Palladium-Catalyzed Synthesis of Indoline-2-ketone-3-acetamide: Scalable Production for Pharmaceutical Applications
This technical commercial insight report analyzes Chinese Patent CN115028569A, which discloses a novel palladium-catalyzed synthesis method for indoline-2-ketone-3-acetamide series compounds, a class of nitrogen-containing heterocyclic derivatives with significant applications in pharmaceutical development. The patented approach represents a substantial advancement in the field of heterocyclic chemistry by eliminating the need for carbon monoxide and amine compounds in the synthesis process, thereby addressing critical safety and cost challenges faced by traditional manufacturing methods. This innovative methodology enables the efficient construction of complex indoline scaffolds that are prevalent in numerous bioactive molecules and drug candidates, offering pharmaceutical manufacturers a safer, more economical route to these valuable intermediates. The process utilizes readily available nitro compounds as nitrogen sources instead of expensive aniline derivatives, while employing metal carbonyl complexes as safe carbonyl sources, thus significantly reducing both operational hazards and raw material costs without compromising product quality or yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing indolinone derivatives typically require indole or indoline precursors as reaction substrates, which are often expensive and difficult to obtain commercially, creating significant supply chain vulnerabilities for pharmaceutical manufacturers. These conventional approaches frequently employ high-pressure carbon monoxide in multi-component reactions, necessitating specialized equipment with stringent safety protocols that substantially increase capital investment and operational complexity for production facilities. The substrate scope in existing methodologies is generally limited to relatively simple structures, resulting in poor structural diversity and inadequate selectivity that restricts the range of accessible compounds for drug discovery and development programs. Furthermore, many established routes rely on amine-based starting materials that require additional synthetic steps for preparation, such as catalytic hydrogenation of nitro compounds, which adds both time and cost to the overall manufacturing process while generating additional waste streams that complicate environmental compliance efforts.
The Novel Approach
The patented methodology overcomes these limitations by utilizing o-halogenated aryl acrylamides and nitro compounds as starting materials, which are commercially available at significantly lower costs compared to traditional indole-based precursors and amine derivatives. By employing metal carbonyl complexes such as molybdenum hexacarbonyl as safe carbonyl sources instead of gaseous carbon monoxide, the process eliminates the need for high-pressure reaction equipment while maintaining excellent reaction efficiency and selectivity under mild conditions of 80-120°C for 12-36 hours. This innovative approach demonstrates remarkable substrate tolerance across a wide range of functional groups including halogens, alkyl, alkoxy, ester, nitro, and cyano substituents, enabling the synthesis of structurally diverse indolinone derivatives with high purity and yield without requiring inert atmosphere protection during the reaction process. The simplified reaction setup and elimination of hazardous reagents translate to reduced capital expenditure for manufacturing facilities while improving operational safety profiles and decreasing overall production costs through streamlined processing and reduced waste generation.
Mechanistic Insights into Palladium-Catalyzed Carbonyl Insertion and Tandem Cyclization
The catalytic cycle begins with oxidative addition of the palladium(0) catalyst into the carbon-halogen bond of the o-halogenated aryl acrylamide substrate, forming an aryl-palladium(II) intermediate that subsequently coordinates with the metal carbonyl complex to facilitate carbonyl insertion. This key step generates an acyl-palladium species that undergoes intramolecular nucleophilic attack by the amide nitrogen, triggering cyclization to form the indolinone core structure with simultaneous reduction of the nitro group to an amine functionality through a reductive amination process. The organic carboxylic acid additive plays a crucial role in promoting the nitro group reduction while maintaining optimal reaction kinetics and selectivity throughout the transformation sequence.
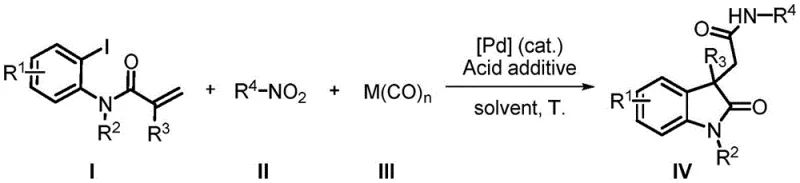
The structural versatility of the resulting indoline-2-ketone-3-acetamide compounds is demonstrated by the general formula IV, where R1, R2, and R3 can independently represent various electron-donating or electron-withdrawing groups including alkyl, alkoxy, halogens, esters, nitro groups, and heterocyclic moieties, while R4 encompasses diverse alkyl, cycloalkyl, alkenyl, alkynyl, aromatic, and heteroaromatic substituents.
