Scaling Green Electrochemical Synthesis of 5-Arylthiouracil Derivatives for Pharmaceutical Manufacturing
Scaling Green Electrochemical Synthesis of 5-Arylthiouracil Derivatives for Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking sustainable and efficient pathways to synthesize bioactive heterocycles, particularly uracil derivatives which serve as critical scaffolds for antiviral and anticancer agents. Patent CN111705329B introduces a groundbreaking electrochemical synthesis method for 5-arylthiouracil compounds that fundamentally shifts the paradigm from traditional chemical oxidation to electron-driven catalysis. This technology utilizes a hexafluoroisopropanol (HFIP) solution containing uracil, aromatic thiophenol, and an iodine salt as the electrolyte, operating within an undivided cell under mild direct current conditions. By replacing hazardous stoichiometric oxidants with electricity, this process achieves high atom efficiency and exceptional yields, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates. The method's reliance on readily available starting materials and its operation at room temperature further underscore its potential for immediate industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 5-arylthiouracil derivatives relied heavily on chemical oxidants that posed significant safety and environmental challenges. For instance, earlier methodologies utilized hydrogen peroxide (H2O2) in conjunction with sodium iodide catalysts, requiring up to 9 equivalents of oxidant and elevated temperatures, which introduced substantial risks of explosion and difficult product isolation. Another common approach employed potassium persulfate (K2S2O8) with molecular iodine, generating large volumes of chemical waste and necessitating complex purification steps to remove inorganic salts. These traditional routes not only inflated production costs due to reagent consumption but also created bottlenecks in cost reduction in pharmaceutical intermediate manufacturing due to the extensive downstream processing required to meet purity standards. The reliance on harsh conditions often led to side reactions, compromising the overall yield and quality of the final active pharmaceutical ingredient (API) precursor.
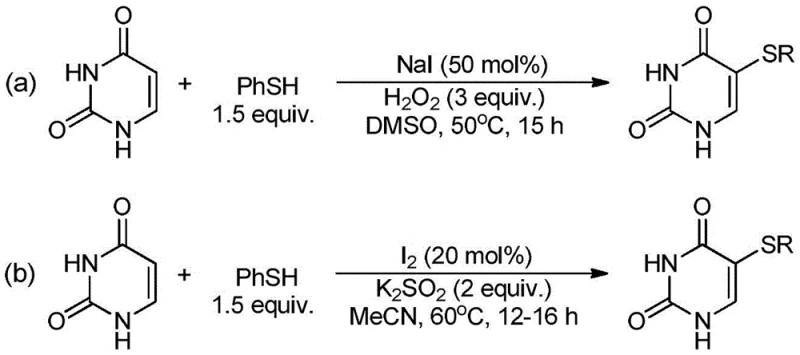
The Novel Approach
In stark contrast, the electrochemical method disclosed in CN111705329B eliminates the need for external chemical oxidants entirely, using electrons as a clean and traceless reagent to drive the oxidative dehydrogenation coupling reaction. The process operates efficiently at room temperature with a constant current of 18 mA, utilizing a reticulated vitreous carbon (RVC) anode and a nickel foam cathode to facilitate the transformation. This setup allows for the direct activation of the iodine salt catalyst at the anode surface, generating reactive species that couple the uracil and thiophenol substrates with high precision. The result is a streamlined workflow where the product can be isolated simply by adding water to the reaction mixture, causing the 5-arylthiouracil to precipitate as high-purity crystals without the need for chromatography. This simplicity represents a major leap forward for any reliable pharmaceutical intermediate supplier aiming to optimize their production lines.

Mechanistic Insights into Iodide-Mediated Electrochemical Oxidation
The success of this synthesis lies in the elegant catalytic cycle mediated by the iodide salt, specifically tetramethylammonium iodide (Me4NI), which acts as a redox mediator. At the anode, iodide ions (I-) lose electrons to form electrophilic iodine species (I+), which subsequently react with the aromatic thiophenol to generate a highly reactive thiophenyl cation intermediate (RS+). This cationic species then attacks the C5 position of the uracil ring, forming a sulfonium ion intermediate that rapidly rearranges into an iminium cation. The final step involves a dehydroaromatization process that restores the aromaticity of the pyrimidine ring, releasing the target 5-arylthiouracil product and regenerating protons. These protons migrate to the cathode, where they are reduced to form hydrogen gas, completing the circuit without generating harmful byproducts. Understanding this mechanism is crucial for R&D teams looking to replicate the high-purity 5-arylthiouracil synthesis, as it highlights the importance of maintaining the correct current density to sustain the catalytic turnover.
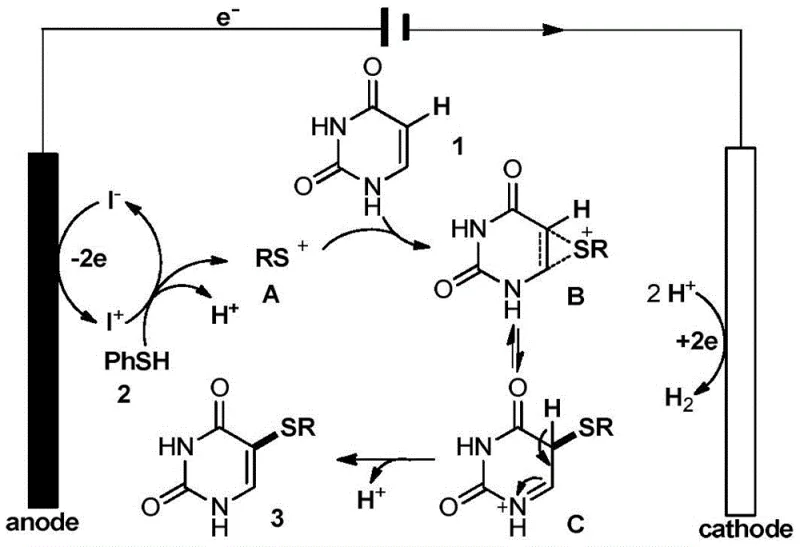
Furthermore, the choice of solvent plays a pivotal role in stabilizing these charged intermediates and ensuring reaction efficiency. The patent data explicitly demonstrates that polar aprotic solvents like acetonitrile or DMF fail to support the reaction, yielding only trace amounts of product. Hexafluoroisopropanol (HFIP), however, provides the necessary environment to solubilize the substrates while stabilizing the cationic intermediates through hydrogen bonding interactions. This specific solvent effect minimizes side reactions such as over-oxidation or polymerization, thereby enhancing the selectivity of the coupling. For procurement managers, this mechanistic robustness translates to consistent batch-to-batch quality, reducing the risk of failed runs and ensuring a steady supply of critical materials for downstream drug development pipelines.
