Advanced Iodine-Catalyzed Triazole Synthesis: Commercial Scale-Up of High-Purity Pharmaceutical Intermediates with Streamlined Supply Chain
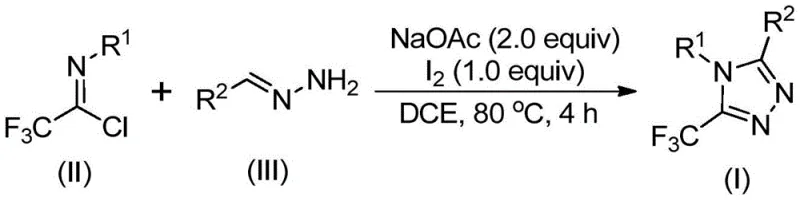
The groundbreaking patent CN110467579B introduces a novel synthetic pathway for producing 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses critical limitations in current manufacturing practices. This method represents a significant advancement over conventional approaches by eliminating the requirement for heavy metal catalysts while maintaining high reaction efficiency under ambient conditions. The process leverages readily available starting materials including sodium acetate, trifluoroethylimidoyl chloride, and hydrazone derivatives to construct these valuable heterocyclic scaffolds. Crucially, the reaction proceeds without stringent anhydrous or anaerobic requirements that typically complicate industrial scale-up. This innovation directly responds to growing industry demands for sustainable manufacturing processes that reduce environmental impact while maintaining pharmaceutical-grade purity standards essential for drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of trifluoromethylated triazoles frequently rely on expensive transition metal catalysts such as palladium or copper complexes that necessitate complex removal procedures to meet pharmaceutical purity specifications. These methods often require strictly controlled anhydrous and anaerobic environments to prevent catalyst deactivation or unwanted side reactions, significantly increasing operational complexity and capital investment in specialized equipment. Furthermore, conventional approaches using trifluorodiazoethane present substantial safety hazards due to its explosive nature and stringent handling requirements. The narrow substrate scope of existing methodologies also limits structural diversity, forcing manufacturers to develop entirely new synthetic routes for each derivative rather than utilizing a flexible platform approach. These combined factors result in higher production costs, extended timelines for process validation, and increased regulatory scrutiny during quality assurance phases.
The Novel Approach
The patented methodology overcomes these challenges through an elegant iodine-catalyzed cyclization that operates under mild conditions without heavy metal involvement. By utilizing elemental iodine as a non-toxic catalyst alongside sodium acetate as a base promoter in dichloroethane solvent at precisely controlled temperatures of 80°C, the process achieves high yields while eliminating hazardous reagents. This approach demonstrates remarkable substrate tolerance across diverse aryl and heteroaryl groups with various functional substituents including halogens and alkyl moieties. The reaction mechanism proceeds through a well-defined sequence involving base-promoted carbon-nitrogen bond formation followed by iodine-mediated oxidative cyclization. Critically, the absence of moisture sensitivity allows straightforward implementation in standard manufacturing facilities without requiring specialized infrastructure for inert atmosphere control.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism begins with base-promoted nucleophilic attack by hydrazone nitrogen on the electrophilic carbon of trifluoroethylimidoyl chloride, forming a key trifluoroacetamidine intermediate through carbon-nitrogen bond formation. This intermediate subsequently undergoes spontaneous isomerization to generate a more stable tautomeric form that positions the molecule for cyclization. The iodine catalyst then facilitates oxidative transformation through single-electron transfer processes that generate reactive iodine species capable of promoting intramolecular electrophilic substitution. This critical step forms the triazole ring structure through precise nitrogen-carbon bond formation at the C5 position while maintaining the trifluoromethyl group's integrity. The mechanism avoids radical pathways that could lead to undesired side products by operating through controlled polar intermediates that ensure regioselective cyclization.
Impurity control is achieved through the reaction's inherent selectivity and mild conditions that minimize decomposition pathways. The absence of strong acids or bases prevents unwanted hydrolysis or rearrangement reactions that commonly generate impurities in traditional syntheses. The precise temperature control at 80°C prevents thermal degradation while ensuring complete conversion without promoting side reactions. Elemental iodine's moderate oxidizing power selectively drives the cyclization without over-oxidizing sensitive functional groups present in diverse substrates. The process design incorporates built-in purification through simple filtration followed by standard column chromatography using silica gel, which effectively removes any residual iodine or minor byproducts without requiring specialized techniques. This streamlined approach consistently delivers high-purity products meeting stringent pharmaceutical requirements without additional costly purification steps.
