Revolutionizing Cross-Coupling Chemistry: Scalable Indole-Based Phosphine Ligand for Pharmaceutical Intermediate Synthesis
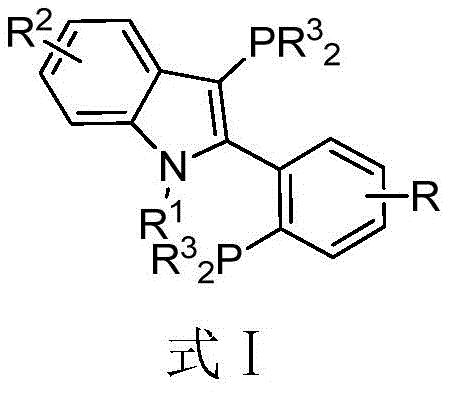
The Chinese patent CN107445989B introduces a groundbreaking class of phosphine ligands featuring an indole skeleton with dual phosphine substitution patterns, representing a significant advancement in transition metal catalysis for complex molecule synthesis. This innovation addresses critical limitations in conventional cross-coupling methodologies by providing a structurally versatile ligand platform that enhances catalytic efficiency while maintaining operational simplicity. The patent specifically details a 3-(disubstituted phosphino)-2-(2-(disubstituted phosphino) substituted phenyl)-1-alkyl-indole skeleton that forms exceptionally stable complexes with palladium and other transition metals. This structural architecture enables unprecedented control over reaction parameters in carbon-carbon and carbon-heteroatom bond formations, particularly demonstrating remarkable efficacy in challenging carbon-sulfur coupling reactions. The technology represents a strategic breakthrough for pharmaceutical manufacturers seeking more efficient routes to complex intermediates while maintaining stringent quality requirements essential for drug substance production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional phosphine ligands used in palladium-catalyzed cross-coupling reactions often suffer from significant limitations including air sensitivity, narrow substrate scope, and requirement for high catalyst loadings that increase production costs and complicate purification processes. Many established ligands such as tri-tert-butylphosphine or Buchwald-type biaryl phosphines demonstrate excellent performance in specific reaction types but lack versatility across different coupling methodologies, forcing manufacturers to maintain multiple specialized catalyst systems. The structural rigidity of conventional ligands frequently results in suboptimal performance when applied to challenging substrates like heteroaromatic systems or sterically hindered partners, leading to inconsistent yields and extended reaction times that disrupt production scheduling. Furthermore, the complex multi-step syntheses required for many advanced ligands create supply chain vulnerabilities and cost inefficiencies that become particularly problematic at commercial scale where consistent quality and availability are paramount for uninterrupted manufacturing operations.
The Novel Approach
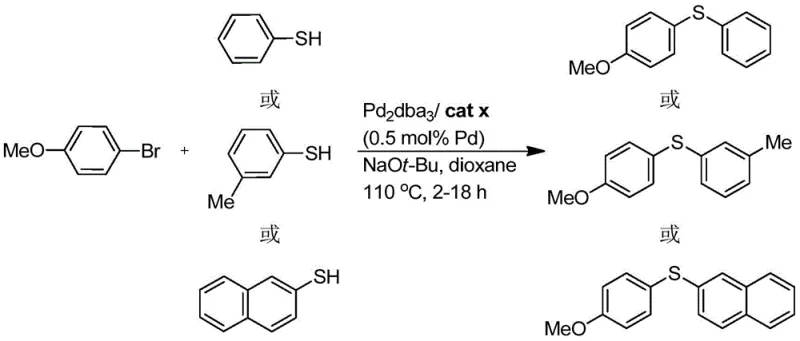
The patented indole-skeleton phosphine ligand overcomes these limitations through its unique molecular architecture that combines structural flexibility with exceptional stability characteristics. The dual phosphine substitution pattern on the indole framework creates an optimal electronic and steric environment that enhances metal coordination while maintaining sufficient lability for catalytic turnover. This design enables remarkably low catalyst loadings of just 0.5 mol% in carbon-sulfur bond formation reactions while achieving high yields previously unattainable with conventional systems. The ligand's inherent stability to air and moisture eliminates the need for specialized handling procedures that complicate manufacturing operations and increase production costs. Most significantly, the straightforward synthetic route from commercially available starting materials using standard organic transformations ensures reliable supply continuity and scalability from laboratory to commercial production volumes without requiring specialized equipment or hazardous reagents that would compromise process safety or environmental compliance.
Mechanistic Insights into Indole-Skeleton Phosphine Ligand Catalysis
The exceptional catalytic performance of this indole-based ligand stems from its unique coordination geometry that facilitates optimal transition state stabilization during cross-coupling reactions. The nitrogen atom in the indole ring participates in secondary coordination with the palladium center, creating a pseudo-chelating effect that stabilizes the active catalytic species while maintaining sufficient lability for substrate association and product dissociation. This dual coordination mode—through both phosphorus atoms and the indole nitrogen—creates an electronic environment that promotes oxidative addition while facilitating reductive elimination steps critical to coupling efficiency. The substituent groups on the phosphorus atoms (R3) can be systematically varied to fine-tune steric bulk and electronic properties, allowing precise optimization for specific reaction types without requiring complete ligand redesign. This molecular tunability represents a significant advantage over conventional monodentate phosphines that lack such sophisticated control over catalytic parameters.
