Advanced One-Pot Synthesis of 1,2,3-Trisubstituted Indolizines for Commercial Scale-Up
Advanced One-Pot Synthesis of 1,2,3-Trisubstituted Indolizines for Commercial Scale-Up
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting broad pharmacological activity. Patent CN111269227A introduces a groundbreaking preparation method for 1,2,3-trisubstituted indolizine derivatives, addressing critical bottlenecks in the production of cyclooxygenase and lipoxygenase inhibitors. This technology represents a paradigm shift from multi-step, hazardous protocols to a streamlined, environmentally benign one-pot process. By leveraging stable and inexpensive starting materials such as brominated substituted acetophenone pyridinium salts and bromoethynylbenzene, the invention achieves high synthesis efficiency without compromising on safety or purity standards. For R&D directors and procurement specialists alike, this patent offers a viable pathway to secure reliable pharmaceutical intermediate supplier status while drastically reducing the ecological footprint of API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolizine core has been plagued by reliance on harsh reagents and complex operational sequences that hinder scalability. As illustrated in the prior art reaction schemes, traditional pathways often necessitate the use of strong bases like sodium hydroxide coupled with carcinogenic solvents such as dichloromethane. Furthermore, oxidative cyclization steps frequently employ chromium trioxide, a highly toxic and corrosive agent that demands rigorous waste management and poses significant occupational health risks. Alternative methods utilizing transition metal catalysts like ferric chloride or cuprous fluoride introduce another layer of complexity, requiring expensive ligands and generating products contaminated with heavy metal residues that are difficult to remove to ppm levels required for pharmaceutical use. These legacy processes are not only operationally cumbersome but also economically inefficient due to low step economy and high disposal costs.
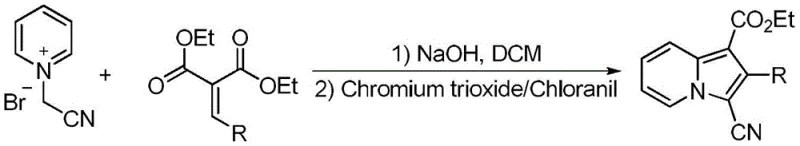
The Novel Approach
In stark contrast to these archaic methodologies, the disclosed invention utilizes a direct, base-mediated cyclization strategy that operates under mild and safe conditions. The process involves reacting a brominated substituted acetophenone pyridinium quaternary ammonium salt with bromoethynylbenzene in the presence of potassium carbonate and dimethyl sulfoxide (DMSO) at a moderate temperature of 90°C. This single-step transformation eliminates the need for transition metal catalysts and high-toxicity oxidants entirely, resulting in a cleaner reaction profile. The use of DMSO, a low-toxicity and non-flammable solvent, further enhances operational safety compared to volatile organic compounds like toluene or dichloroethane. The structural diversity achievable through this method is exemplified by the successful synthesis of various derivatives, including those with methyl, methoxy, chloro, and bromo substituents, all obtained as pure yellow solids with excellent yields ranging from 68% to 92%.
Mechanistic Insights into Base-Mediated Cyclization
The mechanistic elegance of this synthesis lies in its ability to forge the fused bicyclic indolizine system through a concerted sequence of nucleophilic attacks and eliminations without external oxidation. The reaction initiates with the deprotonation of the acidic alpha-proton on the pyridinium salt by the carbonate base, generating a reactive ylide or carbanion species. This nucleophile then attacks the electron-deficient triple bond of the bromoethynylbenzene, triggering a cascade of intramolecular cyclization events. Unlike oxidative methods that require stoichiometric amounts of dangerous oxidants to restore aromaticity, this pathway likely proceeds through a spontaneous aromatization driven by the thermodynamic stability of the indolizine ring system. The absence of radical intermediates typically associated with iron or copper catalysis minimizes the formation of polymeric byproducts and tar, thereby simplifying the impurity profile. This mechanistic clarity allows for precise control over regioselectivity, ensuring that the 1,2,3-substitution pattern is established with high fidelity.
From an impurity control perspective, the exclusion of transition metals is a decisive advantage for regulatory compliance. In conventional copper-catalyzed routes, residual metal levels often exceed ICH Q3D guidelines, necessitating costly scavenging steps using silica-bound thiols or specialized resins. By avoiding these catalysts altogether, the new method inherently produces a crude product with a significantly lower burden of inorganic impurities. Furthermore, the stability of the bromoethynylbenzene reactant under the reaction conditions prevents premature polymerization, a common issue with allene-based precursors used in other methods. The result is a robust process where the primary impurities are likely unreacted starting materials or simple hydrolysis products, which are easily removed during the aqueous workup and subsequent column chromatography. This purity profile is critical for downstream applications where trace contaminants could affect the efficacy or safety of the final drug substance.
