Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough in Pharmaceutical Intermediate Manufacturing
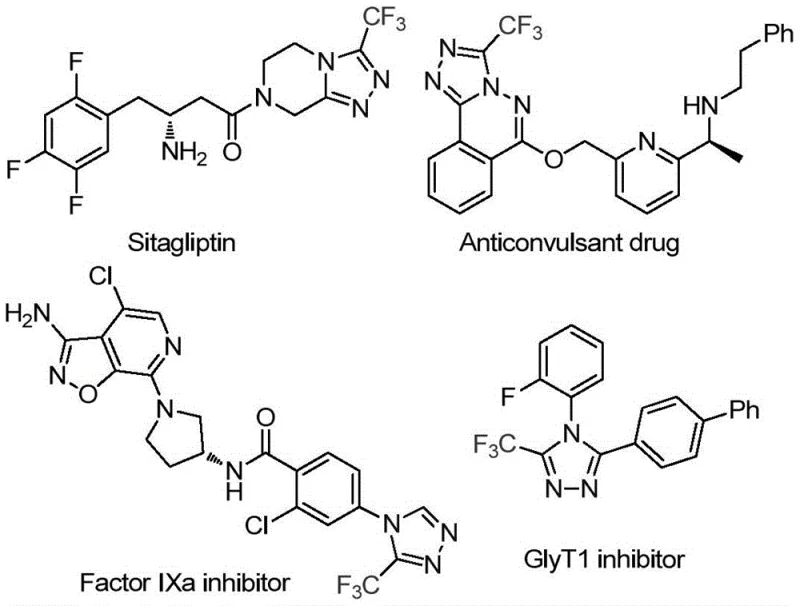
The pharmaceutical and agrochemical industries are constantly seeking more efficient routes to construct nitrogen-rich heterocyclic scaffolds, particularly those bearing fluorinated motifs which are known to enhance metabolic stability and bioavailability. Patent CN114920707B, published in April 2023, discloses a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that fundamentally shifts the paradigm of heterocyclic synthesis. This technology leverages the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as a vital carbon source, thereby streamlining the synthetic sequence. The significance of this chemical architecture cannot be overstated, as the 3-trifluoromethyl-1,2,4-triazole core is a privileged structure found in numerous high-value bioactive molecules, including prominent Factor IXa inhibitors and GlyT1 inhibitors used in treating thrombosis and neurological disorders. By providing a robust, operationally simple pathway to these critical intermediates, this innovation offers a reliable pharmaceutical intermediate supplier with a distinct competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of 1,2,4-triazole rings, especially those substituted with electron-withdrawing trifluoromethyl groups, has been fraught with synthetic challenges that hinder large-scale manufacturing. Conventional methodologies often rely on the cyclization of hydrazides with nitriles, amidines, or orthoesters, reactions that frequently demand harsh thermal conditions, strong acidic or basic media, and the use of specialized, moisture-sensitive reagents. Furthermore, many existing protocols necessitate the pre-functionalization of starting materials, leading to multi-step sequences that erode overall atom economy and generate substantial chemical waste. The requirement for strict anhydrous and anaerobic conditions in many transition-metal catalyzed approaches further complicates the engineering controls needed for production, driving up both capital expenditure and operational costs. These inefficiencies create bottlenecks in the supply chain for high-purity OLED material and pharmaceutical precursors, where consistency and cost-effectiveness are paramount.
The Novel Approach
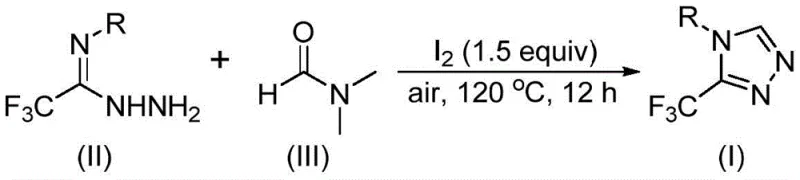
In stark contrast to these cumbersome legacy processes, the method described in CN114920707B introduces an elegant iodine-promoted tandem cyclization that utilizes DMF as a dual-purpose reagent. This approach eliminates the need for external carbon sources or complex catalysts, relying instead on the intrinsic reactivity of the solvent itself under oxidative conditions. As illustrated in the reaction scheme below, the process combines trifluoroethyliminohydrazide with molecular iodine in DMF, heating the mixture under air to effect ring closure. This strategy drastically simplifies the reaction setup, removing the necessity for inert gas lines or glovebox techniques. The ability to use DMF, a low-cost commodity chemical, as a reactant significantly reduces the raw material bill of materials, facilitating cost reduction in API manufacturing. Moreover, the broad substrate scope demonstrated in the patent allows for the introduction of diverse functional groups at the 4-position of the triazole ring, enabling medicinal chemists to rapidly access analog libraries for structure-activity relationship studies.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the versatile activation of DMF by molecular iodine, which unlocks two distinct pathways for carbon incorporation depending on the specific reaction trajectory. In the first plausible pathway, the formyl group of DMF acts as the C1 synthon; it undergoes a condensation reaction with the hydrazide moiety of the starting material to generate a hydrazone intermediate. Subsequent intramolecular cyclization followed by the elimination of dimethylamine yields the desired 3-trifluoromethyl-1,2,4-triazole product. Alternatively, the N-methyl group of DMF can serve as the carbon source. In this scenario, iodine activates the solvent to form an amine salt species, which then engages in nucleophilic addition with the hydrazide. Following the elimination of N-methylformamide, an azadiene intermediate is generated, which undergoes further intramolecular nucleophilic attack and oxidative aromatization to furnish the final heterocyclic core. This dual-reactivity profile underscores the sophistication of the method, allowing for high reaction efficiency without the need for exotic reagents.