Scalable Batch Synthesis of Dihydrogibberellin for Advanced Plant Growth Regulation
Scalable Batch Synthesis of Dihydrogibberellin for Advanced Plant Growth Regulation
The agricultural chemical industry is constantly seeking more efficient and environmentally benign methods for producing plant growth regulators, and the recent technological advancements detailed in patent CN107353264B represent a significant leap forward in this domain. This patent discloses a robust batch synthesis method for dihydrogibberellin, a potent and degradable plant growth regulator known for its ability to control crop height and prevent lodging without the persistence issues associated with older chemicals. By utilizing commercially available gibberellic acid as a starting material, the inventors have engineered a pathway that drastically improves upon the yield and safety profiles of previously documented synthetic routes. The process is characterized by its operation under very mild conditions, often at room temperature, which stands in stark contrast to the harsh thermal requirements of traditional methods. For procurement managers and supply chain directors, this translates to a reliable agrochemical intermediate supplier capability that is both cost-effective and scalable, ensuring a steady flow of high-quality materials for formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of dihydrogibberellin was plagued by significant inefficiencies that hindered its widespread commercial adoption. Literature-reported methods typically involved lengthy synthetic sequences comprising at least seven distinct reaction steps, each introducing potential points of failure and material loss. The cumulative yield of these traditional routes was dismally low, often capping at merely 30%, which made large-scale production economically unviable due to the excessive consumption of raw materials and solvents. Furthermore, the repeatability of these older methods was notoriously poor; for instance, the zinc chloride-catalyzed carboxyl protection steps described in earlier studies were difficult to reproduce consistently in a manufacturing setting. Perhaps most critically, the conventional processes relied on severe and dangerous reaction conditions, such as high-temperature reflux for demethylation and mesylate elimination, which not only posed safety risks but also led to substantial decomposition of the sensitive gibberellin skeleton. The purification of the final product was equally challenging, often requiring strong bases that generated numerous impurity peaks, thereby complicating the isolation of the target molecule and increasing downstream processing costs.
The Novel Approach
The methodology presented in the patent offers a transformative solution by streamlining the synthesis into a highly efficient sequence that maximizes atom economy and operational safety. The core of this novel approach lies in the strategic use of protecting groups and mild catalytic systems that preserve the integrity of the complex diterpenoid structure. Starting with cheap and accessible gibberellic acid, the process employs acetic anhydride to protect hydroxyl groups at the C3 and C13 positions, followed by a specialized benzylation of the C6 carboxyl group using methoxy benzyl alcohol. 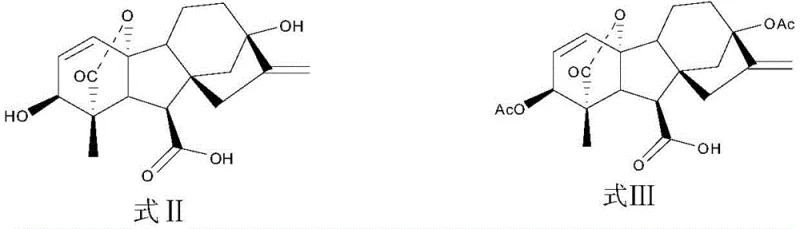 This strategic protection allows for subsequent hydrogenation steps to reduce specific double bonds without affecting other sensitive functionalities. A key breakthrough is the ability to perform the final deprotection and elimination steps in a one-pot fashion or under very mild Lewis acid catalysis at room temperature, completely eliminating the need for dangerous high-temperature reflux. This results in a dramatic improvement in yield, reaching as high as 95% in the final steps, and ensures that the final product possesses a purity of not less than 95%, making it immediately suitable for high-end agrochemical applications without extensive recrystallization.
