Advanced Low-Temperature Synthesis of 2-Methyl-8-Substituent-Quinoline for Commercial Scale-Up
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more efficient synthetic routes that can withstand the rigors of commercial production. A significant breakthrough in this domain is detailed in patent CN114292231A, which introduces a novel preparation method for 2-methyl-8-substituent-quinoline, a critical scaffold in the synthesis of anti-inflammatory agents and benzylamine derivatives. Unlike traditional Skraup-Doebner-Von Miller syntheses that often suffer from harsh conditions and poor selectivity, this invention leverages a specialized dehydrogenation catalyst, specifically yttrium chloride, to achieve aromatization under remarkably mild thermal conditions. This technical advancement not only addresses the chronic issue of tar formation associated with high-temperature quinoline synthesis but also delivers substantial improvements in product purity and overall yield, positioning it as a highly attractive route for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline derivatives has relied heavily on the Skraup reaction or its variations, which typically involve refluxing anilines with glycerol or aldehydes in the presence of strong acids like sulfuric acid at elevated temperatures. These conventional methodologies are plagued by several inherent drawbacks that complicate large-scale manufacturing and compromise economic efficiency. The most significant challenge is the requirement for high reaction temperatures, which not only consumes excessive energy but also promotes uncontrolled side reactions leading to the formation of complex, high-boiling tar-like substances. These polymeric byproducts are notoriously difficult to separate from the desired product, often necessitating elaborate and costly purification steps such as column chromatography or multiple recrystallizations, which drastically reduce the final isolated yield. Furthermore, the slow dehydrogenation kinetics under these traditional conditions often result in incomplete conversion, leaving behind residual dihydro-intermediates that contaminate the final API intermediate, thereby failing to meet the stringent purity specifications required by global regulatory bodies.
The Novel Approach
In stark contrast to these archaic methods, the technology disclosed in patent CN114292231A presents a streamlined, two-stage process that decouples the cyclization and aromatization steps, allowing for precise control over each transformation. The core innovation lies in the second stage, where the 1,2-dihydro-2-methyl-8-substituent-quinoline intermediate is subjected to a dehydrogenation reaction catalyzed by yttrium chloride. This specific catalytic system enables the aromatization to proceed efficiently at a temperature range of merely 53-57°C, a dramatic reduction compared to the vigorous reflux conditions of the past. By operating at these lower temperatures, the generation of thermal degradation products and tar is effectively suppressed, resulting in a much cleaner reaction profile. The visual representation of this superior synthetic pathway highlights the transformation from the substituted aniline precursor through the dihydro-intermediate to the final aromatic quinoline, demonstrating a clear and logical progression that minimizes waste and maximizes atom economy.
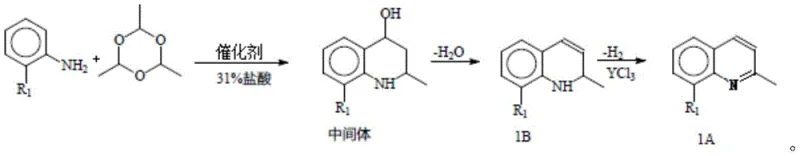
Mechanistic Insights into Yttrium Chloride-Catalyzed Dehydrogenation
The efficacy of this novel process can be attributed to the unique Lewis acidic properties of yttrium chloride (YCl3), which acts as a potent promoter for the removal of hydrogen from the dihydro-quinoline ring system. In the absence of such a catalyst, the dehydrogenation of 1,2-dihydroquinolines typically requires high thermal energy to overcome the activation barrier for aromatization. However, the presence of the yttrium species likely coordinates with the nitrogen atom or the pi-system of the intermediate, weakening the C-H bonds at the 1 and 2 positions and facilitating the elimination of hydrogen. This catalytic assistance lowers the activation energy of the rate-determining step, allowing the reaction to reach completion within a short timeframe of 1 to 3 hours at near-ambient temperatures. The result is a highly selective transformation where the thermodynamic drive towards aromaticity is harnessed without the kinetic penalty of excessive heat, ensuring that the delicate functional groups on the quinoline ring, such as nitro or alkoxy substituents, remain intact and unmodified.
Furthermore, the mechanism extends to the initial cyclization step, which employs a sophisticated dual-catalyst system comprising an oxidant (such as a mixture of potassium iodide and potassium iodate, or hydrogen peroxide) and a phase transfer catalyst. This combination is crucial for managing the heterogeneous nature of the reaction mixture, where the aniline hydrochloride salt must interact with the organic aldehyde (paraldehyde) in a solvent system that may include dichloromethane or toluene. The phase transfer catalyst, selected from options like tetrabutylammonium bromide or crown ethers, shuttles reactive species across the phase boundary, ensuring uniform reaction rates and preventing localized hot spots that could lead to impurity formation. This meticulous control over the reaction environment during the cyclization phase ensures that the dihydro-intermediate is formed with high fidelity, providing a clean substrate for the subsequent yttrium-catalyzed dehydrogenation, ultimately leading to the high purity observed in the final 2-methyl-8-substituent-quinoline product.
