Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)-one Derivatives for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)-one Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those with proven biological significance. Quinolin-2(1H)-one derivatives represent a privileged scaffold in medicinal chemistry, serving as key structural motifs in a vast array of bioactive compounds ranging from immunomodulators to receptor antagonists and MAP kinase inhibitors. As illustrated in the structural diversity of known bioactive molecules, the ability to efficiently access these cores is paramount for drug discovery pipelines. 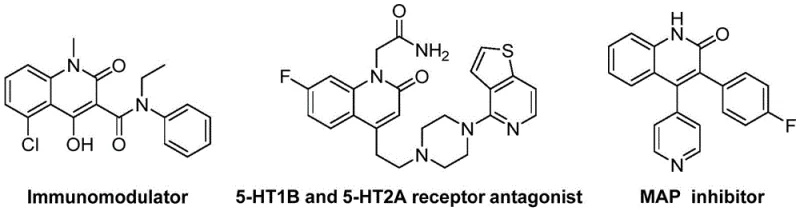 . A recent technological breakthrough detailed in patent CN114478375A introduces a highly efficient preparation method for 3-alkenyl quinolin-2(1H)-one derivatives. This innovation leverages a palladium-catalyzed reductive aminocarbonylation strategy that transforms simple, commercially available precursors into complex heterocyclic architectures with remarkable efficiency. For R&D directors and procurement managers alike, this development signals a shift towards more economical and operationally simple synthetic routes that can significantly streamline the supply chain for high-purity pharmaceutical intermediates.
. A recent technological breakthrough detailed in patent CN114478375A introduces a highly efficient preparation method for 3-alkenyl quinolin-2(1H)-one derivatives. This innovation leverages a palladium-catalyzed reductive aminocarbonylation strategy that transforms simple, commercially available precursors into complex heterocyclic architectures with remarkable efficiency. For R&D directors and procurement managers alike, this development signals a shift towards more economical and operationally simple synthetic routes that can significantly streamline the supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolinone derivatives has often relied on multi-step sequences involving harsh reaction conditions, expensive reagents, or substrates that require tedious pre-functionalization. Traditional cyclization strategies frequently suffer from limited substrate scope, where the presence of sensitive functional groups can lead to decomposition or side reactions, thereby reducing overall yield and purity. Furthermore, many conventional methods utilize allyl chlorides, acetates, or carbonates as electrophiles, which can generate stoichiometric amounts of waste salts and require rigorous purification steps to remove residual halides or acidic byproducts. These factors collectively contribute to higher manufacturing costs and extended lead times, posing significant challenges for supply chain heads aiming to maintain continuity in API production. The reliance on specialized, less abundant starting materials in older protocols further exacerbates supply risks, making the consistent sourcing of high-quality intermediates a persistent bottleneck in the industry.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes o-nitrobenzaldehyde as a dual nitrogen and formyl source, reacting directly with allyl aryl ethers under palladium catalysis. This approach eliminates the need for pre-activated allyl halides, instead employing allyl aryl ethers which are naturally occurring, low-toxicity, and easy to handle. The reaction exhibits exceptional functional group tolerance, accommodating a wide range of substituents on both the aromatic aldehyde and the allyl ether components, including alkyl, alkoxy, halogen, and trifluoromethyl groups. 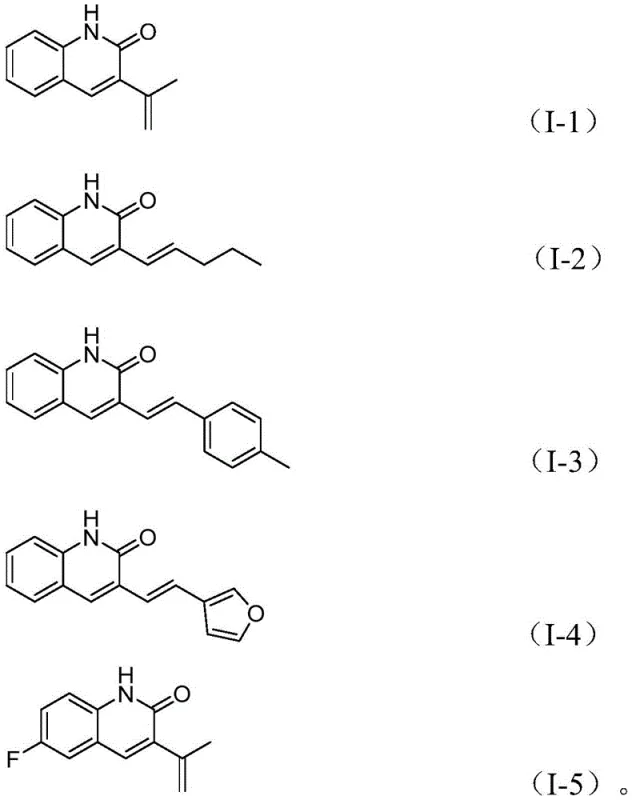 . This versatility allows for the rapid generation of diverse compound libraries, facilitating structure-activity relationship (SAR) studies without the need for route redevelopment. The operational simplicity, combined with the use of cheap and easily obtainable raw materials, positions this method as a superior alternative for cost reduction in pharmaceutical intermediate manufacturing, effectively addressing the economic and logistical pain points of traditional synthesis.
. This versatility allows for the rapid generation of diverse compound libraries, facilitating structure-activity relationship (SAR) studies without the need for route redevelopment. The operational simplicity, combined with the use of cheap and easily obtainable raw materials, positions this method as a superior alternative for cost reduction in pharmaceutical intermediate manufacturing, effectively addressing the economic and logistical pain points of traditional synthesis.
