Advanced Synthesis of 2,7-Dibromo-9,10-Substituted-Phenanthrene Derivatives for High-Performance OLED Manufacturing
Introduction to Next-Generation Blue OLED Intermediates
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only exhibit superior electrochemical performance but also possess the thermal stability and solubility required for scalable manufacturing processes. Patent CN103254044A introduces a groundbreaking series of 2,7-dibromo-9,10-substituted-phenanthrene derivatives that address critical limitations found in earlier generations of blue-emitting host materials. Unlike traditional phenanthrene compounds that suffer from poor solubility and inefficient blue emission due to molecular aggregation, this novel chemical architecture strategically places substituent groups at the 2, 7, 9, and 10 positions. This specific substitution pattern effectively mitigates intermolecular stacking forces, thereby enhancing the material's thermal robustness and spectral stability. For R&D directors and procurement specialists seeking a reliable OLED material supplier, understanding the synthetic pathway detailed in this patent is crucial, as it offers a viable route to high-purity intermediates essential for next-generation display technologies.
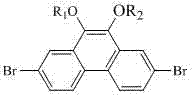
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the field of blue OLED materials, such as the compounds described in WO04/016575 and JP2001/332384, has historically struggled with achieving the perfect balance between emission efficiency and processability. Many existing phenanthrene derivatives are substituted primarily at the 3,6-positions while lacking functionalization at the 9,10-positions, which often results in undesirable shallow blue luminescence and insufficient thermal stability for commercial device lifetimes. Furthermore, previous attempts to introduce bulky groups, such as the fused bicyclic aliphatic systems mentioned in JP2003/055276, necessitated overly complex synthetic routes that are difficult to scale industrially. Another significant drawback identified in earlier patents, specifically the inventor's own prior work (CN201210287739.6), was the presence of active hydroxyl hydrogen atoms which severely limited further functionalization at the 2,7-positions and resulted in poor solubility, leading to excessive solvent consumption during purification.
The Novel Approach
The synthetic strategy outlined in CN103254044A represents a paradigm shift by focusing on the simultaneous functionalization of the 2,7 and 9,10 positions to create a sterically hindered yet chemically versatile scaffold. By converting the reactive hydroxyl groups at the 9,10-positions into alkoxy groups (such as ethoxy or propoxy), the new method eliminates active hydrogen, thereby preventing unwanted side reactions and significantly boosting solubility in common organic solvents. This approach not only simplifies the purification process through standard recrystallization techniques but also creates a superior chemical environment for subsequent coupling reactions at the 2,7-bromo sites. The overall synthetic route is depicted below, showcasing a streamlined progression from commercially available starting materials to the high-value target intermediates.
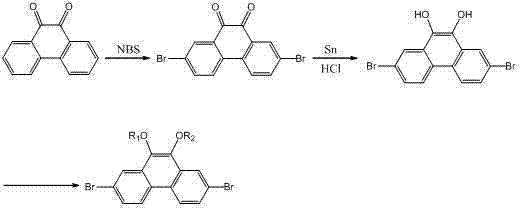
Mechanistic Insights into the Multi-Step Synthesis
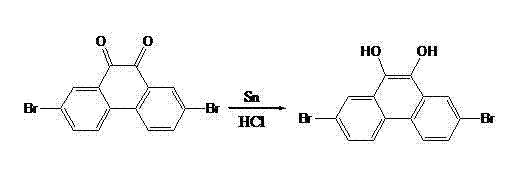
The synthesis begins with a highly regioselective electrophilic aromatic substitution where 9,10-phenanthrenequinone is treated with N-bromosuccinimide (NBS) in concentrated sulfuric acid. This specific reaction condition is critical as the strong acidic medium activates the quinone ring system, directing the bromine atoms exclusively to the 2 and 7 positions due to the electron-withdrawing nature of the carbonyl groups at 9 and 10. Following isolation, the resulting 2,7-dibromophenanthrenequinone undergoes a reduction step using tin (Sn) and concentrated hydrochloric acid in glacial acetic acid under reflux. This reduction converts the carbonyl oxygens into hydroxyl groups, yielding 2,7-dibromo-9,10-dihydroxyphenanthrene with high purity (>98%), a critical parameter for ensuring consistent device performance in downstream applications.
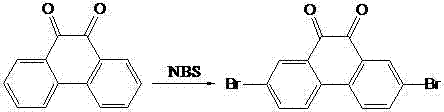
The final and perhaps most commercially significant step involves an acid-catalyzed etherification where the dihydroxy intermediate reacts with short-chain alcohols (C1-C3) in the presence of a strong acid catalyst, preferably trifluoromethanesulfonic acid. Unlike traditional methods that might require harsh conditions or stoichiometric amounts of base, this catalytic approach proceeds efficiently at moderate temperatures (60-120°C), driving the equilibrium towards the ether product while minimizing degradation. The use of trifluoromethanesulfonic acid is particularly advantageous as it acts as a potent proton donor to activate the alcohol nucleophile without introducing metal contaminants that could quench OLED emission. This mechanistic elegance ensures that the final 2,7-dibromo-9,10-diethoxyphenanthrene or dipropoxyphenanthrene products are obtained with excellent yields and minimal impurity profiles.
