Advanced Organocatalytic Synthesis of Axial Chiral Indole-Naphthalene Compounds for Pharmaceutical Applications
Advanced Organocatalytic Synthesis of Axial Chiral Indole-Naphthalene Compounds for Pharmaceutical Applications
The landscape of asymmetric synthesis is continually evolving, driven by the urgent demand for efficient access to complex chiral scaffolds essential for modern drug discovery. Patent CN110452150B introduces a groundbreaking methodology for the preparation of axial chiral indole-naphthalene compounds, a class of molecules with significant potential as organic small-molecule catalysts and ligands. This technology addresses a critical gap in the prior art by enabling the one-step construction of the axial chiral skeleton from racemic starting materials through a dynamic kinetic resolution strategy. Unlike traditional multi-step sequences that often suffer from low atom economy and poor stereocontrol, this novel approach leverages the power of chiral phosphoric acid organocatalysis to achieve high yields and exceptional enantioselectivity under remarkably mild conditions. The structural versatility of the resulting compounds, as depicted in the general formula below, allows for extensive customization to meet specific catalytic requirements in asymmetric transformations.
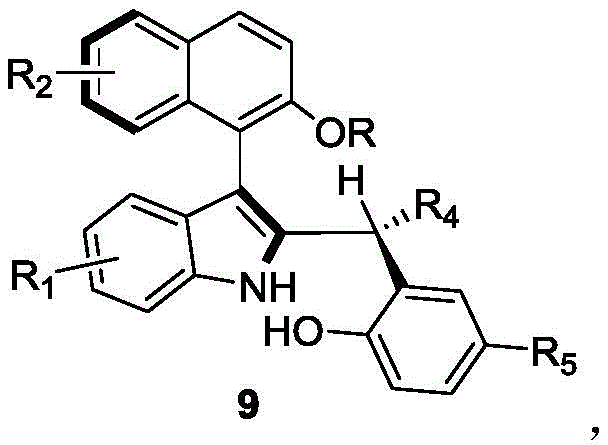
For research directors and process chemists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift in how biaryl axial chirality is installed. The ability to generate high-value chiral building blocks from inexpensive, commercially available racemates significantly lowers the barrier to entry for developing new asymmetric catalytic systems. Furthermore, the operational simplicity of the protocol—requiring only standard filtration and concentration steps post-reaction—makes it an ideal candidate for rapid scale-up and integration into existing manufacturing workflows without the need for specialized high-pressure or cryogenic equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axial chiral indole-naphthalene frameworks has relied heavily on cross-coupling reactions between pre-functionalized indole and naphthalene derivatives. These conventional pathways are fraught with significant drawbacks that hinder their widespread industrial adoption. Primarily, they often necessitate the use of expensive transition metal catalysts, such as palladium or rhodium complexes, which not only inflate raw material costs but also introduce stringent regulatory hurdles regarding heavy metal residues in final pharmaceutical products. Additionally, these coupling reactions frequently require harsh reaction conditions, including elevated temperatures and strong bases, which can lead to the decomposition of sensitive functional groups and result in poor overall yields. The substrate scope is often narrow, limiting the structural diversity of the accessible chiral library, and the stereocontrol is frequently dependent on the use of stoichiometric chiral auxiliaries rather than catalytic amounts of chiral inducers, leading to excessive waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a chiral phosphoric acid-catalyzed asymmetric addition reaction to construct the target architecture. This innovative route bypasses the need for transition metals entirely, relying instead on robust organic small-molecule catalysis. The core transformation involves the reaction of a 2-substituted indole derivative (Formula 7) with a naphthol-based electrophile (Formula 8) in the presence of a tailored chiral phosphoric acid catalyst. As illustrated in the reaction scheme below, this process proceeds smoothly at ambient temperatures (20-30°C) in a mixed solvent system of 1,1,2,2-tetrachloroethane and p-xylene. The use of 5 Angstrom molecular sieves as an additive further drives the equilibrium forward by sequestering water, ensuring high conversion rates. This approach not only simplifies the synthetic sequence to a single step but also dramatically improves the environmental profile of the manufacturing process by eliminating toxic metal waste and reducing energy consumption.
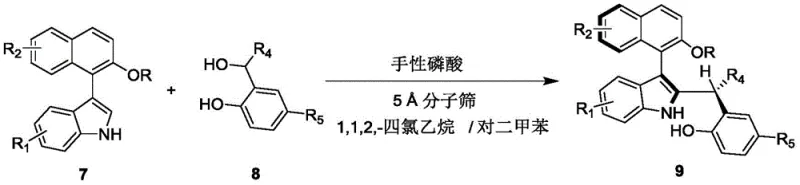
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
The success of this synthesis hinges on the precise molecular recognition and activation facilitated by the chiral phosphoric acid catalyst. These catalysts, typically derived from BINOL or SPINOL backbones, function as dual hydrogen-bond donors that simultaneously activate both the nucleophilic indole and the electrophilic naphthol species. This bifunctional activation creates a highly organized chiral pocket within the transition state, effectively discriminating between the enantiomers of the racemic starting material. The bulky substituents at the 3,3'-positions of the catalyst backbone, such as the 9-anthracenyl groups found in the preferred catalyst (Formula 6), play a crucial role in enforcing this stereochemical discrimination through steric repulsion. By carefully tuning the steric bulk and electronic properties of these substituents, the catalyst directs the incoming nucleophile to attack from a specific face, thereby locking in the desired axial configuration with high fidelity.
