Advanced Copper-Catalyzed Synthesis of N-Substituted Maleimide Intermediates for Commercial Pharmaceutical Manufacturing
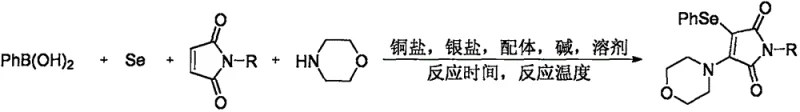
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those exhibiting potent biological activities. A recent technological breakthrough documented in patent CN111978236A introduces a highly innovative preparation method for N-substituted-3-morpholinyl-4-phenylseleno maleimide compounds. This technology represents a significant leap forward in organic synthesis, utilizing a transition metal copper-catalyzed four-component tandem reaction under oxygen conditions. By leveraging cheap and readily available starting materials such as phenylboronic acid, selenium powder, morpholine, and N-substituted maleimides, this method overcomes many historical limitations associated with selenylation reactions. The strategic integration of a silver salt, specific ligands, and a strong base creates a synergistic catalytic environment that ensures high product yields and exceptional purity. For R&D directors and procurement specialists alike, this development signals a new era of accessibility for these valuable intermediates, which serve as core skeletons in marine natural alkaloids and antitumor active molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3,4-difunctionalized maleimides has been fraught with synthetic challenges that hindered their widespread application in drug discovery pipelines. Conventional strategies often relied on the use of precious metal catalysts, such as Ruthenium, which not only escalated the raw material costs but also introduced significant complications regarding heavy metal residue removal in final API products. Furthermore, traditional selenylation protocols frequently necessitated the pre-preparation of diaryl diselenides, adding extra synthetic steps that reduced overall atom economy and increased waste generation. These multi-step sequences inherently amplified the risk of yield loss at each stage and extended the lead time required to produce sufficient quantities for biological testing. Additionally, the harsh reaction conditions often associated with older methodologies limited the functional group tolerance, thereby restricting the structural diversity accessible to medicinal chemists. Such inefficiencies created a bottleneck in the supply chain, making it difficult for manufacturers to secure reliable sources of high-purity maleimide derivatives at a competitive price point.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data revolutionizes this landscape by employing a direct, one-pot four-component cross-coupling strategy. This approach eliminates the need for pre-functionalized selenium reagents by utilizing elemental selenium powder directly, which is both economically advantageous and operationally simpler. The use of a copper catalytic system, specifically optimized with cuprous iodide, offers a cost-effective alternative to noble metals while maintaining high catalytic efficiency. The reaction proceeds smoothly under an oxygen atmosphere, utilizing molecular oxygen as a green oxidant, which aligns with modern principles of sustainable chemistry and environmental compliance. By merging the introduction of the arylselenyl group and the amine functionality in a single operational step, this method drastically reduces the number of unit operations required. This consolidation of synthetic steps not only accelerates the timeline from bench to kilogram scale but also minimizes solvent consumption and waste disposal costs, presenting a compelling value proposition for commercial manufacturing.
Mechanistic Insights into Cu-Catalyzed Oxidative Aminoarylselenation
The success of this transformation hinges on the intricate interplay between the copper catalyst, the silver salt oxidant, and the nitrogen-based ligand. Mechanistically, the cuprous iodide acts as the central activator, likely facilitating the oxidative addition of the selenium species and coordinating the boron reagent for transmetallation. The presence of silver carbonate serves a dual purpose: it acts as an oxidant to regenerate the active copper species and potentially assists in the activation of the selenium powder. The ligand, specifically 1,10-phenanthroline, stabilizes the copper center and modulates its electronic properties to favor the desired cross-coupling pathway over homocoupling side reactions. Experimental data indicates that the precise stoichiometry of these components is critical; deviations from the optimized ratios result in negligible conversion, highlighting the sensitivity and sophistication of this catalytic cycle. Understanding this mechanism allows process chemists to fine-tune reaction parameters for maximum efficiency when scaling up to industrial reactors.
Impurity control is another critical aspect where this mechanistic understanding provides significant value. The high selectivity observed in this reaction minimizes the formation of by-products such as homocoupled diselenides or over-oxidized sulfone derivatives. The use of cesium carbonate as a base ensures complete deprotonation of the intermediate species without promoting hydrolysis of the sensitive maleimide ring. This high level of chemoselectivity translates directly to simplified downstream processing, as the crude reaction mixture contains fewer impurities that require rigorous chromatographic separation. For quality control teams, this means a more consistent impurity profile across different batches, which is essential for regulatory filings and maintaining stringent purity specifications. The ability to predict and control the impurity spectrum based on the catalytic mechanism gives manufacturers a distinct advantage in ensuring product consistency and reliability.
How to Synthesize N-Substituted-3-Morpholinyl-4-Phenylseleno Maleimide Efficiently
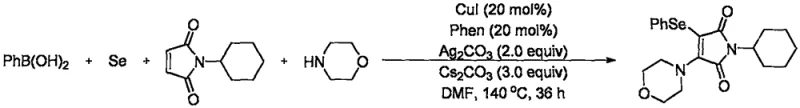
Implementing this synthesis route requires careful attention to the specific reaction parameters outlined in the technical documentation to ensure reproducibility and high yield. The process begins with the precise weighing of phenylboronic acid, selenium powder, morpholine, and the specific N-substituted maleimide substrate in a molar ratio of 3:3:3:1. These reagents are suspended in N,N-dimethylformamide (DMF), which has been identified as the optimal solvent for solubilizing the inorganic salts and facilitating the homogeneous catalytic cycle. The addition of the catalytic system, comprising cuprous iodide, 1,10-phenanthroline, silver carbonate, and cesium carbonate, must be conducted under controlled conditions to prevent premature oxidation or moisture ingress. Once the reaction mixture is assembled, it is subjected to an oxygen atmosphere and heated to 140°C for a duration of approximately 36 hours. Following the completion of the reaction, standard workup procedures involving ethyl acetate extraction and brine washing are employed to isolate the crude product, which is then purified via column chromatography.
