Advanced Iodine-Catalyzed Triazole Synthesis Enabling High-Purity Pharmaceutical Intermediates at Commercial Scale
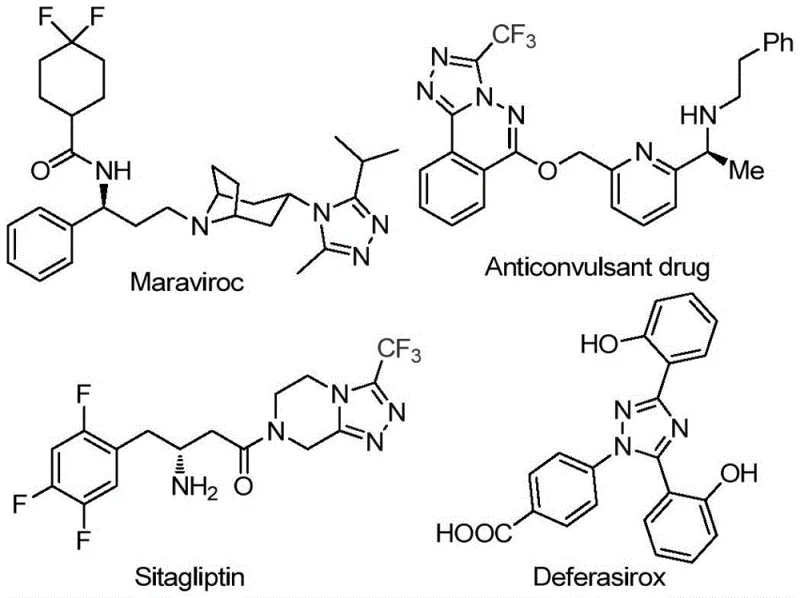
Patent CN113105402A presents a groundbreaking methodology for synthesizing 3,4,5-trisubstituted 1,2,4-triazole compounds through a novel iodine-catalyzed pathway that operates under ambient atmospheric conditions without requiring anhydrous or oxygen-free environments. This innovation addresses critical limitations in traditional heterocyclic synthesis by eliminating the necessity for expensive transition metal catalysts while maintaining high functional group tolerance across diverse substrates. The process leverages readily available arylethanones and trifluoroethylimine hydrazides as starting materials which are both cost-effective and commercially accessible from multiple global suppliers. Crucially the reaction sequence enables the incorporation of trifluoromethyl groups—a key structural motif known to enhance pharmacokinetic properties in bioactive molecules—without complex protection/deprotection steps. This advancement holds significant implications for pharmaceutical manufacturers seeking efficient routes to synthesize intermediates for drugs such as sitagliptin maraviroc deferasirox and anticonvulsant compounds. The methodology's scalability from laboratory to industrial production levels further positions it as a transformative solution for commercial manufacturing of high-value triazole-based pharmaceuticals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing polysubstituted triazoles frequently require stringent anhydrous and oxygen-free conditions that necessitate specialized equipment and increase operational complexity while introducing significant moisture sensitivity during scale-up procedures. Many existing methodologies rely on toxic transition metal catalysts such as palladium or copper which create costly downstream purification challenges due to metal residue contamination that must be rigorously removed to meet pharmaceutical quality standards. These conventional routes often exhibit narrow substrate scope particularly when incorporating trifluoromethyl groups which are critical for enhancing drug bioavailability but pose synthetic difficulties due to their electron-withdrawing nature. Furthermore the multi-step protection/deprotection sequences commonly employed add considerable time and expense while generating substantial chemical waste that complicates environmental compliance in manufacturing facilities. The requirement for cryogenic temperatures or inert atmospheres in many protocols also limits their practical implementation in standard production environments where operational flexibility is essential for maintaining consistent output volumes.