Mechanistic Insights into Palladium-Catalyzed Reductive Aminocarbonylation
The core of this transformative synthesis lies in a sophisticated palladium-catalyzed reductive aminocarbonylation mechanism. The reaction initiates with the oxidative addition of the palladium catalyst to the allyl aryl ether, followed by coordination and insertion processes that construct the carbon framework. Crucially, the use of molybdenum carbonyl serves as an external CO source, facilitating the carbonylation step necessary for forming the lactam ring of the quinolinone. Simultaneously, the nitro group of the o-nitrobenzaldehyde undergoes reduction in situ, likely mediated by the metal carbonyl species, to generate the reactive amine species required for cyclization. 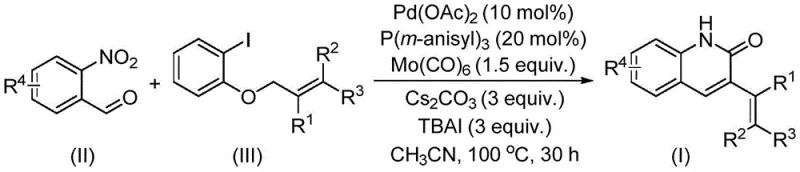 . This tandem process effectively merges reduction, carbonylation, and cyclization into a single pot, minimizing intermediate isolation and maximizing atom economy. The presence of additives like tetrabutylammonium iodide and cesium carbonate plays a vital role in stabilizing catalytic intermediates and promoting the turnover of the active palladium species, ensuring high conversion rates even with sterically demanding substrates.
. This tandem process effectively merges reduction, carbonylation, and cyclization into a single pot, minimizing intermediate isolation and maximizing atom economy. The presence of additives like tetrabutylammonium iodide and cesium carbonate plays a vital role in stabilizing catalytic intermediates and promoting the turnover of the active palladium species, ensuring high conversion rates even with sterically demanding substrates.
From an impurity control perspective, this mechanism offers distinct advantages. The concerted nature of the reaction minimizes the formation of oligomeric byproducts often seen in stepwise condensations. The specific choice of ligands, such as tris(3-methoxyphenyl)phosphine, enhances the selectivity for the desired 3-alkenyl substitution pattern, suppressing potential isomerization of the double bond. For quality control teams, this translates to a cleaner crude reaction profile, simplifying downstream purification and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications. The robustness of the catalytic cycle against various electronic environments on the aromatic rings further ensures consistent product quality across different batches, a critical factor for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Alkenyl Quinolin-2(1H)-one Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The procedure involves charging a sealed tube with the palladium catalyst system, including palladium acetate and the phosphine ligand, along with molybdenum carbonyl and the base cesium carbonate. To this mixture, the o-nitrobenzaldehyde derivative and the chosen allyl aryl ether are added in acetonitrile solvent. The reaction is then heated to approximately 100°C for a duration of roughly 30 hours to ensure complete conversion. Detailed standardized synthesis steps follow below.
- Combine palladium acetate, tris(3-methoxyphenyl)phosphine, molybdenum carbonyl, cesium carbonate, and tetrabutylammonium iodide in a reaction vessel.
- Add o-nitrobenzaldehyde and the specific allyl aryl ether substrate to the mixture along with acetonitrile solvent.
- Heat the reaction mixture to 100°C for 30 hours, then perform filtration and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical elegance. The shift towards using o-nitrobenzaldehyde and allyl aryl ethers as primary feedstocks represents a significant optimization of the raw material basket. These starting materials are commodity chemicals with established global supply chains, reducing the risk of shortages that often plague specialized reagents. By simplifying the synthetic route and eliminating the need for hazardous or expensive activating agents, the overall cost of goods sold (COGS) can be drastically reduced. This economic efficiency is compounded by the simplified workup procedure, which typically involves filtration and standard chromatography, thereby lowering solvent consumption and waste disposal costs associated with complex extractions or crystallizations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts beyond the standard palladium loading, and the use of cheap bases like cesium carbonate, directly lowers reagent costs. Furthermore, the high reaction efficiency and yield minimize material loss, ensuring that every kilogram of input generates maximum output. The avoidance of multi-step protection and deprotection sequences further compresses the manufacturing timeline, leading to substantial cost savings in labor and facility usage without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for any long-term production contract. Since the key raw materials—o-nitrobenzaldehydes and allyl aryl ethers—are widely produced and commercially available from multiple vendors, the supply chain becomes inherently more resilient. This diversification of supply sources mitigates the risk of single-supplier dependency. Additionally, the robustness of the reaction conditions means that minor variations in raw material quality can be accommodated without catastrophic failure, ensuring consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in sealed tube systems that mimic autoclave conditions used in large-scale reactors. The use of acetonitrile, a common industrial solvent, facilitates easy recovery and recycling. Moreover, the atom-economical nature of the carbonylation reaction reduces the generation of stoichiometric waste streams. This aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals while maintaining high throughput capabilities for commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method utilizes inexpensive and readily available starting materials like o-nitrobenzaldehyde and allyl aryl ethers, avoiding complex pre-functionalization. It offers excellent functional group tolerance and operates under relatively mild conditions compared to harsh traditional cyclization methods.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to the use of stable catalysts and simple post-treatment procedures involving filtration and standard column chromatography. The robustness of the reaction conditions supports transition from laboratory to pilot and commercial scales.
Q: What types of substituents are tolerated on the aromatic rings?
A: The methodology demonstrates broad substrate scope, tolerating various substituents including alkyl groups (methyl, propyl, tert-butyl), halogens (fluorine, chlorine), and electron-withdrawing or donating groups (methoxy, trifluoromethyl) on both the aldehyde and ether components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkenyl Quinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed route for the production of valuable quinolinone scaffolds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-alkenyl quinolin-2(1H)-one derivative delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable, cost-effective supply of these critical intermediates.