- Prepare the reaction mixture by combining phenylboronic acid, selenium powder, morpholine, and N-substituted maleimide in a molar ratio of 3: 3:3:1 within an organic solvent system.
- Introduce the synergistic catalytic system comprising cuprous iodide (20 mol%), silver carbonate (2.0 equiv), 1,10-phenanthroline ligand (20 mol%), and cesium carbonate base (3.0 equiv).
- Maintain the reaction at 140°C under an oxygen atmosphere for 36 hours, followed by standard aqueous workup and column chromatography purification to isolate the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented technology offers substantial opportunities for cost optimization and supply chain resilience. The shift from precious metal catalysts to abundant copper salts fundamentally alters the cost structure of the manufacturing process, removing the volatility associated with rhodium or ruthenium pricing. Furthermore, the utilization of elemental selenium and phenylboronic acid as starting materials leverages commodity chemical markets, ensuring a stable and continuous supply of raw materials even during global disruptions. The simplification of the synthetic route from multi-step sequences to a single-pot operation significantly reduces labor costs, energy consumption, and equipment occupancy time. These efficiencies collectively contribute to a lower cost of goods sold, allowing procurement managers to negotiate more competitive pricing structures with suppliers while maintaining healthy margins. The robustness of the reaction conditions also implies a lower risk of batch failure, enhancing the overall reliability of the supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive pre-functionalized selenium reagents and precious metal catalysts drives a significant decrease in direct material costs. By avoiding the separate synthesis of diaryl diselenides, manufacturers save on the reagents, solvents, and labor associated with that precursor step. The use of copper iodide instead of ruthenium complexes further reduces the catalyst cost burden, which is particularly impactful when producing at multi-ton scales. Additionally, the high yield reported in the patent examples minimizes the loss of valuable starting materials, ensuring that a greater proportion of input mass is converted into sellable product. These factors combine to create a leaner manufacturing process that is inherently more cost-effective than legacy technologies.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is streamlined due to the use of widely available commodity chemicals like selenium powder and phenylboronic acid. Unlike specialized reagents that may have single-source suppliers or long lead times, these inputs can be procured from multiple vendors globally, mitigating the risk of supply shortages. The robustness of the reaction against minor variations in conditions also means that production schedules are less likely to be disrupted by technical issues. This stability allows supply chain heads to plan inventory levels with greater confidence and reduce the need for safety stock buffers. Consequently, the overall agility of the supply chain is improved, enabling faster response times to market demands.
- Scalability and Environmental Compliance: The process demonstrates excellent potential for scale-up due to its reliance on standard heating and stirring equipment without the need for extreme pressures or cryogenic temperatures. The use of molecular oxygen as an oxidant generates water as the primary byproduct, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing site. Simplified workup procedures mean less solvent waste is generated per kilogram of product, lowering disposal costs and regulatory compliance burdens. This environmental compatibility is increasingly important for meeting corporate sustainability goals and adhering to strict international environmental regulations. The combination of scalability and eco-friendliness makes this technology a future-proof choice for long-term production partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the detailed experimental data and comparative analysis provided in the patent documentation. Understanding these aspects is crucial for technical teams evaluating the feasibility of adopting this route for their specific project needs. The answers reflect the empirical findings regarding catalyst specificity, reagent availability, and process robustness.
Q: Why is Cuprous Iodide critical for this synthesis compared to other copper salts?
A: According to patent data CN111978236A, experimental comparisons demonstrated that alternative copper catalysts such as copper acetate, cuprous chloride, and cupric bromide failed to yield any target product. Cuprous iodide was identified as the unique effective catalyst essential for activating the selenium species and facilitating the cross-coupling cycle.
Q: What are the primary advantages of this method over traditional Ruthenium-catalyzed routes?
A: Traditional methods often rely on expensive Ruthenium catalysts and pre-synthesized diaryl diselenides, which increase raw material costs and synthetic steps. This novel approach utilizes inexpensive elemental selenium powder and phenylboronic acid directly, significantly simplifying the supply chain and reducing the overall cost of goods sold.
Q: Is this four-component reaction suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available solvents like DMF and standard inorganic bases. The workup involves conventional extraction and chromatography techniques, indicating strong potential for scalability without requiring specialized high-pressure equipment or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted-3-Morpholinyl-4-Phenylseleno Maleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis technology for the global pharmaceutical industry. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative method to fruition. Our state-of-the-art facilities are equipped to handle the specific thermal and atmospheric conditions necessary for this reaction, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and dedicated process development teams, we guarantee the consistent quality and reliability that top-tier drug manufacturers demand. We are committed to leveraging this advanced chemistry to provide our clients with a competitive edge in their drug development programs.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs and timeline constraints. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to cutting-edge synthetic methodologies backed by a supply chain built for performance and reliability.