How to Synthesize 1,2,3-Trisubstituted Indolizine Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings, requiring standard glassware and heating equipment. The protocol begins by charging a reactor with the specific brominated pyridinium salt, the alkyne coupling partner, and potassium carbonate in DMSO, followed by heating to 90°C for a short duration. Detailed standardized operating procedures regarding stoichiometry, addition rates, and specific workup parameters are essential for reproducibility and are outlined in the technical documentation below. Operators should note that the reaction completion is typically achieved within 2 hours, allowing for rapid turnover and high throughput. The simplicity of the workup, involving a simple water quench and extraction, minimizes solvent consumption and labor time, making it an ideal candidate for continuous manufacturing optimization.
- Combine brominated substituted acetophenone pyridinium quaternary ammonium salt, bromoethynylbenzene, and potassium carbonate in dimethyl sulfoxide (DMSO) solvent.
- Heat the reaction mixture to 90°C and maintain stirring for approximately 2 hours to ensure complete cyclization.
- Quench the reaction by pouring into water, followed by extraction, drying, and column chromatography to isolate the pure indolizine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical yield. The elimination of expensive transition metal catalysts and ligands directly reduces the raw material cost base, while the removal of toxic oxidants like chromium trioxide drastically lowers waste disposal fees and regulatory compliance burdens. The use of stable, commercially available starting materials ensures a resilient supply chain that is less susceptible to the volatility often seen with specialized reagents like diazo compounds or unstable allenes. Furthermore, the shortened reaction time of just 2 hours significantly increases asset utilization rates, allowing manufacturing facilities to produce larger volumes of high-purity pharmaceutical intermediates within the same timeframe. This operational efficiency supports the commercial scale-up of complex heterocyclic building blocks, ensuring consistent supply continuity for downstream API production.
- Cost Reduction in Manufacturing: The economic impact of removing transition metal catalysts cannot be overstated, as it eradicates the need for expensive metal scavengers and extensive purification trains. By utilizing inexpensive inorganic bases like potassium carbonate instead of precious metal complexes, the variable cost per kilogram of product is significantly optimized. Additionally, the high step economy of this one-pot process reduces labor costs and energy consumption associated with multiple isolation and purification stages. These cumulative savings contribute to a more competitive pricing structure for the final indolizine derivatives, enhancing margin potential for generic drug manufacturers.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved by relying on commodity chemicals such as acetophenones and pyridines rather than bespoke, unstable intermediates. The robustness of the reaction conditions means that production is less likely to be disrupted by minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining just-in-time inventory levels and meeting tight delivery schedules for global pharmaceutical clients. The ability to source raw materials from multiple vendors further mitigates the risk of supply chain bottlenecks, securing long-term production viability.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry principles, facilitating easier permitting and regulatory approval for large-scale manufacturing sites. The absence of halogenated solvents like dichloromethane and the use of non-flammable DMSO reduce fire hazards and VOC emissions, creating a safer working environment. Scalability is inherently supported by the homogeneous nature of the reaction mixture and the lack of exothermic hazards associated with strong oxidants. This allows for seamless technology transfer from gram-scale R&D to multi-ton commercial production without the need for major process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indolizine synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the process capabilities. Stakeholders are encouraged to review these points to assess the feasibility of integrating this method into their existing manufacturing portfolios. Comprehensive technical support is available to assist with process validation and customization for specific derivative targets.
Q: Why is this new indolizine synthesis method superior to traditional chromium-based oxidation?
A: Traditional methods often rely on highly toxic and corrosive oxidants like chromium trioxide, which pose severe environmental and safety hazards. This novel protocol eliminates the need for such hazardous reagents, utilizing a benign base-mediated cyclization instead.
Q: Does this process involve transition metal catalysts that require removal?
A: No, unlike copper or iron-catalyzed routes found in prior art, this method is completely transition-metal-free. This ensures the final pharmaceutical intermediate is free from heavy metal residues, simplifying downstream purification.
Q: What are the typical yields and reaction conditions for this synthesis?
A: The reaction operates efficiently at 90°C in DMSO solvent and typically completes within 2 hours. Reported isolated yields for various substituted derivatives range robustly from 68% to 92%, demonstrating high step economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Trisubstituted Indolizine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,2,3-trisubstituted indolizine derivatives meets the highest international standards. Our infrastructure is designed to handle complex heterocyclic chemistry with precision, offering a secure partnership for companies seeking to optimize their API supply chains.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener, more efficient protocol. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as a trusted partner in fine chemical manufacturing. Contact us today to explore collaborative opportunities that drive value and innovation in your pharmaceutical projects.
