Scalable Synthesis of Optically Active Imidazolones via Metal-Free Asymmetric Conjugate Addition
Introduction to Advanced Asymmetric Synthesis Technology
The landscape of modern organic synthesis is continuously evolving towards more sustainable and efficient methodologies, particularly in the construction of chiral carbon-carbon bonds. A significant breakthrough in this domain is detailed in patent CN114920702A, which discloses a novel method for synthesizing optically active imidazolone compounds via asymmetric conjugate addition. This technology represents a paradigm shift from traditional transition-metal-catalyzed processes to a more benign organocatalytic approach. By utilizing alpha,beta-unsaturated 2-acyl imidazole compounds and organic boronic acids as raw materials, the process achieves high yields and excellent enantioselectivity under mild reaction conditions. The core innovation lies in the use of chiral binaphthol or tetrabenzocyclooctatetraene catalysts combined with molecular sieves, offering a robust platform for generating high-value chiral intermediates essential for pharmaceutical development.
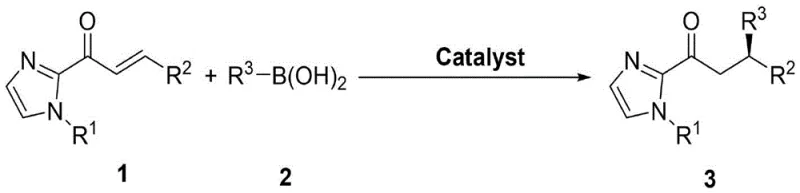
The significance of this invention cannot be overstated for the fine chemical industry. Imidazolone derivatives serve as critical scaffolds in medicinal chemistry, often acting as key precursors for bioactive molecules. The ability to introduce chirality with high precision using simple, non-toxic reagents addresses long-standing challenges in process chemistry. Unlike previous methods that relied on expensive and potentially hazardous metal complexes, this new protocol leverages the inherent stability and low toxicity of organoboron reagents. Furthermore, the operational simplicity—requiring only standard inert atmosphere techniques and common organic solvents—makes this technology immediately accessible for both laboratory research and large-scale manufacturing, positioning it as a vital tool for reliable pharmaceutical intermediate supplier networks seeking to optimize their synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral ketones via conjugate addition has heavily relied on transition metal catalysis, specifically utilizing copper-N-heterocyclic carbene complexes or chiral rhodium complexes under photoredox conditions. While effective, these conventional methods present substantial drawbacks for industrial application. The use of heavy metals such as copper and rhodium introduces significant regulatory hurdles, as residual metal levels in pharmaceutical ingredients must be strictly controlled to parts-per-million levels. Removing these trace metals often requires additional purification steps, such as specialized scavenging resins or extensive recrystallization, which drastically increases production costs and reduces overall process efficiency. Moreover, the catalysts themselves can be air-sensitive, expensive to synthesize, and difficult to recover, leading to higher waste generation and economic inefficiency. The reliance on alkylboranes like alkyl-9-BBN also adds complexity due to their sensitivity and handling requirements, limiting the scope of substrates that can be practically employed in a commercial setting.
The Novel Approach
The methodology described in patent CN114920702A offers a compelling solution to these limitations by employing a metal-free organocatalytic system. This novel approach utilizes chiral binaphthol derivatives or tetrabenzocyclooctatetraene phenols as catalysts, which are not only easier to prepare but also exhibit remarkable stability compared to their metal counterparts. The reaction proceeds smoothly in common organic solvents like toluene at moderate temperatures (typically 60°C), eliminating the need for cryogenic conditions or specialized photochemical equipment. By replacing toxic transition metals with earth-abundant magnesium additives (such as Mg(OtBu)2) and stable organoboronic acids, the process inherently reduces the environmental footprint and safety risks associated with chemical manufacturing. This transition to organocatalysis ensures that the resulting optically active imidazolone compounds are free from heavy metal contamination from the outset, streamlining the downstream purification process and significantly enhancing the economic viability of producing high-purity chiral intermediates for the global market.
