Advanced Photoredox Synthesis of 1-Trifluoroethyl Isoquinoline for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to access fluorinated heterocyclic scaffolds, particularly isoquinoline derivatives, which serve as critical building blocks for a wide array of bioactive molecules. Patent CN108484500B introduces a groundbreaking preparation method for 1-trifluoroethyl isoquinoline that leverages visible-light photoredox catalysis to overcome the limitations of classical synthetic approaches. This innovation utilizes alkenyl isocyanide and trifluoroiodoethane compounds as key starting materials, mediated by an iridium salt photosensitizer and inorganic bases under mild thermal conditions. The strategic incorporation of the trifluoroethyl group, rather than a simple trifluoromethyl group, offers unique physicochemical properties that are highly desirable in modern drug design, acting as a lipophilic hydrogen bond donor mimic. By shifting away from harsh acidic conditions or expensive noble metal catalysts typically required for such transformations, this technology provides a robust platform for the reliable pharmaceutical intermediate supplier market, ensuring high purity and consistent quality for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoquinoline derivatives has relied heavily on classical methodologies such as the Bischler-Napieralski reaction or variations of the Pomeranz-Fritsch synthesis, which often necessitate the use of aggressive reagents and extreme reaction parameters. For instance, previous attempts to introduce trifluoroethyl groups involved dehydroxylation reactions using strong acids like trifluoromethanesulfonic acid in benzene solvents at elevated temperatures, posing significant safety risks and environmental concerns due to the volatility and toxicity of the solvents employed. Furthermore, alternative transition-metal catalyzed approaches utilizing rhodium or copper bimetallic systems, while effective, frequently demand high temperatures and stoichiometric amounts of expensive oxidants, leading to substantial cost burdens and complex waste streams that complicate purification processes. These conventional routes often suffer from poor functional group tolerance, limiting the structural diversity accessible to medicinal chemists and creating bottlenecks in the development of novel therapeutic candidates that require specific substitution patterns on the isoquinoline core.
The Novel Approach
In stark contrast, the methodology disclosed in CN108484500B represents a paradigm shift by employing a radical tandem reaction mechanism driven by visible light irradiation, which allows for the construction of the isoquinoline skeleton under exceptionally mild conditions. This novel approach eliminates the need for corrosive strong acids or high-energy thermal inputs, thereby enhancing the operational safety profile and reducing the energy consumption associated with the manufacturing process. The use of readily available alkenyl isocyanides and trifluoroiodoethane precursors ensures a cost-effective supply chain, while the catalytic system based on iridium salts operates at low loading levels, minimizing the residual metal content in the final product. This streamlined process not only simplifies the workup procedure through standard extraction and crystallization techniques but also significantly improves the overall atom economy, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing where efficiency and sustainability are paramount.
Mechanistic Insights into Iridium-Catalyzed Photoredox Cyclization
The core of this technological advancement lies in the sophisticated interplay between the iridium photosensitizer and the trifluoroiodoethane substrate under blue light irradiation, which initiates a single-electron transfer process to generate reactive trifluoroethyl radicals. Upon excitation by visible light, the iridium catalyst enters a highly energetic state capable of reducing the carbon-iodine bond in the trifluoroiodoethane molecule, thereby releasing the trifluoroethyl radical species that serves as the key nucleophilic component in the subsequent cascade. This radical species then adds selectively to the electron-deficient carbon of the alkenyl isocyanide moiety, triggering a rapid intramolecular cyclization that constructs the isoquinoline ring system with high regioselectivity and stereochemical control. The presence of the inorganic base plays a crucial role in neutralizing the acidic byproducts generated during the reaction cycle, ensuring that the catalytic turnover remains efficient and that the reaction proceeds to completion without stalling due to acid accumulation or catalyst deactivation.
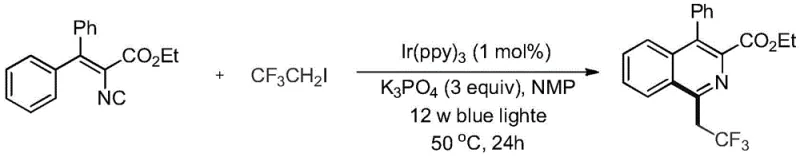
Beyond the primary bond-forming events, the mechanism inherently includes built-in safeguards against the formation of common impurities that plague traditional synthetic routes, such as over-alkylation or polymerization byproducts. The mild nature of the radical generation step prevents the degradation of sensitive functional groups that might be present on the aromatic rings of the starting materials, allowing for a broader scope of substrate compatibility compared to ionic mechanisms. Additionally, the use of polar aprotic solvents like N-methylpyrrolidone facilitates the dissolution of both the organic substrates and the inorganic base, creating a homogeneous reaction environment that promotes consistent heat and mass transfer. This precise control over the reaction microenvironment ensures that the impurity profile remains clean and predictable, which is a critical factor for R&D directors evaluating the feasibility of scaling this process for the production of high-purity pharmaceutical intermediates intended for clinical trials.
How to Synthesize 1-Trifluoroethyl Isoquinoline Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding the exclusion of oxygen and moisture, as the radical intermediates generated during the photoredox cycle are sensitive to quenching by atmospheric components. The detailed standardized synthesis steps involve the precise weighing of the iridium catalyst and inorganic base, followed by the sequential addition of the alkenyl isocyanide and trifluoroiodoethane reagents into a dry reaction vessel purged with inert gas. Maintaining the correct ratio of reagents and ensuring uniform illumination of the reaction mixture are essential for achieving the high yields reported in the patent examples, and the reaction progress should be monitored via thin-layer chromatography to determine the optimal endpoint for quenching.
