Advanced Palladium-Catalyzed Synthesis of Sulfur-Containing Gamma-Gamma-Diarylamine Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Sulfur-Containing Gamma-Gamma-Diarylamine Compounds for Commercial Scale-Up
The development of efficient synthetic routes for complex nitrogen and sulfur-containing heterocycles remains a critical challenge in modern medicinal chemistry, particularly when dealing with transition metal catalysis where heteroatoms often act as catalyst poisons. Patent CN113861086A introduces a groundbreaking methodology for the synthesis of sulfur-containing gamma,gamma-diarylamine butyrylamide compounds, utilizing a sophisticated N,S-bidentate directed olefin difunctionalization strategy. This technology represents a significant leap forward by successfully employing palladium catalysis in the presence of sulfur atoms, traditionally known to deactivate palladium centers, thereby opening new avenues for constructing valuable pharmacophores. The process leverages unactivated olefins and electron-rich aromatic amines under relatively mild oxidative conditions, achieving high regioselectivity and functional group tolerance without the need for pre-functionalized substrates. For R&D directors and process chemists, this patent offers a robust platform for accessing diverse chemical space that was previously difficult to explore due to catalyst incompatibility issues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the introduction of sulfur and nitrogen functionalities onto aliphatic chains has relied on multi-step sequences involving harsh reagents, protecting group manipulations, and stoichiometric amounts of toxic heavy metals. Conventional approaches often struggle with the inherent reactivity of sulfur towards palladium, leading to rapid catalyst deactivation and poor turnover numbers, which necessitates excessive catalyst loading and drives up production costs significantly. Furthermore, classical methods for gamma,gamma-diarylation typically require pre-activated halides or organometallic reagents that are moisture-sensitive and expensive, creating substantial logistical burdens for supply chain managers. The lack of atom economy in these older routes generates significant waste streams, complicating environmental compliance and increasing the overall carbon footprint of the manufacturing process. Additionally, controlling regioselectivity in the functionalization of unactivated olefins without a strong directing group often results in complex mixtures of isomers, requiring energy-intensive purification steps that reduce overall yield and throughput.
The Novel Approach
In stark contrast, the methodology disclosed in CN113861086A utilizes a clever N,S-bidentate coordination motif that not only prevents catalyst poisoning but actively directs the palladium center to the specific gamma-position of the olefin. This innovative approach allows for the direct coupling of simple, commercially available arylamines with N-(2-(ethylthio)phenyl)but-3-enamide in a single operational step, drastically simplifying the synthetic workflow. By employing a dual oxidant system consisting of ferric chloride and ferrous acetate, the reaction maintains the active Pd(II) species throughout the catalytic cycle, ensuring consistent performance over extended reaction times. The use of benign solvents like acetonitrile and the absence of cryogenic conditions further enhance the practicality of this method for industrial applications. This streamlined process eliminates the need for intermediate isolation, allowing for a telescoped workflow that reduces solvent consumption and labor costs while improving the overall safety profile of the operation.

Mechanistic Insights into N,S-Bidentate Directed Olefin Difunctionalization
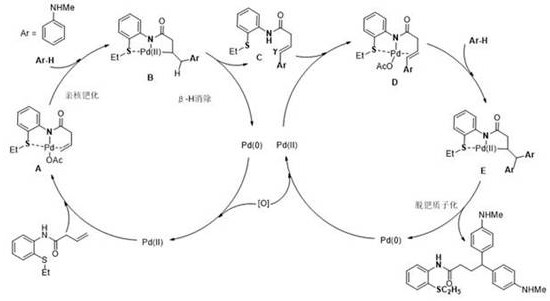
The core innovation of this technology lies in its unique mechanistic pathway, which effectively turns a potential liability—the sulfur atom—into a crucial asset for regiocontrol. The reaction initiates with the coordination of the palladium(II) catalyst to the nitrogen and sulfur atoms of the substrate, forming a stable five-membered palladacycle intermediate that directs subsequent reactivity. This N,S-chelation stabilizes the metal center against reduction and prevents the formation of inactive palladium-sulfur clusters that typically plague sulfur-containing reactions. Following coordination, the nucleophilic arylamine attacks the activated olefin moiety, facilitated by the electron-withdrawing nature of the coordinated palladium, leading to the formation of a key alkyl-palladium species. The presence of benzoic acid as an additive plays a critical role in proton shuttling and maintaining the appropriate acidity to facilitate the nucleophilic attack without decomposing the sensitive catalytic complex.
Subsequent beta-hydride elimination generates a mono-arylated olefin intermediate and a Pd(0) species, which is rapidly re-oxidized to Pd(II) by the iron-based oxidant system to re-enter the catalytic cycle. This redox-neutral balance is essential for sustaining catalytic turnover and achieving the high yields reported in the patent examples. The mono-arylated intermediate then undergoes a second coordination and nucleophilic attack sequence, ultimately delivering the gamma,gamma-diarylamine product with high fidelity. Understanding this detailed catalytic cycle is vital for process optimization, as it highlights the importance of maintaining precise oxidant ratios and temperature control to prevent side reactions such as polymerization or over-oxidation. The mechanistic clarity provided by this patent allows chemists to confidently scale the reaction, knowing exactly which parameters influence the rate-determining steps and product distribution.
