Optimized Synthetic Route for Imperatorin Derivatives Enhancing Commercial Viability
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the production of bioactive furocoumarins, specifically imperatorin derivatives, due to their profound therapeutic potential ranging from antitumor to anti-inflammatory activities. The patent CN101805350B discloses a groundbreaking synthetic approach that addresses the longstanding inefficiencies associated with traditional linear furocoumarin synthesis. By leveraging a strategic combination of Pechmann condensation and a novel Lewis acid-mediated dealkylation sequence, this technology offers a pathway to high-purity intermediates with exceptional step economy. For research and development directors focusing on impurity profiles and process robustness, this method represents a significant leap forward, eliminating the need for cumbersome functional group protection strategies that often plague legacy syntheses. The ability to access these complex molecular architectures through a streamlined five-step sequence not only accelerates timeline-to-market but also ensures a cleaner crude profile, thereby reducing the burden on downstream purification units.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of imperatorin derivatives has been hindered by convoluted reaction pathways that necessitate multiple protection and deprotection steps to manage the reactivity of phenolic hydroxyl groups. These conventional routes often suffer from low overall yields due to material loss at each sequential transformation, alongside the generation of significant chemical waste that complicates environmental compliance. The reliance on harsh reagents for deprotection can lead to the degradation of the sensitive furan ring system, resulting in difficult-to-remove impurities that compromise the quality of the final active pharmaceutical ingredient. Furthermore, the extended reaction times and stringent conditions required for these older methods drive up operational expenditures, making the final product less competitive in a cost-sensitive global market. For procurement managers, these inefficiencies translate into volatile pricing and unreliable supply chains, as manufacturers struggle to maintain consistent output amidst complex processing requirements.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a highly efficient route that bypasses traditional bottlenecks through the innovative use of an acetonyl side chain as a temporary masking group. This strategy allows for the direct construction of the coumarin core followed by a precise cyclization step using sodium hydroxide and isopropanol, which facilitates the formation of the furan ring under relatively mild conditions. The subsequent dealkylation utilizing a Lewis acid catalyst is particularly noteworthy, as it enables the selective removal of the acetonyl group to reveal the critical phenolic functionality without damaging the molecular scaffold. This streamlined approach not only enhances the total yield but also simplifies the workup procedures, leading to a more sustainable and economically viable manufacturing process. 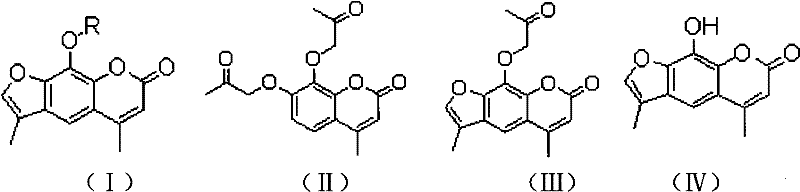
Mechanistic Insights into Lewis Acid-Mediated Dealkylation
The core innovation of this synthetic pathway lies in the mechanistic elegance of the dealkylation step, where a Lewis acid is employed to cleave the ether linkage of the acetonyl side chain. This reaction proceeds through a coordination complex that activates the ether oxygen, facilitating the departure of the alkyl group under thermal conditions that are significantly milder than those required for strong acid hydrolysis. For technical teams evaluating process safety and scalability, this mechanism offers a distinct advantage by minimizing the risk of exothermic runaways and reducing the corrosion load on reactor equipment. The selectivity of this transformation ensures that the methyl groups at the 4 and 6 positions remain intact, preserving the structural integrity required for the biological activity of the final imperatorin derivative. Understanding this mechanistic nuance is crucial for scaling the process, as it dictates the precise stoichiometry and temperature control needed to maximize conversion while suppressing side reactions.
Furthermore, the initial cyclization step involving sodium hydroxide and isopropanol demonstrates a sophisticated control over regioselectivity, ensuring that the furan ring closes exclusively at the desired position on the coumarin nucleus. The use of isopropanol as a co-solvent plays a pivotal role in solubilizing the intermediates while maintaining the basicity required for the intramolecular nucleophilic attack. This specific solvent system helps to stabilize the transition state, thereby enhancing the rate of reaction and improving the purity of the resulting acetonyl-imperatorin intermediate. From a quality control perspective, this high degree of regiocontrol minimizes the formation of isomeric byproducts, which simplifies the chromatographic purification steps and leads to a final product with a superior impurity profile. Such mechanistic precision is essential for meeting the stringent specifications demanded by regulatory bodies for pharmaceutical intermediates.