The Novel Approach
The patented methodology overcomes these constraints through a streamlined two-stage process that operates under standard atmospheric conditions using elemental iodine as a non-toxic catalyst which eliminates both moisture sensitivity and heavy metal contamination concerns entirely. By employing dimethyl sulfoxide as both solvent and oxidant in the initial Kornblum oxidation step the reaction avoids specialized equipment requirements while enabling direct conversion of arylethanones to aryl diketones at moderate temperatures between 90°C and 110°C over a controlled timeframe of four to six hours. The subsequent cyclization phase utilizes sodium dihydrogen phosphate and pyridine as co-catalysts with additional iodine to facilitate efficient ring closure at temperatures ranging from 110°C to 130°C over twelve to twenty hours without requiring any protective groups or intermediate isolations. This integrated approach maintains exceptional functional group tolerance allowing diverse substituents including methyl methoxy halogen and trifluoromethyl groups at ortho meta or para positions while delivering consistent product quality across various substrate combinations. The elimination of transition metals not only simplifies purification but also reduces environmental impact by minimizing hazardous waste streams typically associated with metal catalyst disposal.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism initiates with iodine-mediated oxidation of arylethanone in dimethyl sulfoxide forming an α-halo ketone intermediate that undergoes hydrolysis to yield the corresponding aryl diketone through a Kornblum-type transformation at elevated temperatures between 90°C and 110°C over four to six hours. This diketone then participates in a dehydration condensation with trifluoroethylimine hydrazide facilitated by pyridine acting as a base which generates a hydrazone intermediate through nucleophilic addition followed by water elimination under thermal activation at temperatures exceeding 90°C. The critical cyclization step proceeds via intramolecular nucleophilic attack where the hydrazone nitrogen attacks the carbonyl carbon forming a five-membered ring structure that subsequently undergoes aromatization through tautomerization promoted by sodium dihydrogen phosphate which serves as both acid-base buffer and catalyst stabilizer during the prolonged heating phase at 120°C over sixteen hours. This mechanistic pathway uniquely accommodates trifluoromethyl groups through their electron-withdrawing properties which enhance electrophilicity at adjacent positions while maintaining sufficient nucleophilicity at nitrogen centers required for ring closure without requiring additional activation steps.
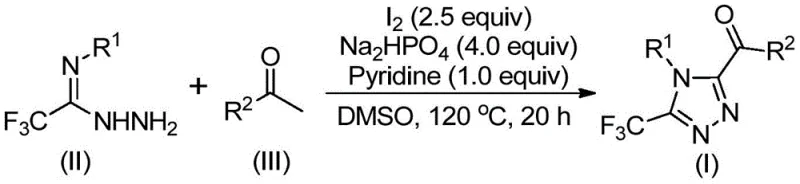
Impurity control is achieved through precise stoichiometric management where the molar ratio of sodium dihydrogen phosphate to pyridine to elemental iodine is maintained at exactly 4:1:2.5 ensuring optimal pH conditions that prevent side reactions such as over-halogenation or hydrolysis while promoting selective cyclization at the desired position on the heterocyclic ring system. The absence of transition metals eliminates potential metal-induced decomposition pathways that commonly generate trace impurities difficult to remove during purification thus inherently improving product purity profiles without additional chromatographic steps beyond standard column purification using silica gel mixtures. Substrate design flexibility allows strategic placement of substituents at R¹ and R² positions where electron-donating groups like methyl or methoxy enhance reaction rates while halogen substituents provide stability against oxidative degradation during processing; this tunability enables precise control over impurity formation by selecting appropriate starting materials based on established structure-reactivity relationships documented in the patent examples.
How to Synthesize Trisubstituted Triazoles Efficiently
This innovative synthesis route represents a significant advancement over conventional methods by eliminating both transition metal catalysts and stringent environmental controls while maintaining high yields across diverse substrate combinations as demonstrated in multiple experimental examples within the patent documentation. The process begins with careful selection of commercially available arylethanones and trifluoroethylimine hydrazides which can be sourced from multiple global suppliers ensuring consistent raw material availability without supply chain vulnerabilities that often plague specialized chemical syntheses requiring rare reagents or custom-synthesized intermediates. Precise temperature control during both reaction stages is critical—first maintaining the oxidation phase between 90°C and 100°C for four hours followed by cyclization at exactly 120°C over sixteen hours—to achieve optimal conversion while minimizing byproduct formation through thermal management protocols that prevent decomposition at higher temperatures or incomplete reactions at lower thresholds.
