Scalable Electrocatalytic Production of High-Purity 3-Alkylthio Quinoxalinone Derivatives for Pharma
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex heterocyclic scaffolds, particularly those with significant biological activity. Patent CN111041515B introduces a groundbreaking methodology for the synthesis of 3-alkylthio substituted quinoxalinone derivatives utilizing advanced electrocatalysis technology. This innovation represents a paradigm shift from traditional stoichiometric oxidation methods to a greener, electricity-driven Cross Dehydrogenative Coupling (CDC) process. By leveraging direct anodic oxidation, this technique enables the direct functionalization of the C-3 position on the quinoxalinone ring without the necessity for pre-installed halogen handles or toxic heavy metal catalysts. For R&D directors and procurement strategists, this patent offers a compelling value proposition: a streamlined synthetic route that significantly reduces raw material complexity while enhancing the environmental profile of the manufacturing process. The ability to generate high-value sulfur-containing heterocycles under mild conditions opens new avenues for the rapid development of enzyme inhibitors and anticancer agents, positioning this technology as a critical asset for modern API intermediate supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-S bonds at the C-3 position of quinoxalinone scaffolds has relied heavily on nucleophilic substitution reactions involving pre-halogenated precursors. As illustrated in the reaction scheme below, traditional protocols typically require the synthesis of 3-chloro or 3-bromo quinoxalinone derivatives as starting materials, which are then subjected to reflux conditions with thiols in the presence of strong bases like sodium ethoxide. 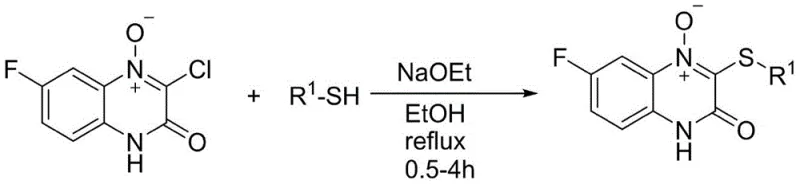 This classical approach suffers from inherent inefficiencies, primarily due to poor atom economy; the halogen atom installed solely for the purpose of leaving group capability constitutes wasted mass that must be disposed of as salt waste. Furthermore, these reactions often necessitate harsh thermal conditions and stoichiometric amounts of base, which can lead to side reactions, decomposition of sensitive functional groups, and difficult purification profiles. From a supply chain perspective, the reliance on halogenated intermediates adds an extra synthetic step upstream, increasing the overall lead time and the cumulative cost of goods sold (COGS). Additionally, the potential for residual halogenated impurities poses a significant regulatory burden for pharmaceutical applications, requiring rigorous and costly purification steps to meet stringent safety specifications.
This classical approach suffers from inherent inefficiencies, primarily due to poor atom economy; the halogen atom installed solely for the purpose of leaving group capability constitutes wasted mass that must be disposed of as salt waste. Furthermore, these reactions often necessitate harsh thermal conditions and stoichiometric amounts of base, which can lead to side reactions, decomposition of sensitive functional groups, and difficult purification profiles. From a supply chain perspective, the reliance on halogenated intermediates adds an extra synthetic step upstream, increasing the overall lead time and the cumulative cost of goods sold (COGS). Additionally, the potential for residual halogenated impurities poses a significant regulatory burden for pharmaceutical applications, requiring rigorous and costly purification steps to meet stringent safety specifications.