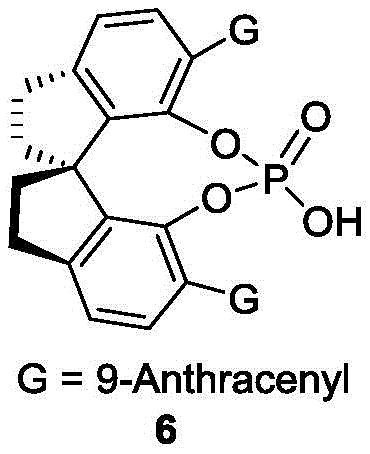
Beyond the primary bond formation, the mechanism inherently controls the impurity profile of the final product. The dynamic nature of the kinetic resolution ensures that the undesired enantiomer of the starting material is continuously racemized and recycled into the productive catalytic cycle, theoretically allowing for 100% yield of the desired chiral product from a racemic feed. This stands in contrast to classical resolutions which are capped at a maximum of 50% yield. The result is a process that delivers products with enantiomeric ratios (er) consistently exceeding 95:5, and in optimized cases reaching >99:1 after simple recrystallization. Such high optical purity is paramount for downstream applications, particularly when these compounds are employed as ligands in sensitive asymmetric catalytic cycles where even trace amounts of the opposite enaantiomer can degrade the performance of the final drug substance.
How to Synthesize Axial Chiral Indole-Naphthalene Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol begins with the preparation of a homogeneous reaction mixture containing the indole substrate and the naphthol electrophile in the specified binary solvent system. The addition of the chiral catalyst and molecular sieves initiates the transformation, which is monitored via thin-layer chromatography (TLC) to ensure complete consumption of the starting materials. Following the reaction period, typically around 12 hours at room temperature, the workup involves a straightforward filtration to remove the solid molecular sieves, followed by solvent evaporation. The crude product is then purified using standard silica gel column chromatography with a petroleum ether and ethyl acetate gradient. For a detailed breakdown of the specific molar ratios, solvent volumes, and purification parameters, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining compound of formula 7 and compound of formula 8 in a mixed solvent of 1,1,2,2-tetrachloroethane and p-xylene.
- Add 5 Angstrom molecular sieves and a chiral phosphoric acid catalyst (such as compound of formula 6) to the mixture.
- Stir the reaction at 20-30°C until completion, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages that directly impact the bottom line and operational resilience. The shift from transition-metal catalysis to organocatalysis eliminates the dependency on volatile precious metal markets, stabilizing raw material costs and insulating the supply chain from geopolitical disruptions associated with mining and refining rare earth elements. Furthermore, the use of readily available, commodity-grade solvents and starting materials ensures a robust and continuous supply stream, reducing the risk of production delays caused by specialty reagent shortages. The simplified downstream processing, which avoids complex metal scavenging steps, translates into shorter batch cycle times and reduced utility consumption, contributing to substantial cost savings in large-scale manufacturing operations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated purification infrastructure significantly lowers the cost of goods sold (COGS). By removing the need for specialized metal removal resins or extensive aqueous workups to meet residual metal specifications, the process streamlines the production workflow. This efficiency gain allows for a more competitive pricing structure for the final chiral intermediates, making high-value asymmetric building blocks accessible for a broader range of therapeutic programs without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous organic catalysts and common solvents enhances the overall reliability of the supply chain. Unlike processes requiring air-sensitive reagents or cryogenic conditions, this method can be executed in standard glass-lined reactors with minimal safety risks. This operational robustness facilitates easier technology transfer between sites and reduces the likelihood of batch failures due to environmental fluctuations, ensuring consistent delivery schedules for downstream pharmaceutical customers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The mild reaction conditions and high atom economy of this process align perfectly with green chemistry principles, simplifying regulatory compliance and waste management. The absence of heavy metal waste streams reduces the burden on wastewater treatment facilities and lowers disposal costs. Moreover, the scalability of the reaction has been demonstrated through its tolerance to various substrate substitutions, indicating that the process can be adapted to produce a diverse library of analogs without requiring fundamental changes to the manufacturing infrastructure, thus future-proofing the production capacity against evolving market demands.
Frequently Asked Questions (FAQ)
To assist our partners in evaluating the technical feasibility and commercial viability of this technology, we have compiled answers to common inquiries regarding the synthesis and application of these axial chiral compounds. The following insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a transparent view of the process capabilities and limitations. We encourage potential collaborators to review these details closely to understand how this methodology can be integrated into their specific development pipelines.
Q: What is the primary advantage of this synthesis method over traditional coupling reactions?
A: This method utilizes dynamic kinetic resolution via asymmetric addition, allowing for the construction of the axial chiral skeleton in a single step from racemic raw materials under mild conditions, avoiding the harsh conditions and limited substrate scope of traditional coupling methods.
Q: What level of enantioselectivity can be achieved with this process?
A: The process demonstrates excellent stereocontrol, consistently achieving enantiomeric ratios (er) greater than 95:5, with specific examples reaching up to 99.8:0.2 after recrystallization, ensuring high optical purity suitable for pharmaceutical applications.
Q: Can the resulting compounds be further derivatized into functional catalysts?
A: Yes, the synthesized axial chiral indole-naphthalene compounds serve as versatile precursors that can be chemically modified into chiral phosphine catalysts, which have been proven effective in catalyzing asymmetric cyclization reactions such as the [4+1] cyclization of o-hydroxy-p-benzoquinone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Indole-Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing high-quality chiral intermediates for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of axial chiral indole-naphthalene compounds meets the exacting standards required by the global pharmaceutical industry. Our state-of-the-art facilities are equipped to handle the specific solvent systems and purification requirements of this organocatalytic process, delivering products with consistent optical purity and chemical integrity.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your drug development timeline while optimizing your overall production budget. Contact us today to discuss how we can support your supply chain with reliable, high-performance chiral building blocks.
