Advanced One-Pot Synthesis of 2-Aminobenzofuran Compounds for Pharmaceutical Manufacturing
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and environmentally benign synthetic routes for bioactive scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN109851599B, which discloses a novel preparation method for 2-aminobenzofuran compounds. These structures are privileged motifs found in numerous natural products and pharmaceutical agents, exhibiting a broad spectrum of biological activities including antibacterial, analgesic, antifungal, and antiviral properties. The patented methodology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot transformation utilizing hypervalent iodine chemistry. By leveraging the unique reactivity of 1-[(triisopropyl)silyl]ethynyl]-1,2-benziodoxol-3(1H)-one (often referred to as TIPS-EBX), this process achieves high atom economy and operational simplicity. For R&D directors and procurement specialists in the fine chemical sector, this innovation offers a compelling alternative for sourcing high-purity pharmaceutical intermediates, promising reduced processing times and enhanced supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
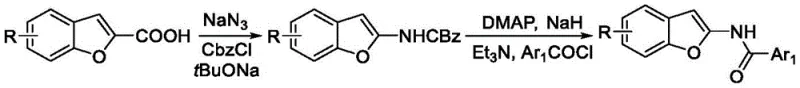
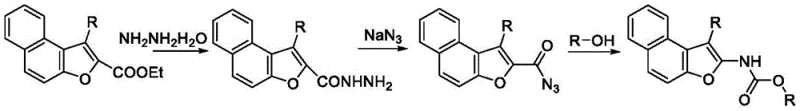
Historically, the construction of the 2-aminobenzofuran core has been fraught with synthetic challenges that hinder large-scale manufacturing efficiency. Prior art, such as the methods developed by the Saku project group in 2010, relied on ammoniation and coupling reactions starting from o-hydroxybenzaldehyde. While effective on a small laboratory scale, these routes often suffer from poor atom economy and require harsh reaction conditions that generate significant environmental pollution. Furthermore, alternative strategies reported by the Hema topic group in 2013 involved a cumbersome four-step reaction sequence starting from 3-aminonaphthofuran-2-carboxylic acid ethyl ester. Such multi-step processes inherently accumulate impurities at each stage, necessitating rigorous purification between steps which drastically lowers the overall yield and increases production costs. The reliance on complex starting materials and the generation of stoichiometric waste make these conventional pathways less attractive for modern, green chemistry-compliant manufacturing environments.
The Novel Approach
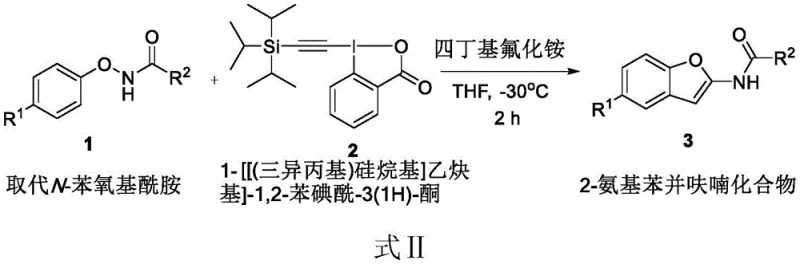
In stark contrast to the laborious historical precedents, the method disclosed in CN109851599B introduces a direct and highly efficient cyclization strategy. This novel approach utilizes substituted N-phenoxyamides as the primary substrates, reacting them directly with the hypervalent iodine reagent TIPS-EBX in the presence of tetrabutylammonium fluoride (TBAF). The reaction proceeds smoothly in tetrahydrofuran (THF) at a controlled low temperature of -30°C, completing within just 2 hours. This single-step transformation constructs the furan ring and installs the amino functionality simultaneously, effectively collapsing what was previously a multi-day synthesis into a matter of hours. The simplicity of the work-up procedure, involving merely concentration and silica gel column chromatography, underscores the practical utility of this method for generating diverse libraries of 2-aminobenzofuran derivatives with various substituents on the aromatic ring.