How to Synthesize 2-Methyl-8-Substituent-Quinoline Efficiently
Implementing this advanced synthetic route requires careful attention to reagent stoichiometry and temperature control, particularly during the transition from the cyclization phase to the dehydrogenation phase. The process begins with the salification of the substituted aniline, followed by the cyclization with paraldehyde under inert atmosphere to generate the dihydro-intermediate. Once this intermediate is secured, the critical dehydrogenation step is initiated by the addition of the yttrium chloride catalyst. The following guide outlines the standardized operational parameters derived from the patent data to ensure optimal yield and purity, serving as a foundational reference for process chemists aiming to replicate this high-efficiency methodology in a pilot or production setting.
- Salify 2-substituted aniline with hydrochloric acid at 20-55°C to form the hydrochloride salt.
- Perform cyclization with paraldehyde using a phase transfer catalyst and oxidant (KI/KIO3 or H2O2) at 20-25°C to obtain the dihydro-intermediate.
- Conduct dehydrogenation of the intermediate using yttrium chloride catalyst at 53-57°C to yield the final aromatic quinoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this yttrium-catalyzed synthesis offers compelling strategic advantages that extend far beyond simple chemical curiosity. The primary value proposition lies in the drastic simplification of the downstream processing workflow. By effectively eliminating the formation of heavy tars and high-boiling impurities, the need for extensive purification protocols is significantly reduced. This translates directly into shorter batch cycles and higher throughput, allowing manufacturing facilities to produce more material in less time without compromising on quality standards. The ability to operate at lower temperatures also reduces the load on heating utilities and cooling systems, contributing to a leaner operational expenditure profile that enhances the overall competitiveness of the supply chain.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of expensive and hazardous high-temperature operations. Traditional quinoline synthesis often requires specialized equipment capable of withstanding aggressive acidic conditions and high thermal stress, which incurs high capital and maintenance costs. In contrast, the mild conditions of 53-57°C allow for the use of standard glass-lined or stainless steel reactors, reducing equipment depreciation and maintenance overheads. Additionally, the high yield reported in the patent examples, reaching up to 90% in optimized scenarios, means that less raw material is wasted per kilogram of finished product. This improvement in material efficiency directly lowers the cost of goods sold (COGS), providing a buffer against fluctuating raw material prices and enabling more aggressive pricing strategies in the competitive pharmaceutical intermediate market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that are sensitive to minor variations in conditions or that produce inconsistent quality. The robustness of the yttrium chloride catalytic system ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term contracts with multinational pharmaceutical clients. The use of readily available and stable reagents, such as paraldehyde and common phase transfer catalysts, mitigates the risk of supply disruptions associated with exotic or hard-to-source chemicals. Furthermore, the reduced formation of hazardous waste and tar simplifies waste disposal logistics, removing a potential bottleneck in the production schedule and ensuring that delivery timelines are met with greater predictability and reliability.
- Scalability and Environmental Compliance: As the industry moves towards greener manufacturing practices, the environmental footprint of chemical processes is under increasing scrutiny. This novel method aligns perfectly with sustainability goals by minimizing energy consumption through low-temperature operation and reducing the volume of organic waste generated. The suppression of tar formation means less solvent is required for cleaning and purification, lowering the total volume of volatile organic compounds (VOCs) released. This inherent cleanliness makes the process easier to scale from laboratory grams to multi-ton commercial production without encountering the nonlinear scale-up issues often associated with exothermic, high-temperature reactions. Consequently, this technology offers a future-proof solution for the commercial scale-up of complex pharmaceutical intermediates that meets both economic and environmental regulatory standards.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing production lines, we have compiled a set of frequently asked questions based on the specific technical disclosures within the patent documentation. These answers address common concerns regarding catalyst loading, substrate scope, and process safety, providing a clear understanding of the operational realities of this synthesis. Understanding these details is essential for R&D teams planning tech transfer activities and for procurement officers assessing the long-term viability of this supply source.
Q: What is the primary advantage of using Yttrium Chloride in quinoline synthesis?
A: According to patent CN114292231A, Yttrium Chloride allows the dehydrogenation reaction to proceed at significantly lower temperatures (53-57°C) compared to traditional methods, reducing tar formation and improving yield up to 90%.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the process utilizes mild reaction conditions and common solvents like dichloromethane or toluene, avoiding extreme heat and pressure, which facilitates safer and more reliable commercial scale-up for pharmaceutical manufacturing.
Q: What substituents are compatible with this synthetic route?
A: The method is versatile and supports various R1 groups including hydrogen, nitro, methyl, ethyl, propyl, butyl, pentyl, and alkoxy groups, making it suitable for synthesizing a wide range of quinoline derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-8-Substituent-Quinoline Supplier
The technical superiority of the yttrium-catalyzed dehydrogenation route represents a significant opportunity for pharmaceutical companies seeking to secure a stable and high-quality supply of quinoline intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this advanced chemistry for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed in the laboratory are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-methyl-8-substituent-quinoline meets the exacting standards required for API synthesis, providing you with peace of mind and regulatory confidence.
We invite you to explore how this innovative synthesis can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals, ensuring a partnership built on transparency, quality, and mutual success.
