Revolutionizing Maleimide Synthesis: A Cost-Effective Four-Component Coupling Strategy for Pharmaceutical Intermediates
The pharmaceutical industry's relentless pursuit of potent kinase inhibitors has placed maleimide derivatives at the forefront of medicinal chemistry research. Patent CN109651225B introduces a groundbreaking methodology for synthesizing 1-methyl-3-morpholinyl-4-arylmercaptomaleimide compounds, addressing critical bottlenecks in traditional synthetic routes. This innovation leverages a transition metal copper-catalyzed relay reaction that simultaneously constructs carbon-sulfur and carbon-nitrogen bonds on the maleimide scaffold. By utilizing inexpensive and readily available starting materials such as iodobenzene, elemental sulfur powder, N-methylmaleimide, and morpholine, this protocol offers a robust alternative to legacy methods. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-value pharmaceutical intermediates while adhering to stricter environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-amino-4-arylmercaptomaleimides has been plagued by reliance on hazardous and costly precursors. As illustrated in earlier literature, such as the work by Dubinina et al., traditional pathways often necessitate the use of 3,4-dichloromaleimide, a specialized and expensive building block that significantly inflates raw material costs. Furthermore, these conventional routes typically require thiophenols as sulfur sources, which are notorious for their overpowering, unpleasant odors and high toxicity, posing severe challenges for industrial hygiene and waste management. The reliance on such malodorous reagents not only complicates reactor design and containment but also increases the operational expenditure related to scrubbing and environmental compliance. Additionally, the multi-step nature of accessing these functionalized maleimides often leads to cumulative yield losses and complex purification burdens, making the overall process economically unviable for large-scale commercial manufacturing.
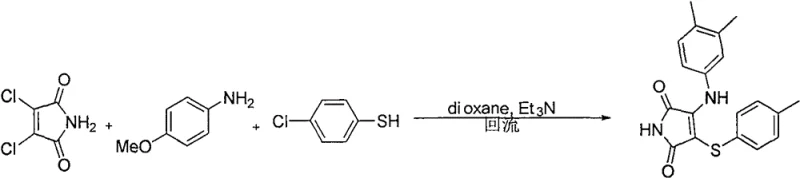
The Novel Approach
In stark contrast, the methodology disclosed in CN109651225B revolutionizes the construction of the maleimide core by employing a direct four-component coupling strategy. This novel approach bypasses the need for pre-functionalized dichloromaleimides, instead utilizing simple N-methylmaleimide as the central scaffold. The introduction of elemental sulfur powder as the sulfur source is a masterstroke in green chemistry, eliminating the handling risks associated with liquid thiophenols while drastically reducing material costs. The reaction proceeds through an oxidative coupling mechanism where the carbon-carbon double bond of the maleimide serves as the acceptor for both the aryl group from iodobenzene and the sulfur moiety. This convergent synthesis not only shortens the synthetic timeline but also enhances atom economy, providing a streamlined pathway that is inherently more suitable for the reliable supply of high-purity pharmaceutical intermediates.
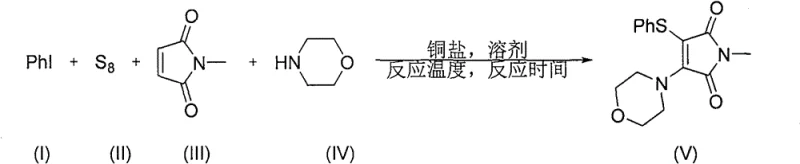
Mechanistic Insights into Copper-Catalyzed Oxidative Thioamination
The success of this transformation hinges on the precise orchestration of a copper-catalyzed relay reaction under an oxygen atmosphere. Mechanistically, the process likely initiates with the oxidative addition of the aryl iodide to the copper center, generating an active aryl-copper species. Concurrently, the activation of elemental sulfur (S8) by the copper catalyst facilitates the insertion of sulfur into the reaction manifold, potentially forming a copper-sulfur intermediate. This species then engages with the electron-deficient double bond of the N-methylmaleimide. The presence of morpholine as a nucleophile allows for the subsequent amination step, completing the functionalization of the maleimide ring. The use of molecular oxygen as the terminal oxidant is crucial, as it regenerates the active copper catalyst and drives the thermodynamic equilibrium forward, ensuring high conversion rates without the need for stoichiometric oxidants that generate excessive waste.