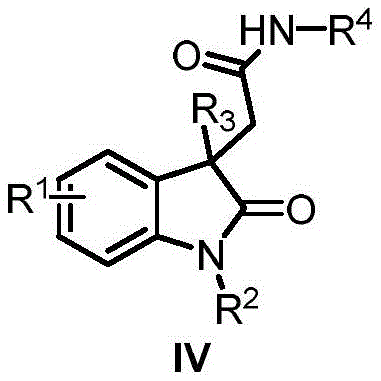
The high chemoselectivity observed in this transformation stems from the precise control of reaction parameters that prevent undesired side reactions such as homocoupling or hydrodehalogenation, while the mild reaction conditions preserve sensitive functional groups throughout the synthesis. The absence of amine compounds in the reaction mixture eliminates potential issues with overalkylation or imine formation that commonly plague traditional synthetic routes, resulting in cleaner reaction profiles with fewer impurities that require removal during purification. This inherent selectivity translates directly to higher product purity without the need for extensive chromatographic separation or additional purification steps, which is particularly advantageous for large-scale manufacturing where process efficiency and yield are critical economic factors.
How to Synthesize Indoline-2-ketone-3-acetamide Efficiently
This patented methodology provides a robust and scalable approach to synthesizing indoline-2-ketone-3-acetamide derivatives through a palladium-catalyzed carbonyl insertion and tandem cyclization process that eliminates traditional limitations associated with carbon monoxide usage and amine-based starting materials. The process demonstrates exceptional functional group tolerance across diverse substrates while maintaining high yields under operationally simple conditions that do not require inert atmosphere protection or specialized high-pressure equipment. Detailed standardized synthesis procedures for specific compound variants are provided in the following step-by-step guide to facilitate seamless implementation in pharmaceutical manufacturing environments.
- Prepare reaction mixture by combining o-halogenated aryl acrylamide substrate (1 equivalent), nitro compound (1 equivalent), metal carbonyl complex (e.g., molybdenum hexacarbonyl), palladium catalyst (e.g., palladium acetate), organic carboxylic acid additive, and solvent (e.g., tetrahydrofuran or dioxane) under air atmosphere.
- Heat reaction mixture to 80-120°C in a pressure-resistant vessel for 12-36 hours with stirring until complete conversion is confirmed by TLC analysis.
- Perform post-reaction processing by filtering through silica gel to remove catalyst residues followed by column chromatography purification to isolate pure indoline-based product with yields typically ranging from 40% to 90% depending on specific substrate combinations.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology addresses critical pain points in pharmaceutical intermediate procurement by offering a more economical and reliable production route for indoline-based compounds that are essential building blocks in numerous drug development programs. The elimination of hazardous carbon monoxide usage reduces both capital investment requirements for specialized equipment and ongoing operational safety costs while improving facility flexibility for multi-product manufacturing environments.
- Cost Reduction in Manufacturing: The substitution of expensive aniline derivatives with readily available nitro compounds as nitrogen sources significantly reduces raw material costs while eliminating the need for additional catalytic hydrogenation steps required to prepare amine precursors from nitro compounds in traditional syntheses. The use of solid metal carbonyl complexes instead of gaseous carbon monoxide removes requirements for high-pressure reaction vessels and associated safety infrastructure, substantially lowering capital expenditure while simplifying facility design and maintenance requirements for manufacturing operations.
- Enhanced Supply Chain Reliability: The broad substrate scope and functional group tolerance enable reliable production of diverse indolinone derivatives from commercially available starting materials with minimal supply chain constraints. The simplified reaction setup that operates effectively under air atmosphere eliminates dependency on specialized inert gas handling systems, reducing potential points of failure in production processes while improving overall manufacturing resilience against supply chain disruptions.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production scales without requiring significant process re-engineering, with successful demonstration from 100 kgs to 100 MT/annual commercial production using standard glassware or stainless steel reactors. The elimination of hazardous reagents and high-pressure operations substantially reduces environmental impact while simplifying waste stream management and regulatory compliance documentation required for pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding the implementation of this patented synthesis methodology for indoline-based pharmaceutical intermediates, with answers derived directly from the technical specifications and experimental data provided in Chinese Patent CN115028569A.
Q: How does this method eliminate the need for carbon monoxide while maintaining efficient carbonylation?
A: The patented process utilizes solid metal carbonyl complexes such as molybdenum hexacarbonyl or dicobalt octacarbonyl as stable carbonyl sources that release CO in situ under reaction conditions without requiring external pressurized CO gas handling systems.
Q: Why is using nitro compounds more advantageous than traditional aniline-based approaches?
A: Nitro compounds are significantly more cost-effective than aniline derivatives and eliminate the need for additional catalytic hydrogenation steps required to prepare amine precursors from nitro compounds in conventional syntheses.
Q: What makes this process suitable for large-scale pharmaceutical manufacturing?
A: The method operates effectively under air atmosphere without inert gas protection requirements, uses standard glassware or stainless steel reactors without specialized high-pressure equipment, demonstrates excellent scalability from laboratory to commercial production scales, and produces high-purity products with minimal purification needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indoline-2-ketone-3-acetamide Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation. As a leading CDMO specializing in challenging heterocyclic syntheses, we have successfully implemented this patented methodology to produce high-purity indoline-based intermediates for multiple global pharmaceutical clients with consistent quality and reliable delivery performance that meets the most demanding regulatory requirements.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target compounds through our Customized Cost-Saving Analysis service, which will provide detailed insights into how this innovative synthesis approach can optimize your supply chain economics while ensuring uninterrupted access to these critical pharmaceutical building blocks.