Impurity control is significantly enhanced through the ligand's ability to promote cleaner reaction profiles with minimal side product formation. The well-defined coordination sphere created by the indole scaffold suppresses common side reactions such as homocoupling or protodehalogenation that typically complicate purification of cross-coupled products. The mild reaction conditions enabled by this catalytic system (often operating at or near room temperature) further reduce thermal degradation pathways that generate impurities difficult to remove during downstream processing. The ligand's stability profile also eliminates concerns about ligand decomposition contributing to metal contamination in final products—a critical consideration for pharmaceutical applications where strict metal residue limits must be met. This combination of factors results in cleaner reaction profiles that simplify purification processes and improve overall process efficiency while maintaining the high purity standards required for pharmaceutical intermediates.
How to Synthesize Indole-Based Phosphine Ligands Efficiently
This patented synthesis route represents a significant advancement in ligand manufacturing methodology by combining operational simplicity with exceptional scalability potential for commercial production environments. The three-stage process begins with Fischer indole synthesis using readily available starting materials, proceeds through selective bromination at the critical 3-position, and concludes with dual phosphination under controlled conditions to install the key functional groups responsible for the ligand's superior performance characteristics. Each step has been optimized for high yield and minimal byproduct formation, ensuring efficient material utilization and simplified purification requirements that translate directly to cost advantages at scale. The following standardized procedure provides a reliable pathway for manufacturing this advanced ligand system while maintaining the quality attributes essential for consistent catalytic performance across diverse applications.
- Fischer indole synthesis using 2'-bromoacetophenone and N-alkyl phenylhydrazine with phosphoric acid catalyst followed by polyphosphoric acid treatment at 80-120°C
- Bromination at the 3-position using N-bromosuccinimide in dimethylformamide at room temperature to form the dibromo intermediate
- Phosphination through lithiation at -78°C followed by reaction with disubstituted phosphine chloride to introduce dual phosphine groups
Commercial Advantages for Procurement and Supply Chain Teams
This innovative ligand technology delivers substantial value across procurement and supply chain functions by addressing multiple pain points inherent in traditional catalyst systems used for complex molecule synthesis. The simplified manufacturing process reduces dependency on specialized raw materials and complex synthesis routes that often create supply vulnerabilities in global chemical markets. By enabling more efficient catalytic transformations with lower metal loadings, this technology directly contributes to reduced material consumption and waste generation throughout the production process. The enhanced stability profile eliminates costly handling requirements while improving shelf life and reducing the risk of material degradation during transportation and storage—factors that significantly impact total cost of ownership for specialty chemical inputs.
- Cost Reduction in Manufacturing: The elimination of complex purification steps required for air-sensitive ligands reduces operational complexity and associated labor costs while minimizing solvent consumption during manufacturing. The ability to achieve high yields with minimal catalyst loading directly reduces raw material expenses without compromising product quality or process reliability, creating substantial cost savings through process intensification rather than component cost reduction alone.
- Enhanced Supply Chain Reliability: Sourcing from readily available starting materials through a straightforward synthetic route ensures consistent supply continuity even during market volatility, eliminating dependency on specialized intermediates with limited supplier options. The ligand's stability profile enables standard shipping and storage conditions without requiring specialized handling protocols that often create logistical bottlenecks in global supply chains.
- Scalability and Environmental Compliance: The manufacturing process demonstrates excellent scalability from laboratory to commercial production volumes without requiring specialized equipment modifications or hazardous reagents that would complicate regulatory compliance. Reduced metal loading requirements minimize heavy metal waste streams while simplified reaction profiles decrease overall solvent usage, contributing significantly to environmental sustainability goals without compromising production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented ligand technology in industrial manufacturing environments. These answers are derived directly from the technical specifications and performance data documented in patent CN107445989B, providing factual guidance for technical evaluation and implementation planning without speculative projections or unsubstantiated claims.
Q: How does the indole-skeleton phosphine ligand improve catalytic efficiency compared to conventional ligands?
A: The indole-based ligand forms stable complexes with palladium through its dual phosphine groups and nitrogen coordination site, enabling lower catalyst loading (down to 0.5 mol%) while maintaining high yields in carbon-sulfur bond formation reactions.
Q: What are the key advantages of this ligand system for pharmaceutical intermediate production?
A: The ligand demonstrates exceptional air and moisture stability, simplifies purification processes, and enables high-yield transformations under mild conditions, significantly reducing production complexity for sensitive pharmaceutical intermediates.
Q: How does this technology address supply chain challenges in specialty chemical manufacturing?
A: The straightforward synthesis from commercially available starting materials using standard reactions (Fischer indole, alkylation, bromination) ensures reliable supply continuity and scalability from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-Based Phosphine Ligand Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex specialty chemicals while maintaining stringent purity specifications through rigorous QC labs and advanced analytical capabilities. As a CDMO expert specializing in challenging catalytic transformations, we have successfully implemented this patented ligand technology across multiple client projects requiring high-purity intermediates for pharmaceutical applications where consistent quality and reliable supply are non-negotiable requirements. Our manufacturing infrastructure is specifically designed to handle air-sensitive compounds while ensuring complete traceability throughout the production process, providing clients with confidence in both product quality and regulatory compliance.
We invite you to request our Customized Cost-Saving Analysis which details how this technology can optimize your specific manufacturing process while reducing total cost of ownership. Contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your production requirements, enabling informed decision-making based on actual technical capabilities rather than theoretical projections.