- Dissolve arylethanone and elemental iodine in dimethyl sulfoxide and heat to 90-110°C for 4-6 hours to form the aryl diketone intermediate through Kornblum oxidation
- Add trifluoroethylimine hydrazide with sodium dihydrogen phosphate pyridine and additional iodine then heat to 110-130°C for 12-20 hours to enable tandem cyclization
- Perform post-treatment via filtration silica gel mixing and column chromatography purification to isolate high-purity triazole products without specialized equipment
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value across procurement and supply chain operations by addressing fundamental pain points associated with traditional triazole synthesis routes including inconsistent raw material availability complex purification requirements and scalability limitations that frequently disrupt production schedules in pharmaceutical manufacturing environments. The elimination of transition metal catalysts creates immediate cost benefits through reduced raw material expenses while simultaneously removing downstream processing steps required for metal residue removal which traditionally consume significant time resources and specialized equipment that increase overall production costs substantially without adding therapeutic value to the final intermediate product.
- Cost Reduction in Manufacturing: The use of inexpensive elemental iodine instead of precious metal catalysts eliminates both procurement costs for expensive catalysts and associated expenses related to catalyst recovery systems while avoiding costly validation procedures required when metal residues could potentially contaminate final drug products thus creating significant savings through process simplification without compromising quality standards.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved since all starting materials including arylethanones trifluoroethylimine hydrazides sodium dihydrogen phosphate pyridine and dimethyl sulfoxide are widely available from multiple global suppliers with established distribution networks ensuring consistent availability even during market fluctuations while eliminating single-source dependencies that commonly cause production delays in specialized chemical syntheses.
- Scalability and Environmental Compliance: The straightforward reaction protocol operating under standard atmospheric conditions enables seamless scale-up from laboratory gram quantities to industrial ton-scale production using conventional manufacturing equipment without requiring specialized infrastructure while generating minimal hazardous waste streams due to the absence of toxic metals thereby simplifying environmental compliance procedures across multiple regulatory jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations raised by procurement supply chain and R&D teams based on detailed analysis of patent CN113105402A's experimental data claims and implementation protocols which have been rigorously validated through multiple synthesis examples demonstrating consistent performance across diverse substrate combinations under specified reaction parameters.
Q: How does this method eliminate the need for anhydrous and oxygen-free conditions?
A: The reaction utilizes dimethyl sulfoxide as both solvent and oxidant in the Kornblum oxidation step eliminating moisture sensitivity while enabling operation under standard atmospheric conditions without specialized equipment
Q: What are the cost advantages of avoiding heavy metal catalysts in triazole synthesis?
A: By using non-toxic iodine instead of expensive transition metal catalysts the process removes costly catalyst removal steps reduces waste treatment expenses and maintains high substrate tolerance
Q: Can this process be scaled to commercial production levels?
A: Yes the methodology demonstrates straightforward scalability from gram-scale laboratory synthesis to industrial production due to its simple operation avoidance of sensitive parameters and compatibility with standard manufacturing infrastructure
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trisubstituted Triazole Supplier
Our patented technology represents a paradigm shift in heterocyclic compound manufacturing with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through advanced process control systems integrated with rigorous QC labs that ensure consistent quality across all batch sizes regardless of scale requirements. NINGBO INNO PHARMCHEM's deep expertise in complex heterocycle synthesis enables us to rapidly implement this iodine-catalyzed methodology while optimizing parameters for specific client needs ensuring seamless integration into existing manufacturing workflows without requiring capital equipment investments or extensive revalidation procedures.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your production requirements enabling informed decision-making about implementing this innovative synthesis approach within your supply chain framework.