How to Synthesize Sulfur-Containing Gamma-Gamma-Diarylamine Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the oxidant system and the purity of the starting olefin substrate to ensure reproducible results. The protocol involves dissolving the N-(2-(ethylthio)phenyl)but-3-enamide and the chosen arylamine nucleophile in dry acetonitrile, followed by the sequential addition of palladium acetate, ferric chloride, ferrous acetate, and benzoic acid. Maintaining the reaction temperature at precisely 90°C is critical, as lower temperatures may result in incomplete conversion while higher temperatures could lead to decomposition of the sensitive amine components. The detailed standardized synthesis steps below outline the specific molar ratios and workup procedures required to achieve the optimal 71% yield demonstrated in the patent examples.
- Combine N-(2-(ethylthio)phenyl)but-3-enamide substrate with arylamine nucleophile, palladium acetate catalyst, and iron-based oxidants in acetonitrile solvent.
- Heat the reaction mixture to 90°C and stir continuously for 24 hours to ensure complete conversion via the N,S-bidentate directed mechanism.
- Filter the reaction mixture, remove solvent via rotary evaporation, and purify the residue using silica gel column chromatography to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by relying on commodity chemicals that are readily available in the global market, reducing dependency on exotic or custom-synthesized starting materials. The ability to use simple arylamines and unactivated olefins means that sourcing risks are minimized, and bulk purchasing power can be leveraged to drive down raw material costs significantly. Furthermore, the simplified workup procedure, which involves basic filtration and solvent removal followed by standard column chromatography, reduces the need for specialized equipment or complex downstream processing infrastructure. This operational simplicity translates directly into lower capital expenditure for manufacturing facilities and reduced training requirements for plant operators, making it an attractive option for contract manufacturing organizations looking to optimize their asset utilization.
- Cost Reduction in Manufacturing: The elimination of expensive pre-functionalized reagents and the use of catalytic rather than stoichiometric amounts of palladium significantly lowers the direct material costs associated with production. By avoiding the need for cryogenic conditions or inert atmosphere gloveboxes, the process also reduces energy consumption and utility costs, contributing to a leaner manufacturing budget. The high atom economy of the difunctionalization reaction ensures that a greater proportion of the input mass ends up in the final product, minimizing waste disposal fees and maximizing resource efficiency. Additionally, the robustness of the catalyst system allows for potential recycling strategies or reduced catalyst loading upon further optimization, offering further opportunities for cost savings in large-scale campaigns.
- Enhanced Supply Chain Reliability: Utilizing widely available feedstocks such as acetonitrile and common arylamines ensures that supply chain disruptions are less likely to impact production schedules compared to routes relying on niche intermediates. The mild reaction conditions and tolerance to various functional groups mean that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply fluctuations. This resilience is crucial for maintaining consistent delivery timelines to pharmaceutical clients who require reliable access to key intermediates for their own drug development pipelines. The scalability of the reaction from gram to kilogram scales without significant modification further enhances supply security, allowing for rapid ramp-up in response to market demand.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods, as it avoids the use of heavy metal stoichiometric oxidants like chromium or manganese salts. The primary byproducts are iron salts, which are easier to handle and dispose of in accordance with environmental regulations, reducing the regulatory burden on the manufacturing site. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, supports green chemistry initiatives and helps companies meet their sustainability goals. This environmental compatibility not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, derived directly from the experimental data and scope defined in the intellectual property documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this technology for integration into their existing portfolios. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: How does this method overcome sulfur poisoning of palladium catalysts?
A: The patent utilizes an N,S-bidentate directing group strategy where the sulfur atom coordinates with palladium rather than poisoning it, facilitating a stable catalytic cycle for olefin difunctionalization.
Q: What are the optimal reaction conditions for maximum yield?
A: Optimal results are achieved using palladium acetate (20 mol%) with ferric chloride and ferrous acetate as co-oxidants in acetonitrile at 90°C for 24 hours, yielding up to 71%.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process avoids complex intermediate separations and uses commercially available raw materials like olefins and arylamines, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-Containing Gamma-Gamma-Diarylamine Butyrylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed technology for accelerating the discovery and development of novel therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of sulfur-containing intermediates meets the highest quality standards required by global regulatory agencies. We understand the complexities of managing sulfur chemistry and have developed specialized protocols to handle these materials safely and effectively, minimizing cross-contamination risks and ensuring product integrity.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can be tailored to your specific project needs, offering a Customized Cost-Saving Analysis that highlights the economic benefits of switching to this more efficient methodology. Please contact our technical procurement team today to request specific COA data for related analogues and comprehensive route feasibility assessments for your target molecules. By partnering with us, you gain access to a wealth of chemical expertise and manufacturing capacity dedicated to bringing your most challenging projects to market faster and more cost-effectively.