How to Synthesize Imperatorin Derivative Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the Lewis acid dealkylation and the final Williamson etherification steps. The process begins with the preparation of the coumarin precursor, followed by the critical cyclization and dealkylation sequence that defines the novelty of this patent. Operators must ensure strict moisture control during the Lewis acid addition to prevent catalyst deactivation, and the subsequent heating under vacuum must be monitored closely to achieve complete conversion without thermal degradation. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature profiles optimized for reproducibility.
- Execute Pechmann reaction using pyrogallol and methyl acetoacetate to form 7,8-dihydroxyl-4-methylcoumarin.
- Perform Williamson etherification to introduce the acetonyl side chain, followed by base-catalyzed cyclization in sodium hydroxide and isopropanol.
- Conduct Lewis acid-mediated dealkylation to obtain 4,6-dimethyl xanthoxylin, followed by final Williamson etherification to yield the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply chain resilience, this synthetic route offers compelling advantages derived directly from its chemical efficiency and raw material selection. The reduction in the number of synthetic steps inherently lowers the consumption of solvents and reagents, which translates into a significant decrease in variable manufacturing costs without compromising on product quality. By utilizing commodity chemicals such as pyrogallol and methyl acetoacetate as starting materials, the process mitigates the risk of supply disruptions associated with exotic or proprietary reagents, ensuring a more stable and predictable procurement landscape. Additionally, the milder reaction conditions reduce the energy footprint of the manufacturing process, aligning with modern sustainability goals and potentially lowering utility expenses for large-scale production facilities.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection sequences drastically reduces the consumption of auxiliary chemicals and the labor hours required for monitoring complex reactions. This step economy directly correlates to a lower cost of goods sold, allowing for more competitive pricing strategies in the global market for pharmaceutical intermediates. Furthermore, the high overall yield reported in the patent examples suggests that less raw material is wasted per kilogram of final product, enhancing the atom economy of the entire process. These factors combine to create a robust economic model that can withstand fluctuations in raw material pricing while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that production schedules are not held hostage by the lead times of specialized reagents. This accessibility allows for greater flexibility in sourcing and inventory management, reducing the risk of stockouts that can delay downstream drug development programs. The robustness of the reaction conditions also means that the process can be transferred between manufacturing sites with minimal re-validation, providing supply chain leaders with the option to diversify production geography. Such flexibility is invaluable in maintaining continuity of supply for critical API intermediates in a volatile global market.
- Scalability and Environmental Compliance: The process design inherently supports scale-up, as the reaction exotherms are manageable and the workup procedures involve standard filtration and extraction techniques familiar to plant operators. The reduction in hazardous waste generation, owing to the shorter synthetic route and higher selectivity, simplifies waste treatment protocols and reduces the environmental compliance burden. This alignment with green chemistry principles not only improves the corporate sustainability profile but also minimizes the regulatory hurdles associated with expanding production capacity. Consequently, this technology offers a clear path from laboratory bench to commercial tonnage with reduced friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the specific data and claims found within the patent documentation. These insights are designed to clarify the operational benefits and feasibility of adopting this route for commercial manufacturing of imperatorin derivatives. Understanding these details is essential for making informed decisions about process adoption and partnership opportunities.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method significantly shortens the synthetic route to five steps, eliminating complex protection and deprotection sequences, which results in higher overall yields and reduced operational costs.
Q: How does the Lewis acid dealkylation step improve process safety?
A: The use of Lewis acid for dealkylation allows for milder reaction conditions compared to traditional harsh hydrolysis, reducing energy consumption and minimizing the formation of hazardous byproducts.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials like pyrogallol and operates under manageable temperature and pressure conditions, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imperatorin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial supplies for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis method are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of imperatorin derivative meets the exacting standards required for drug substance manufacturing. Our commitment to technical excellence ensures that the complex chemistry involved in the Lewis acid dealkylation and cyclization steps is managed with precision and consistency.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this more efficient methodology. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to enhance your supply chain efficiency and accelerate your development timelines with our high-quality chemical solutions.