Mechanistic Insights into TIPS-EBX Mediated Cyclization
The success of this synthesis hinges on the unique reactivity profile of the TIPS-EBX reagent, a stable hypervalent iodine(III) species capable of transferring an ethynyl group. In the presence of a fluoride source like TBAF, the silicon-protecting group on the alkyne is cleaved, generating a reactive iodine-acetylide intermediate in situ. This species acts as a potent electrophile towards the oxygen atom of the N-phenoxyamide substrate. The subsequent intramolecular nucleophilic attack by the amide nitrogen onto the activated alkyne triggers a cascade cyclization event. This mechanism avoids the need for transition metal catalysts, such as copper or palladium, which are often required in traditional Sonogashira-type couplings. The absence of heavy metals is a critical advantage for pharmaceutical manufacturing, as it eliminates the need for expensive and time-consuming metal scavenging steps to meet stringent regulatory limits on residual metals in API intermediates.
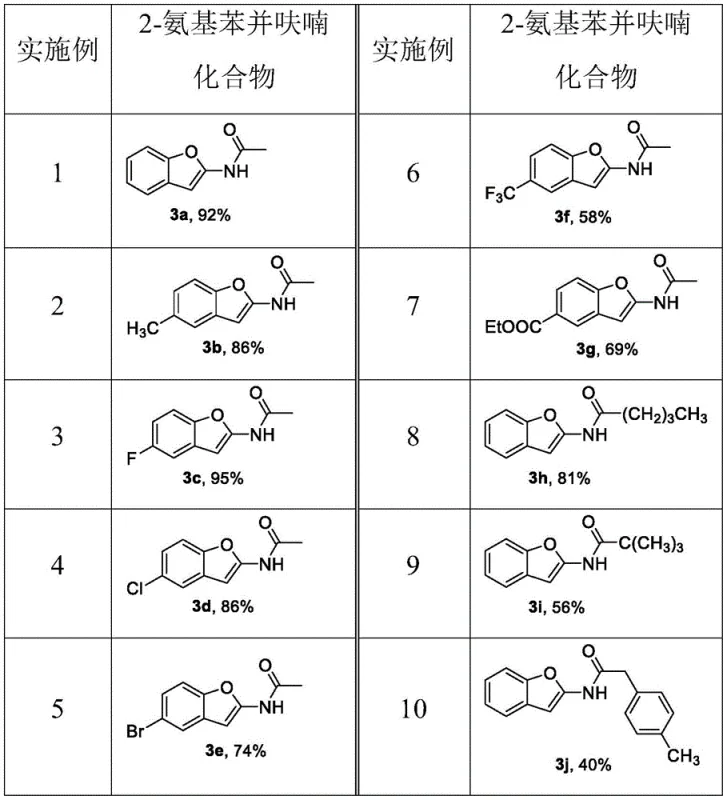
Furthermore, the reaction exhibits remarkable tolerance to a wide range of functional groups, allowing for the introduction of diverse substituents at the R1 position of the benzene ring. As demonstrated in the patent examples, electron-donating groups like methyl, as well as electron-withdrawing groups such as fluorine, chlorine, bromine, trifluoromethyl, and even ester functionalities, are well-tolerated under the reaction conditions. The mechanism ensures that the integrity of these sensitive functional groups is maintained throughout the cyclization process. This chemoselectivity is vital for R&D teams aiming to explore structure-activity relationships (SAR) without needing to employ orthogonal protection strategies. The high yields observed across different substrates, ranging from 40% to 95%, indicate a robust and reliable catalytic cycle that minimizes side reactions and byproduct formation, thereby simplifying the downstream purification burden.
How to Synthesize 2-Aminobenzofuran Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise control over reaction parameters to maximize yield and purity. The protocol dictates a specific molar ratio of 1:1.3:1.3 for the N-phenoxyamide, TIPS-EBX, and TBAF respectively, ensuring complete consumption of the starting material while minimizing excess reagent waste. The low temperature of -30°C is critical to suppress potential decomposition of the hypervalent iodine species and to control the exothermicity of the cyclization. Following the reaction, the isolation of the product is straightforward, relying on standard evaporation techniques followed by chromatographic separation. For detailed operational parameters and safety considerations regarding the handling of hypervalent iodine reagents, please refer to the standardized synthesis guide below.