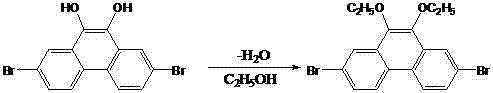
How to Synthesize 2,7-Dibromo-9,10-Substituted-Phenanthrene Efficiently
Executing this synthesis requires precise control over reaction parameters, particularly temperature and acid concentration, to maximize yield and purity. The process is designed to be robust, utilizing standard laboratory glassware and readily available reagents, making it highly suitable for translation to pilot and commercial scales. Operators should pay close attention to the workup procedures, specifically the recrystallization steps using solvents like DMSO or methanol, which are essential for removing trace metal residues and unreacted starting materials. For a detailed breakdown of the specific operational parameters, stoichiometry, and safety protocols required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide provided below.
- Brominate 9,10-phenanthrenequinone using NBS in concentrated sulfuric acid to obtain 2,7-dibromophenanthrenequinone.
- Reduce the quinone intermediate using Tin (Sn) and concentrated hydrochloric acid in glacial acetic acid to form 2,7-dibromo-9,10-dihydroxyphenanthrene.
- Perform acid-catalyzed etherification with C1-C3 alcohols (e.g., ethanol or propanol) using trifluoromethanesulfonic acid to yield the final substituted derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits, primarily driven by the accessibility of raw materials and the simplicity of the unit operations involved. The starting material, 9,10-phenanthrenequinone, is a commodity chemical with a stable global supply, reducing the risk of bottlenecks that often plague specialty chemical manufacturing. Furthermore, the elimination of complex transition metal catalysts in favor of organic acids and tin-based reagents significantly lowers the cost of goods sold (COGS) by removing the need for expensive ligand systems and rigorous heavy metal scavenging steps typically required in palladium-catalyzed cross-couplings.
- Cost Reduction in Manufacturing: The process achieves cost optimization through the use of catalytic amounts of trifluoromethanesulfonic acid rather than stoichiometric reagents, which drastically reduces reagent costs per kilogram of product. Additionally, the high crude purity achieved before recrystallization minimizes solvent usage and waste generation, leading to significant operational expenditure savings in waste treatment and solvent recovery systems.
- Enhanced Supply Chain Reliability: By relying on a linear three-step synthesis with high-yielding transformations, the production lead time is inherently shorter compared to multi-step convergent syntheses often seen in complex OLED material manufacturing. The robustness of the reaction conditions ensures consistent batch-to-batch quality, allowing supply chain managers to forecast inventory needs with greater confidence and reduce safety stock levels.
- Scalability and Environmental Compliance: The synthetic pathway avoids the use of highly toxic or volatile organic compounds that pose severe environmental hazards, aligning well with modern green chemistry principles and regulatory compliance standards. The scalability is further supported by the fact that the exothermic profiles of the bromination and reduction steps are manageable in large-scale reactors, facilitating a smooth transition from gram-scale R&D to ton-scale commercial production without extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and handling of these phenanthrene derivatives. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the material's value proposition for electronic chemical applications.
Q: What are the key advantages of 9,10-substitution in phenanthrene derivatives for OLEDs?
A: Substitution at the 9,10-positions effectively reduces intermolecular stacking interactions, which significantly improves thermal stability and enhances blue light-emitting properties compared to unsubstituted or 3,6-substituted analogs.
Q: How does this synthesis method improve solubility compared to previous art?
A: By introducing alkoxy groups at the 9,10-positions, the active hydrogen atoms are eliminated, and steric hindrance is increased, leading to markedly improved solubility in organic solvents like THF, facilitating solution processing.
Q: Is the catalyst used in the final step recoverable or expensive?
A: The process utilizes trifluoromethanesulfonic acid as a preferred catalyst, which operates efficiently in catalytic amounts, reducing the overall cost burden associated with stoichiometric reagents and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-Dibromo-9,10-Substituted-Phenanthrene Derivatives Supplier
As the demand for high-performance blue OLED materials continues to surge, partnering with a manufacturer that possesses deep technical expertise in complex organic synthesis is paramount. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure that your supply of critical intermediates remains uninterrupted. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities that could compromise device efficiency, ensuring that every batch of 2,7-dibromo-9,10-substituted-phenanthrene derivatives meets the exacting standards of the optoelectronics industry.
We invite you to engage with our technical procurement team to discuss how this patented technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain visibility into potential reductions in your manufacturing overhead. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements and quality targets.