The Novel Approach
In stark contrast, the novel electrocatalytic method disclosed in the patent utilizes a direct Cross Dehydrogenative Coupling (CDC) strategy that bypasses the need for pre-functionalization entirely. Instead of relying on chemical oxidants or metal catalysts to drive the reaction, this method employs electricity as the primary oxidant to activate the C-H bond at the C-3 position of the quinoxalin-2-one core. 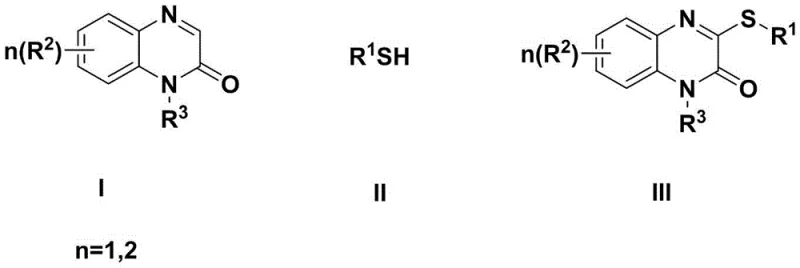 This approach allows for the direct coupling of readily available quinoxalin-2-one compounds with various thiol reagents under remarkably mild conditions, typically at room temperature or slightly elevated temperatures up to 40°C. The elimination of the halogenation step not only simplifies the synthetic sequence but also drastically improves the overall atom economy of the process. By avoiding the use of transition metals such as copper or palladium, which are common in traditional cross-coupling reactions, this method mitigates the risk of heavy metal contamination in the final product. This is a critical advantage for pharmaceutical manufacturers who face increasingly strict limits on residual metals in active pharmaceutical ingredients (APIs), thereby reducing the need for expensive scavenging resins and complex downstream processing.
This approach allows for the direct coupling of readily available quinoxalin-2-one compounds with various thiol reagents under remarkably mild conditions, typically at room temperature or slightly elevated temperatures up to 40°C. The elimination of the halogenation step not only simplifies the synthetic sequence but also drastically improves the overall atom economy of the process. By avoiding the use of transition metals such as copper or palladium, which are common in traditional cross-coupling reactions, this method mitigates the risk of heavy metal contamination in the final product. This is a critical advantage for pharmaceutical manufacturers who face increasingly strict limits on residual metals in active pharmaceutical ingredients (APIs), thereby reducing the need for expensive scavenging resins and complex downstream processing.
Mechanistic Insights into Electrochemical C-H Thiolation
The core of this technological advancement lies in the electrochemical generation of reactive radical species through anodic oxidation. In the proposed mechanism, the quinoxalin-2-one substrate undergoes single-electron oxidation at the anode surface to form a radical cation intermediate. This highly reactive species facilitates the deprotonation at the C-3 position, generating a carbon-centered radical that is poised for nucleophilic attack. Simultaneously, the thiol component may also undergo electrochemical activation or participate directly as a nucleophile depending on the specific electrolyte and additive conditions employed. The presence of acidic additives, such as trifluoroacetic acid or acetic acid, plays a crucial role in modulating the oxidation potential of the substrates and stabilizing the intermediate species, ensuring high selectivity for the desired C-S bond formation. This precise control over the reaction pathway via electrical parameters (current and voltage) allows chemists to fine-tune the process for different substrate classes, minimizing the formation of over-oxidized byproducts or disulfide dimers that often plague chemical oxidation methods.
From an impurity control perspective, the metal-free nature of this electrocatalytic cycle offers a distinct advantage in managing the impurity profile of the final product. Traditional metal-catalyzed reactions often generate complex mixtures of metal-ligand complexes and homocoupling byproducts that are structurally similar to the target molecule and difficult to separate. In the electrochemical protocol, the primary byproducts are typically hydrogen gas evolved at the cathode and simple salts derived from the supporting electrolyte, both of which are easily removed during the aqueous workup phase. The use of constant current electrolysis ensures a steady rate of electron transfer, preventing local hotspots of high oxidation potential that could lead to substrate degradation. This results in a cleaner crude reaction mixture, which translates directly to higher isolated yields and reduced solvent consumption during the purification stage, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
How to Synthesize 3-Alkylthio Quinoxalinone Derivatives Efficiently
The practical implementation of this electrocatalytic synthesis is designed to be straightforward and adaptable to existing laboratory and pilot-scale infrastructure. The general procedure involves dissolving the quinoxalin-2-one starting material and the thiol reagent in a polar aprotic solvent such as DMF, DMSO, or acetonitrile, along with a supporting electrolyte like tetrabutylammonium bromide. An acid additive is introduced to facilitate the reaction kinetics, and the mixture is subjected to constant current electrolysis (typically 10-15 mA) using undivided cells with inert electrodes. The reaction progress is conveniently monitored by thin-layer chromatography (TLC), and upon completion, the product is isolated through standard extraction and column chromatography techniques. For a detailed breakdown of the specific operational parameters, stoichiometry, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining quinoxalin-2-one compound (Formula I), thiol compound (Formula II), and an acid additive in an organic solvent containing a supporting electrolyte.