- Prepare the reaction mixture by adding tris(2-phenylpyridine)iridium (1 mol%), potassium phosphate (3.0 equiv.), ethyl 2-isocyano-3,3-diphenylacrylate (1.0 equiv.), trifluoroiodoethane (1.0 equiv.), and anhydrous N-methylpyrrolidone solvent into a dry, nitrogen-blanketed reaction tube.
- Place the reaction flask in an oil bath maintained at 80°C and irradiate with 12W blue light for approximately 12 hours to drive the radical tandem cyclization to completion.
- Upon reaction completion confirmed by TLC, quench with water, extract with ethyl acetate, wash the organic phase with saturated NaCl, dry over anhydrous sodium sulfate, and purify via column chromatography to isolate the final yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this photoredox-mediated synthesis route offers transformative benefits that directly address the pain points of cost volatility and raw material scarcity often encountered in the fine chemical sector. By replacing expensive and toxic noble metal catalysts like rhodium with more economical iridium complexes used at low loadings, manufacturers can achieve substantial cost savings without compromising on reaction efficiency or product quality. The elimination of hazardous strong acids and volatile organic solvents like benzene further reduces the costs associated with waste disposal and regulatory compliance, creating a leaner and more sustainable manufacturing operation that aligns with modern environmental standards. Moreover, the simplicity of the workup procedure, which relies on standard liquid-liquid extraction and crystallization rather than complex chromatographic separations, significantly shortens the production cycle time and enhances the overall throughput of the facility.
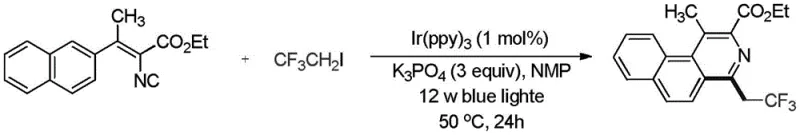
The robustness of this synthetic method also translates into enhanced supply chain reliability, as the starting materials such as alkenyl isocyanides and trifluoroiodoethane are commercially available and can be sourced from multiple vendors to mitigate supply risk. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for specialized corrosion-resistant equipment, lowering the capital expenditure required for technology transfer and scale-up. Furthermore, the high conversion rates and selectivity observed in this process minimize the formation of difficult-to-remove byproducts, ensuring a consistent supply of high-quality intermediates that meet stringent purity specifications. This reliability is crucial for supply chain heads who need to guarantee uninterrupted material flow for downstream drug substance manufacturing, reducing the lead time for high-purity pharmaceutical intermediates and enabling faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The replacement of stoichiometric oxidants and expensive rhodium catalysts with a catalytic amount of iridium and visible light leads to a drastic simplification of the reagent cost structure. By avoiding the use of corrosive strong acids, the process eliminates the need for specialized acid-resistant infrastructure and reduces the costs associated with neutralization and waste treatment. The high yield and selectivity of the reaction minimize raw material waste, ensuring that every kilogram of input contributes maximally to the final output, which drives down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially accessible starting materials ensures that production schedules are not disrupted by the scarcity of exotic reagents. The mild operating conditions reduce the risk of equipment failure or safety incidents that could halt production, providing a stable and predictable manufacturing timeline. Additionally, the simplified purification process reduces the dependency on specialized chromatography resins or columns, which can often be a bottleneck in large-scale operations, thereby smoothing out the supply chain logistics.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is highly amenable to scale-up using modern flow chemistry technologies or large-scale batch photoreactors, allowing for seamless transition from gram to ton scale. The absence of heavy metal waste and volatile toxic solvents aligns the process with green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing site. This compliance advantage is increasingly valuable in global markets where environmental regulations are becoming stricter, positioning the manufacturer as a responsible and forward-thinking partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their existing portfolios. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this photoredox method over traditional Friedel-Crafts acylation?
A: Unlike traditional Friedel-Crafts methods which often require harsh strong acids like trifluoromethanesulfonic acid and high temperatures, this photoredox protocol operates under mild conditions with visible light. This significantly improves functional group tolerance and reduces safety hazards associated with corrosive reagents.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial production due to its short process flow, easy separation of products, and the use of readily available raw materials. The mild reaction conditions also facilitate easier scale-up compared to high-pressure or high-temperature alternatives.
Q: What specific substrates are compatible with this trifluoroethylation reaction?
A: The method demonstrates broad substrate scope, accommodating various alkenyl isocyanides with different ester groups (methyl, ethyl, tert-butyl) and beta-position substituents including phenyl, naphthyl, and heterocycles. It also tolerates electron-donating and electron-withdrawing groups on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Trifluoroethyl Isoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this photoredox cyclization can be successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch, giving our clients complete confidence in the materials they receive for their drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project, whether you require custom synthesis of complex intermediates or full-scale commercial manufacturing services. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements, and ask for specific COA data and route feasibility assessments to see how we can optimize your supply chain. By partnering with us, you gain access to a wealth of chemical expertise and manufacturing capacity dedicated to delivering high-value solutions that accelerate your path to market.