How to Synthesize 5-Arylthiouracil Efficiently
Implementing this electrochemical protocol requires precise control over reaction parameters to achieve the reported yields of up to 98%. The standard procedure involves charging an undivided cell with uracil and aromatic thiophenol in a 1:1 molar ratio, along with 15 mol% of tetramethylammonium iodide in 8 mL of HFIP. The electrode configuration is critical, with the RVC anode and nickel foam cathode providing the optimal surface area for electron transfer. Maintaining a constant current of 18 mA at room temperature for 12 hours ensures complete conversion while preventing the degradation of sensitive functional groups. Once the reaction is complete, the workup is remarkably simple: adding excess water precipitates the product, which can be filtered and dried to obtain the pure compound. Detailed standardized synthesis steps follow below for technical reference.
- Prepare the electrolyte by dissolving uracil compound, aromatic thiophenol, and tetramethylammonium iodide (15 mol%) in hexafluoroisopropanol (HFIP) solvent within an undivided cell.
- Insert a reticulated vitreous carbon (RVC) anode and a nickel foam cathode into the solution, ensuring proper spacing for efficient current distribution.
- Apply a constant direct current of 18 mA at room temperature for approximately 12 hours, then precipitate the product by adding excess water and filter to obtain the pure compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the transition to this electrochemical methodology offers profound strategic benefits beyond mere technical novelty. By removing the dependency on hazardous chemical oxidants like hydrogen peroxide and potassium persulfate, manufacturers can significantly reduce the costs associated with reagent procurement, storage, and hazardous waste disposal. The elimination of chromatographic purification steps further drives down operational expenses, as it reduces solvent consumption and labor time, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. Additionally, the use of electricity as the primary driving force decouples production from the volatility of chemical reagent markets, providing a more stable and predictable cost structure for long-term contracts. This stability is essential for maintaining competitive pricing in the global API market.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive stoichiometric oxidants and complex purification protocols, leading to substantial savings in raw material and processing costs. The ability to use a 1:1 molar ratio of substrates without excess reagents minimizes waste and maximizes atom economy, ensuring that every gram of input material contributes to the final product value. Furthermore, the simple water precipitation workup avoids the use of large volumes of organic extraction solvents, reducing both material costs and environmental compliance burdens.
- Enhanced Supply Chain Reliability: The reliance on electricity and readily available commodity chemicals like uracil and thiophenols ensures a robust supply chain that is less susceptible to disruptions. Unlike methods requiring specialized or hazardous oxidants that may face shipping restrictions, this electrochemical approach can be implemented in standard facilities with basic electrical infrastructure. This accessibility facilitates reducing lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more agilely to fluctuating market demands and urgent client requests.
- Scalability and Environmental Compliance: Operating at room temperature and atmospheric pressure makes this process inherently safer and easier to scale from laboratory to pilot and commercial production. The absence of explosive oxidants removes significant safety barriers to scaling, while the generation of hydrogen gas as the only byproduct aligns with green chemistry principles. This environmental profile simplifies regulatory approvals and supports corporate sustainability goals, making it an attractive option for companies aiming to minimize their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 5-arylthiouracil derivatives. These answers are derived directly from the experimental data and mechanistic studies presented in patent CN111705329B, providing clarity on the feasibility and advantages of this technology. Understanding these details helps stakeholders make informed decisions about integrating this green chemistry approach into their existing manufacturing portfolios.
Q: Why is hexafluoroisopropanol (HFIP) critical for this electrochemical reaction?
A: According to patent CN111705329B, HFIP is the optimal solvent because other common solvents like acetonitrile, ethanol, DMSO, and DMF result in trace or no reaction. Its strong polarity and unique hydrogen-bonding properties likely stabilize the charged intermediates generated during the anodic oxidation process.
Q: What are the advantages of using electricity over chemical oxidants like hydrogen peroxide?
A: Traditional methods require hazardous chemical oxidants like hydrogen peroxide or potassium persulfate, which create safety risks (explosion) and significant chemical waste. This electrochemical method uses electrons as a 'traceless' oxidant, eliminating the need for external oxidizing agents, thereby simplifying purification and reducing environmental impact.
Q: Can this method tolerate various functional groups on the thiophenol substrate?
A: Yes, the method demonstrates excellent functional group compatibility. It successfully synthesizes derivatives with methyl, fluoro, and other substituents on the aromatic ring with high yields (e.g., 93-96%), indicating robustness for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Arylthiouracil Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of vital pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN111705329B can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-arylthiouracil delivered meets the highest international standards for drug substance manufacturing. We are committed to leveraging our technical expertise to help clients navigate the complexities of process development and scale-up.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full capabilities of this electrochemical route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for efficiency, safety, and sustainability in the evolving landscape of fine chemical synthesis.