- Apply a constant current of 10-15 mA to the reaction mixture while stirring at a controlled temperature between 20°C and 40°C for 5 to 20 hours.
- Upon completion monitored by TLC, perform aqueous workup with saturated brine, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrocatalytic technology presents a multitude of strategic benefits that extend beyond mere technical feasibility. The most significant impact is observed in the reduction of manufacturing costs driven by the simplification of the raw material portfolio. By eliminating the requirement for expensive pre-halogenated intermediates and precious metal catalysts, the direct cost of materials is substantially lowered. Furthermore, the operational expenditure is optimized through the use of electricity as a traceless reagent, removing the need for purchasing, storing, and disposing of hazardous chemical oxidants. This shift not only reduces the direct financial outlay but also minimizes the logistical complexities associated with handling dangerous goods, thereby enhancing overall site safety and regulatory compliance.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior to traditional methods due to the drastic simplification of the synthetic route. By removing the halogenation step and the subsequent metal-catalyzed coupling, the number of unit operations is reduced, leading to lower labor costs and decreased equipment occupancy time. The absence of noble metals like palladium or copper eliminates the substantial cost associated with catalyst recovery and the specialized resin treatments required to meet residual metal specifications. Additionally, the mild reaction conditions reduce energy consumption related to heating and cooling, contributing to a leaner and more cost-efficient production profile that enhances margin potential for high-volume commercial campaigns.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity-grade starting materials that are widely available from multiple global suppliers. Unlike specialized halogenated building blocks which may have limited sources and long lead times, quinoxalin-2-ones and simple thiols are bulk chemicals with robust supply networks. This diversification of sourcing options mitigates the risk of supply disruptions and price volatility. Moreover, the scalability of electrochemical processes allows for flexible production scheduling; capacity can be increased simply by adding more electrode pairs or running multiple cells in parallel, ensuring that demand surges can be met without the need for massive capital investment in new reactor vessels or high-pressure infrastructure.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent, the ability to demonstrate a green manufacturing process is a key competitive differentiator. This electrocatalytic method generates minimal waste, primarily consisting of benign salts and hydrogen gas, avoiding the generation of heavy metal sludge or halogenated organic waste streams. This simplifies wastewater treatment and reduces the environmental footprint of the facility. The process is inherently safer as it operates at ambient pressure and low temperatures, reducing the risk of thermal runaways. These factors collectively streamline the regulatory approval process for new drug filings and ensure long-term operational continuity in jurisdictions with strict environmental enforcement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of this electrochemical method over traditional halogenation routes?
A: The electrochemical method eliminates the need for pre-functionalized halogenated starting materials and expensive transition metal catalysts. This results in superior atom economy, reduced heavy metal contamination risks, and simplified downstream purification processes compared to conventional dehalogenation coupling methods.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process operates under mild conditions (room temperature to 40°C) and uses electricity as a clean reagent. The absence of hazardous oxidants and the use of common organic solvents make it highly adaptable for scale-up in standard electrochemical flow reactors or batch cells.
Q: What levels of purity can be achieved with this electrocatalytic protocol?
A: Experimental data from the patent indicates that the method consistently yields products with high HPLC purity, often exceeding 96% to 98% after standard column chromatography, making it suitable for the production of high-specification pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkylthio Quinoxalinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic technologies in the synthesis of complex pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111041515B can be successfully translated into robust industrial processes. We are committed to delivering high-purity 3-alkylthio quinoxalinone derivatives that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our state-of-the-art electrochemical facilities allow us to offer this green synthesis route as a viable alternative to traditional methods, providing our clients with a sustainable and cost-effective supply solution.
We invite pharmaceutical companies and agrochemical manufacturers to collaborate with us to evaluate the feasibility of this route for their specific pipeline candidates. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free protocol for your specific target molecules. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments tailored to your project requirements.