This strategic protection allows for subsequent hydrogenation steps to reduce specific double bonds without affecting other sensitive functionalities. A key breakthrough is the ability to perform the final deprotection and elimination steps in a one-pot fashion or under very mild Lewis acid catalysis at room temperature, completely eliminating the need for dangerous high-temperature reflux. This results in a dramatic improvement in yield, reaching as high as 95% in the final steps, and ensures that the final product possesses a purity of not less than 95%, making it immediately suitable for high-end agrochemical applications without extensive recrystallization.
Mechanistic Insights into Lewis Acid Catalyzed Hydrolysis and Elimination
The chemical elegance of this synthesis is best exemplified in the final stages where the complex protecting group architecture is dismantled to reveal the active dihydrogibberellin molecule. The mechanism involves a sophisticated interplay between trifluoromethanesulfonic anhydride activation and Lewis acid catalysis. In the critical elimination step, the hydroxyl group at the C3 position is activated, likely forming a transient triflate or similar leaving group species which facilitates the elimination reaction. This is coupled with the removal of the carboxyl protecting groups in a concerted manner, a feat that is chemically challenging due to the steric hindrance and sensitivity of the gibberellin core. The use of Lewis acids such as ferric chloride, zinc chloride, or boron trifluoride is pivotal here, as they coordinate with the oxygen atoms of the protecting groups, lowering the energy barrier for hydrolysis and cleavage. 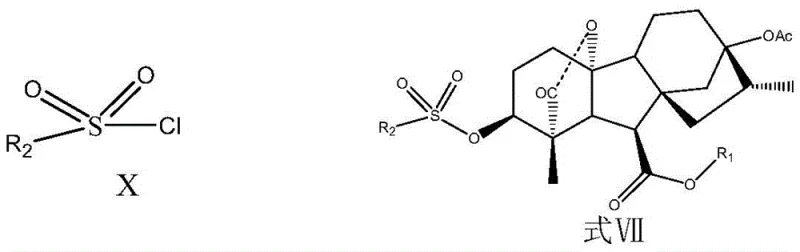 This catalytic activation allows the reaction to proceed rapidly at room temperature, avoiding the thermal degradation pathways that plague non-catalytic thermal hydrolysis. The selectivity of this mechanism ensures that only the intended protecting groups are removed while the newly formed exo-16,17-dihydro structure remains intact, preserving the biological activity of the final plant growth regulator.
This catalytic activation allows the reaction to proceed rapidly at room temperature, avoiding the thermal degradation pathways that plague non-catalytic thermal hydrolysis. The selectivity of this mechanism ensures that only the intended protecting groups are removed while the newly formed exo-16,17-dihydro structure remains intact, preserving the biological activity of the final plant growth regulator.
From an impurity control perspective, the mildness of these reaction conditions is paramount. Harsh basic or acidic conditions at elevated temperatures often lead to skeletal rearrangements or epimerization in gibberellins, generating isomers that are difficult to separate and may possess different biological activities or toxicological profiles. By maintaining the reaction environment at neutral to mildly acidic conditions during the workup and utilizing specific organic bases like DBU or pyridine in controlled stoichiometric ratios, the process minimizes the formation of these deleterious side products. The result is a clean reaction profile where the target dihydrogibberellin is the predominant species, simplifying the purification process to a straightforward column chromatography or crystallization step. This high level of chemical fidelity is essential for meeting the stringent regulatory requirements for agrochemical intermediates, ensuring that the final product delivered to formulators is consistent and reliable.
How to Synthesize Dihydrogibberellin Efficiently
The synthesis of dihydrogibberellin via this patented route is designed for practical implementation in a pilot or commercial plant setting, balancing chemical complexity with operational simplicity. The process begins with the bulk protection of gibberellic acid, a step that can be easily monitored by TLC and quenched safely with methanol. Following the isolation of the fully protected intermediate, the subsequent hydrogenation and selective deacetylation steps utilize standard reagents and solvents like ethyl acetate and methanol, which are familiar to any competent chemical manufacturing team. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature controls that ensure reproducibility.
- Perform peracetylation of gibberellic acid at C3 and C13 positions using acetic anhydride and DMAP to obtain the fully protected intermediate.
