustrated in the specific reaction examples, electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens and trifluoromethyl, are all tolerated without significant degradation in yield. This broad substrate scope is invaluable for medicinal chemists seeking to optimize the pharmacokinetic properties of lead compounds through structural modification. The robustness of the reaction conditions, which can even be performed in an air atmosphere rather than requiring strict inert gas protection, further underscores its suitability for commercial scale-up. This combination of simplicity, safety, and efficiency positions the technology as a superior alternative for the reliable supply of high-purity pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Cyclization
The underlying chemical mechanism driving this transformation involves a sophisticated interplay between the palladium catalyst and the organic substrates, facilitated by the acidic promoter. The cycle initiates with the transmetallation of the arylboronic acid with the palladium(II) species, generating an active aryl-palladium intermediate. This electrophilic metal center then coordinates with the nitrile group of the acetonitrile derivative, activating it for subsequent intramolecular nucleophilic attack. The coordination geometry allows for the precise alignment of reactive centers, promoting the formation of the new carbon-nitrogen bonds required to close the pyrazine ring. This step is critical for ensuring regioselectivity and preventing the formation of unwanted byproducts that could complicate downstream purification efforts.
Following the cyclization event, protonation of the resulting imine-palladium complex by methanesulfonic acid releases the final heterocyclic product and regenerates the active palladium catalyst for the next turnover. This regeneration step is vital for maintaining catalytic efficiency and minimizing the total loading of expensive metal required for the process. The use of an acidic promoter also helps to suppress side reactions that might occur under neutral or basic conditions, thereby enhancing the overall purity of the crude reaction mixture. Understanding these mechanistic nuances allows process engineers to fine-tune reaction parameters such as pH and temperature to maximize throughput. The ability to control impurity profiles at the molecular level is a key advantage for manufacturers aiming to meet stringent regulatory specifications for active pharmaceutical ingredients.
How to Synthesize Pyrazino-fused Quinazolinone Efficiently
Implementing this synthesis on a pilot or production scale requires careful attention to reagent quality and mixing dynamics to ensure consistent results. The protocol is designed to be operationally simple, involving the sequential addition of the acetonitrile derivative, arylboronic acid, palladium catalyst, solvent, and acid promoter into a standard reaction vessel. Once the components are mixed, the system is heated to the optimal temperature range, where the reaction progresses over a period of 12 to 36 hours depending on the specific substrate reactivity. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the technical guide below to assist your team in replicating these high-yield results.
- Charge the reactor with 2-(2-benzoyl-4-oxoquinazolin-3(4H)-yl)acetonitrile derivative, arylboronic acid, palladium catalyst, solvent, and acid promoter under air atmosphere.
- Stir the mixture thoroughly to ensure uniform dispersion of reactants, particularly when using water as the primary solvent medium.
- Heat the reaction system to 70-90°C and maintain stirring for 12 to 36 hours to complete the cyclization and obtain the target pyrazino-fused quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative benefits that directly impact the bottom line and operational resilience. The shift to an aqueous-based process eliminates the dependency on volatile and flammable organic solvents, significantly reducing storage hazards and insurance costs associated with chemical inventory. Moreover, the high atom economy and single-step nature of the reaction minimize raw material consumption, leading to substantial cost savings in the manufacturing of complex heterocyclic intermediates. For supply chain managers, the ability to source readily available starting materials like arylboronic acids ensures a stable and continuous supply flow, mitigating the risks associated with specialized or scarce reagents. The simplified workflow also translates to shorter production cycles, enabling faster response times to market demands.
- Cost Reduction in Manufacturing: The elimination of toxic reagents and the use of water as a primary solvent drastically reduce waste disposal costs and solvent recovery expenses. By avoiding the need for extensive purification steps to remove heavy metal residues, the overall processing time and labor costs are significantly lowered. This streamlined approach allows for a more competitive pricing structure for the final pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which do not require inert atmospheres or high-pressure equipment, simplifies the infrastructure requirements for production facilities. This ease of implementation means that multiple manufacturing sites can potentially adopt the process, diversifying the supply base and reducing the risk of single-source bottlenecks. The availability of diverse arylboronic acids on the global market further secures the raw material supply chain against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild thermal profile and lack of exothermic hazards, making it suitable for expansion from kilogram to multi-ton scales. Operating in an aqueous medium aligns with increasingly strict environmental regulations regarding volatile organic compound emissions, ensuring long-term compliance and sustainability. This green chemistry profile enhances the corporate reputation of manufacturers and facilitates smoother regulatory approvals for new drug applications containing these intermediates.
![Specific reaction example showing the synthesis of 1,3-diphenyl-6H-pyrazino[2,1-b]quinazoline](/insights/img/pyrazino-quinazolinone-synthesis-palladium-pharma-supplier-20260305132932-04.webp)
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Whether you are evaluating the technical feasibility for a specific derivative or assessing the economic impact on your supply chain, these answers offer the necessary clarity to move forward with confidence in adopting this advanced manufacturing technology.
Q: What are the primary advantages of using water as a solvent in this synthesis?
A: Using water significantly reduces organic solvent waste, lowers material costs, and improves operator safety by eliminating volatile organic compounds, while maintaining high reaction yields up to 94%.
Q: Does this method require inert gas protection like nitrogen or argon?
A: No, the process is robust enough to be conducted under an air atmosphere, which simplifies operational requirements and reduces equipment costs associated with inert gas purging.
Q: What is the substrate scope for the arylboronic acid component?
A: The method demonstrates strong universality, accommodating various substituents including methyl, methoxy, halogens, trifluoromethyl, and naphthyl groups on the arylboronic acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazino-fused Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthetic routes in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced technologies like the aqueous palladium-catalyzed cyclization described here, we can offer you a competitive edge in terms of both cost and speed to market.
We invite you to collaborate with us to explore the full potential of this synthesis for your specific drug candidates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for pyrazino-fused quinazolinone derivatives.