- Charge a reactor with substituted N-phenoxyamide, 1-[(triisopropyl)silyl]ethynyl]-1,2-benziodoxol-3(1H)-one (TIPS-EBX), and tetrabutylammonium fluoride in a 1: 1.3:1.3 molar ratio.
- Add tetrahydrofuran (THF) as the solvent and maintain the reaction mixture at a strict low temperature of -30°C while stirring for 2 hours.
- Concentrate the reaction mixture using a rotary evaporator to obtain the crude product, followed by purification via silica gel column chromatography to isolate the target 2-aminobenzofuran.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this TIPS-EBX mediated synthesis offers substantial strategic advantages for procurement managers and supply chain heads. The primary benefit lies in the drastic simplification of the manufacturing process. By reducing a multi-step sequence to a single pot operation, manufacturers can significantly reduce the total processing time and labor costs associated with production. This efficiency gain translates directly into cost reduction in pharmaceutical intermediate manufacturing, allowing for more competitive pricing structures without compromising on quality. Additionally, the elimination of transition metal catalysts removes a major bottleneck in the supply chain related to the sourcing and disposal of precious metals, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis inherently lowers operational expenditures. Traditional methods often require multiple isolation and purification steps, each consuming solvents, energy, and manpower. By consolidating the synthesis into a single reaction vessel with a simple work-up, the overall consumption of resources is minimized. Moreover, the high atom economy of the TIPS-EBX reagent ensures that a greater proportion of the raw materials end up in the final product, reducing waste disposal costs. Although specific percentage savings depend on local utility costs, the qualitative reduction in unit operations provides a clear pathway to lower cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, specifically substituted N-phenoxyamides and TIPS-EBX, are becoming increasingly accessible through global chemical suppliers. Unlike specialized catalysts that may have long lead times or single-source dependencies, the key components of this reaction are supported by a robust supply network. This diversity in sourcing options mitigates the risk of supply disruptions, ensuring consistent availability of the 2-aminobenzofuran intermediates. For supply chain planners, this reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturing.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not involve extreme pressures or temperatures, making the process highly amenable to scale-up from gram to kilogram scales. The use of THF as a solvent is standard in the industry, and the waste stream is relatively clean compared to heavy metal-catalyzed processes. This aligns well with modern environmental, health, and safety (EHS) standards, facilitating easier regulatory approval for commercial production facilities. The ability to scale complex heterocyclic syntheses without generating hazardous heavy metal waste is a significant compliance advantage for manufacturers operating in strictly regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of using TIPS-EBX for benzofuran synthesis compared to traditional methods?
A: The use of TIPS-EBX (1-[(triisopropyl)silyl]ethynyl]-1,2-benziodoxol-3(1H)-one) allows for a direct, one-pot cyclization that bypasses the multi-step sequences required in conventional routes. This significantly improves atom economy and reduces the generation of hazardous waste associated with older coupling strategies.
Q: What is the typical yield and purity profile for this synthesis route?
A: Experimental data from the patent indicates excellent yields, with unsubstituted analogues achieving up to 92% isolated yield. The process utilizes standard silica gel chromatography for purification, ensuring high purity suitable for downstream pharmaceutical applications without requiring complex recrystallization protocols.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the reaction operates under mild conditions (-30°C in THF) and uses commercially available reagents. The simplicity of the work-up procedure (concentration followed by chromatography) suggests strong potential for scale-up, provided that low-temperature control is maintained effectively in larger reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobenzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has extensively evaluated the TIPS-EBX mediated cyclization method and confirmed its potential for delivering high-purity 2-aminobenzofuran compounds. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the identity and purity of every batch, guaranteeing consistency and reliability for your drug development programs.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of novel derivatives or bulk supply of standard intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and accelerate your path to market.