- Protect the C6 carboxyl group via benzylation using methoxy benzyl alcohol and EDCI coupling agents.
- Execute catalytic hydrogenation to reduce double bonds at C1-C2 and C16-C17, followed by selective deacetylation and final Lewis acid mediated hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers profound strategic benefits that extend beyond simple chemistry. The primary advantage lies in the drastic reduction of production costs driven by the exceptional yield improvements. Where older methods struggled to achieve 30% yield, this process consistently delivers yields approaching 95% in the critical final steps. This three-fold increase in efficiency means that for every ton of raw gibberellic acid purchased, the output of valuable dihydrogibberellin is significantly higher, effectively lowering the cost of goods sold (COGS) without needing to negotiate lower raw material prices. Furthermore, the elimination of hazardous high-temperature reflux steps reduces the energy consumption of the manufacturing process and lowers the risk of safety incidents, which in turn reduces insurance premiums and operational downtime associated with safety audits and maintenance.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the synthetic route and the high recovery of materials. By reducing the number of steps and avoiding the need for complex purification to remove thermal degradation byproducts, the consumption of solvents and stationary phases for chromatography is substantially decreased. The ability to precipitate intermediates directly from methanol in the early stages eliminates the need for column chromatography at those specific junctures, saving both time and expensive silica gel resources. Additionally, the use of inexpensive and commercially available starting materials like gibberellic acid ensures that the supply chain is not vulnerable to the price volatility of exotic or custom-synthesized precursors, providing a stable cost base for long-term budgeting.
- Enhanced Supply Chain Reliability: The robustness of this synthesis method directly translates to improved supply chain continuity. Because the reaction conditions are mild and the reagents are common industrial chemicals, the risk of batch failure due to equipment limitations or reagent quality variance is minimized. The high yield and purity mean that production schedules can be met with greater certainty, reducing the lead time for high-purity agrochemical intermediates. This reliability is crucial for downstream formulators who depend on a steady supply of active ingredients to meet seasonal agricultural demands. The scalability of the process from gram to kilogram scales without loss of efficiency ensures that suppliers can ramp up production quickly in response to market spikes without compromising on quality.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns perfectly with modern green chemistry principles. The avoidance of strong bases and high-temperature reflux reduces the generation of hazardous waste streams and volatile organic compound (VOC) emissions. The milder conditions also extend the lifespan of reactor vessels and piping by reducing corrosion and thermal stress, contributing to lower capital expenditure on equipment replacement. The high purity of the final product reduces the burden on wastewater treatment facilities, as there are fewer complex organic impurities to degrade. This environmental compatibility makes the process easier to permit and operate in regions with strict environmental regulations, securing the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of dihydrogibberellin synthesized via this advanced method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply chains.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The patented method significantly reduces the synthetic steps compared to literature reports which often require at least 7 steps with yields as low as 30%. This new route achieves yields up to 95% under much milder conditions, avoiding dangerous high-temperature reflux steps required for deprotection in older methods.
Q: Is the starting material gibberellic acid readily available for large-scale production?
A: Yes, the process utilizes commercially available and inexpensive gibberellic acid as the primary raw material. This ensures a stable supply chain foundation and facilitates cost-effective manufacturing of the final dihydrogibberellin product.
Q: What is the purity profile of the final dihydrogibberellin product?
A: The synthesis method is designed to produce high-purity dihydrogibberellin with a purity level not less than 95%. The mild reaction conditions minimize the formation of side products and impurities that typically complicate purification in harsher synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydrogibberellin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern agrochemical landscape. Our team of expert chemists has thoroughly analyzed the technological potential of this batch synthesis method and is fully prepared to implement it for our global partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for plant growth regulators, guaranteeing that every batch of dihydrogibberellin meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your product lines. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates exactly how this optimized route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a competitive advantage in the agrochemical market with a supply of dihydrogibberellin that is both economically superior and technically robust.