How to Synthesize 5-Trifluoromethyl Triazole Efficiently
This innovative synthesis route represents a significant advancement in triazole chemistry by providing a robust platform for producing high-value pharmaceutical intermediates with exceptional efficiency. The patented method eliminates multiple pain points associated with conventional approaches through its clever use of non-metallic catalysis and operationally simple procedures. Detailed standardized synthesis steps are provided below to enable seamless implementation in manufacturing environments while maintaining consistent product quality across different scales.
- Combine sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in dichloroethane solvent under ambient conditions without requiring anhydrous or anaerobic environments.
- Heat the reaction mixture to precisely 80°C and maintain this temperature for three hours to facilitate carbon-nitrogen bond formation and isomerization.
- Introduce elemental iodine into the system and continue heating at 80°C for one additional hour before initiating standard purification protocols.
Commercial Advantages for Procurement and Supply Chain Teams
This manufacturing innovation delivers substantial strategic benefits that directly address critical pain points in pharmaceutical supply chains while enhancing overall operational efficiency. The elimination of heavy metal catalysts fundamentally transforms cost structures by removing expensive purification requirements that typically consume significant resources in traditional processes. The use of readily available starting materials creates inherent supply chain resilience by reducing dependency on specialized chemical suppliers while enabling multiple sourcing options for key precursors. These advantages collectively strengthen procurement positions through improved cost predictability and reduced vulnerability to market fluctuations in specialty chemical markets.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes both material costs and complex removal processes required to meet regulatory purity standards. This simplification reduces capital expenditure by avoiding specialized equipment for metal residue testing while lowering operational costs through decreased solvent consumption during purification. The use of inexpensive sodium acetate and elemental iodine as catalytic system components provides significant material cost savings compared to precious metal alternatives without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through the use of commercially available starting materials that are widely accessible from multiple global suppliers. The absence of moisture-sensitive reagents eliminates complex storage requirements while enabling straightforward transportation logistics without specialized packaging or environmental controls. This robustness ensures consistent production continuity even during supply chain disruptions as alternative vendors can be rapidly qualified due to standardized material specifications.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production volumes due to its straightforward thermal profile and absence of hazardous intermediates. Environmental impact is minimized through reduced waste generation from eliminated metal removal steps while maintaining high atom economy in the core transformation. The operationally simple procedure requires minimal specialized infrastructure investment when scaling from pilot plant to full commercial production capacity.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations based on patent specifications and practical implementation experience with this innovative synthesis platform. These insights have been developed through rigorous analysis of the patented methodology and its application across diverse manufacturing scenarios.
Q: How does this method eliminate the need for anhydrous and anaerobic conditions compared to traditional triazole syntheses?
A: The iodine-catalyzed process operates effectively under ambient atmospheric conditions without moisture sensitivity. Unlike conventional methods requiring strict anhydrous environments to prevent catalyst decomposition or side reactions, this approach leverages robust non-metallic catalysis that tolerates trace water. The reaction mechanism proceeds through stable iodine-mediated oxidative steps that remain unaffected by oxygen or humidity, significantly reducing operational complexity while maintaining high product integrity.
Q: What are the key advantages of using elemental iodine instead of heavy metal catalysts in this synthesis?
A: Elemental iodine provides substantial environmental and economic benefits by eliminating toxic heavy metal residues. This avoids costly purification steps required to remove palladium or copper catalysts from final products. The iodine catalyst is inexpensive, readily available, and generates minimal waste streams. Crucially, it enables direct synthesis without requiring specialized equipment for metal removal, thereby reducing both capital expenditure and operational costs while ensuring cleaner product profiles suitable for pharmaceutical applications.
Q: How does substrate design flexibility enable production of diverse trifluoromethyl triazole derivatives?
A: The modular reaction design allows systematic variation of R¹ and R² substituents through precursor selection. By choosing different aryl groups on trifluoroethylimidoyl chloride or modifying hydrazone structures with various alkenyl/aryl moieties, manufacturers can precisely tailor molecular properties. This flexibility accommodates diverse functional groups including halogens and alkyl chains without compromising yield, enabling customized synthesis of target compounds for specific pharmaceutical applications while maintaining consistent process parameters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. We specialize in transforming patented methodologies like this iodine-catalyzed triazole synthesis into robust manufacturing processes that deliver consistent quality across all production scales. Our technical team possesses deep expertise in optimizing reaction parameters for maximum yield while ensuring complete regulatory compliance throughout the production lifecycle.
Leverage our technical procurement team's expertise by requesting a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements. We provide comprehensive support including specific COA data and route feasibility assessments to help you evaluate this innovative process against your current supply chain needs.