From an impurity control perspective, the specificity of the CuI catalyst plays a pivotal role in minimizing side reactions. Experimental data within the patent indicates that the choice of copper salt is non-trivial; while cuprous iodide affords a respectable 57% yield, switching to cupric chloride or bromide results in complete reaction failure. This suggests that the iodide ligand is essential for stabilizing the key organometallic intermediates or facilitating the specific oxidative addition step required for aryl iodide activation. Furthermore, the solvent system, specifically N,N-dimethylformamide (DMF), provides the necessary polarity to solubilize the inorganic sulfur and stabilize the charged transition states. This rigorous optimization ensures that the final product profile is clean, reducing the burden on downstream purification processes and ensuring that the resulting intermediate meets the stringent purity specifications required for API synthesis.
How to Synthesize 1-Methyl-3-Morpholinyl-4-Phenylmercaptomaleimide Efficiently
To implement this synthesis effectively, operators must adhere to the optimized conditions defined in the patent examples, particularly regarding the catalyst loading and atmospheric control. The reaction requires a precise molar ratio of substrates, typically employing an excess of iodobenzene and sulfur to drive the equilibrium towards the desired tetra-substituted product. Maintaining an oxygen-rich environment is critical for the catalytic cycle turnover, and the temperature must be carefully controlled around 120°C to balance reaction kinetics with thermal stability. Detailed standardized operating procedures regarding reagent addition sequences and workup protocols are essential for reproducibility.
- Combine iodobenzene, elemental sulfur, N-methylmaleimide, and morpholine in DMF solvent with CuI catalyst under oxygen atmosphere.
- Heat the reaction mixture to 120°C and stir for 24 hours to facilitate the oxidative coupling and sulfur insertion.
- Perform workup via ethyl acetate extraction, brine washing, and purification using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this four-component coupling technology offers profound strategic advantages beyond mere technical novelty. The substitution of specialized, high-cost starting materials with commodity chemicals fundamentally alters the cost structure of the manufacturing process. By eliminating the dependency on 3,4-dichloromaleimide and malodorous thiophenols, manufacturers can secure a more stable and predictable supply chain that is less susceptible to market volatility associated with niche fine chemicals. This transition not only simplifies logistics but also aligns with global trends towards greener, safer chemical manufacturing, thereby reducing regulatory friction and potential liability.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive dichloromaleimides with bulk N-methylmaleimide and elemental sulfur cannot be overstated. Elemental sulfur is one of the most abundant and inexpensive chemical feedstocks available globally, offering a drastic reduction in raw material expenditure compared to functionalized thiol reagents. Furthermore, the elimination of thiophenols removes the need for specialized odor-control infrastructure and expensive personal protective equipment, leading to substantial operational cost savings. The simplified workflow reduces the number of unit operations required, thereby lowering energy consumption and labor costs associated with multi-step syntheses.
- Enhanced Supply Chain Reliability: Sourcing iodobenzene and morpholine is significantly more straightforward than procuring specialized chlorinated maleimides, which often have limited supplier bases. This diversification of raw material sources mitigates the risk of supply disruptions and ensures continuous production capability. The robustness of the reaction conditions, utilizing common solvents like DMF and standard heating equipment, means that this process can be easily transferred between different manufacturing sites or contract manufacturing organizations without requiring bespoke reactor modifications. This flexibility is crucial for maintaining supply continuity in the face of geopolitical or logistical challenges.
- Scalability and Environmental Compliance: The use of solid sulfur powder and the avoidance of volatile, toxic thiols greatly improve the safety profile of the process, making it inherently safer for scale-up from pilot plant to commercial tonnage. The reduced generation of hazardous waste streams simplifies effluent treatment and lowers the environmental footprint of the facility. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies focused on ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed maleimide synthesis. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on catalyst selection, solvent effects, and process scalability for potential licensees and manufacturing partners.
Q: Why is elemental sulfur preferred over thiophenols in this maleimide synthesis?
A: Elemental sulfur (S8) is significantly cheaper, less toxic, and avoids the severe odor and environmental hazards associated with handling volatile thiophenols, leading to safer industrial operations.
Q: What is the critical catalyst for this four-component coupling reaction?
A: Cuprous iodide (CuI) is identified as the optimal catalyst, providing a 57% yield, whereas other copper salts like CuCl2 or CuBr2 result in no reaction, highlighting the specific mechanistic requirement for iodide ligands.
Q: Can this process be scaled for commercial production of kinase inhibitors?
A: Yes, the use of readily available commodity chemicals like iodobenzene and sulfur powder, combined with standard DMF solvent systems, makes this route highly amenable to kilogram-to-ton scale-up for API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Methyl-3-Morpholinyl-4-Arylmercapto Maleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed four-component coupling technology for the production of next-generation kinase inhibitors. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative coupling reactions, including rigorous oxygen control and high-temperature processing, while our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this efficient synthetic route for your pipeline projects. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this methodology can optimize your COGS. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the development of high-value maleimide-based therapeutics.