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity heterocyclic compounds. The use of air as the terminal oxidant ensures that the oxidative aromatization step proceeds cleanly, minimizing the formation of over-reduced byproducts that are common in metal-catalyzed reductions. Furthermore, the byproducts of the reaction, such as dimethylamine salts or iodide species, are generally water-soluble and easily removed during the aqueous workup phase. This inherent cleanliness of the reaction profile reduces the burden on downstream purification processes, such as column chromatography or recrystallization, which are often the most costly and time-consuming steps in fine chemical production. For R&D directors focused on impurity profiles, understanding that the reaction proceeds through well-defined hydrazone or azadiene intermediates allows for better prediction and monitoring of potential side products, ensuring the final API intermediate meets stringent regulatory specifications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the trifluoroethyliminohydrazide substrate, molecular iodine, and DMF solvent, followed by heating under ambient air pressure. The detailed standardized synthesis steps, including precise stoichiometric ratios, temperature ramping profiles, and specific workup procedures to maximize yield and purity, are outlined in the guide below.
- Combine molecular iodine (1.5 equivalents) and trifluoroethyliminohydrazide substrate in a reaction vessel containing DMF solvent.
- Heat the reaction mixture to 110-130°C under an air atmosphere and maintain stirring for 10-15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, and silica gel column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis route presents a compelling value proposition centered on cost stability and operational resilience. The shift away from specialized, high-cost reagents towards commodity chemicals directly impacts the bottom line, offering substantial cost savings in raw material procurement. Additionally, the robustness of the reaction conditions mitigates the risk of batch failures due to environmental factors, ensuring a more consistent supply of critical intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the elimination of expensive transition metal catalysts and specialized C1 sources. By utilizing DMF, a solvent that is already present in vast quantities in most chemical facilities, as a reactant, the process effectively turns a cost center (solvent disposal) into a value-added input. This dual usage drastically lowers the material cost per kilogram of the final product. Furthermore, the avoidance of noble metals like palladium or rhodium removes the need for costly metal scavenging steps to meet residual metal limits in pharmaceutical products, thereby simplifying the purification train and reducing processing time and associated utility costs.
- Enhanced Supply Chain Reliability: The reliance on globally available, commodity-grade starting materials such as molecular iodine and DMF ensures that the supply chain is not vulnerable to the geopolitical or logistical disruptions that often plague the supply of specialized fine chemicals. Since the reaction does not require strict anhydrous or anaerobic conditions, the manufacturing infrastructure requirements are significantly lowered. Facilities do not need to invest in complex nitrogen blanketing systems or dried solvent loops, allowing for production in a wider range of multipurpose reactors. This flexibility enhances supply continuity and reduces lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles. The atom economy is improved by incorporating the solvent into the product structure, reducing the overall volume of chemical waste generated per unit of product. The use of air as an oxidant avoids the generation of stoichiometric amounts of heavy metal waste or hazardous oxidizing agents. The simplicity of the workup, involving basic filtration and washing, minimizes the consumption of auxiliary solvents and silica gel. These factors collectively contribute to a lower environmental footprint, facilitating easier regulatory compliance and reducing waste disposal costs, which is increasingly critical for sustainable commercial scale-up of complex polymer additives and fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this route for adoption.
Q: Does this synthesis require strict anhydrous or anaerobic conditions?
A: No, one of the primary advantages of this patented method (CN114920707B) is that it operates efficiently under a standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, significantly simplifying operational requirements.
Q: What is the role of DMF in this reaction mechanism?
A: DMF serves a dual function as both the reaction solvent and the critical C1 carbon source. Depending on the pathway, either the formyl group or the N-methyl group of DMF participates in the tandem cyclization to construct the triazole ring.
Q: Is this method scalable for industrial production of API intermediates?
A: Yes, the use of commodity chemicals like DMF and molecular iodine, combined with mild reaction conditions and simple workup procedures, makes this process highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology disclosed in CN114920707B for the production of next-generation pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this iodine-promoted cyclization, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this innovative technology for your pipeline projects. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our triazole derivatives and to discuss route feasibility assessments for your target molecules, ensuring your supply chain remains robust and competitive.