Mechanistic Insights into Chiral Binaphthol-Catalyzed Conjugate Addition
The success of this asymmetric transformation hinges on the precise stereochemical control exerted by the chiral catalyst. The proposed mechanism involves a cooperative activation mode where the chiral binaphthol or tetrabenzocyclooctatetraene catalyst interacts with the magnesium additive to form a chiral Lewis acid-base complex. This complex activates the alpha,beta-unsaturated 2-acyl imidazole substrate, likely through coordination with the carbonyl oxygen, thereby increasing its electrophilicity. Simultaneously, the chiral environment created by the bulky naphthyl or phenyl groups of the catalyst directs the approach of the nucleophilic organoboron species. The rigidity of the binaphthyl backbone, as seen in catalysts like Cat 3, ensures a well-defined chiral pocket that discriminates effectively between the two enantiotopic faces of the double bond. This high level of stereocontrol is evidenced by the exceptional enantiomeric ratios (often >95:5 e.r.) observed across a wide range of substrates, demonstrating the robustness of the catalyst design in maintaining chiral integrity throughout the bond-forming event.
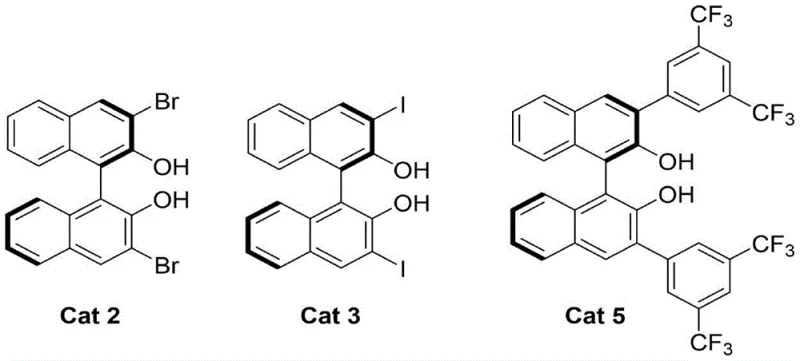
Furthermore, the choice of substituents on the catalyst backbone plays a critical role in tuning the reactivity and selectivity. The patent highlights that catalysts bearing specific halogen or electron-withdrawing groups, such as the iodine-substituted Cat 3 or the trifluoromethyl-substituted Cat 5, can significantly enhance catalytic performance. These electronic modifications likely influence the acidity of the phenolic hydroxyl groups and the steric bulk around the active site, optimizing the transition state energy for the desired enantiomer. The presence of molecular sieves in the reaction mixture is also mechanistically crucial, as they sequester water generated during the transmetallation or activation steps, preventing catalyst deactivation and hydrolysis of the sensitive boronic acid reagents. This careful balance of steric and electronic factors, combined with rigorous moisture control, allows for the consistent production of optically active products with minimal formation of undesired byproducts or racemic impurities, ensuring a clean impurity profile suitable for sensitive pharmaceutical applications.
How to Synthesize Optically Active Imidazolone Derivatives Efficiently
Implementing this synthesis route requires attention to detail regarding reagent quality and reaction conditions to maximize yield and enantioselectivity. The process begins with the preparation of anhydrous reaction vessels, typically Schlenk tubes, which are subjected to vacuum and backfilled with nitrogen to ensure an oxygen-free environment. The specific protocol involves charging the vessel with activated molecular sieves, the optimized chiral catalyst (such as Cat 3 at 10 mol%), and the magnesium additive Mg(OtBu)2. To this mixture, the alpha,beta-unsaturated 2-acyl imidazole substrate and the organoboronic acid coupling partner are added in a stoichiometric ratio, typically 1:2, dissolved in dry toluene. The reaction is then heated to 60°C and stirred for approximately 48 hours. Monitoring via TLC is recommended to confirm the complete consumption of the starting material before proceeding to workup. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining alpha,beta-unsaturated 2-acyl imidazole, organoboronic acid, chiral catalyst (e.g., Cat 3), and Mg(OtBu)2 in anhydrous toluene with molecular sieves.
- Stir the reaction under nitrogen atmosphere at 60°C for approximately 48 hours until TLC indicates complete consumption of the starting material.
- Remove the solvent under reduced pressure and purify the crude residue via flash silica gel column chromatography to isolate the optically active imidazolone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers tangible strategic benefits beyond mere chemical elegance. The shift away from precious metal catalysts directly translates to cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for expensive rhodium or palladium complexes and the associated metal scavenging agents, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the use of commercially available and stable organoboronic acids as coupling partners ensures a reliable supply chain, as these reagents are produced on a multi-ton scale globally and are not subject to the same geopolitical supply constraints as rare earth metals. This stability in raw material sourcing mitigates the risk of production delays and price volatility, allowing for more accurate long-term budgeting and inventory planning.
- Cost Reduction in Manufacturing: The elimination of transition metals removes the necessity for costly purification steps dedicated to heavy metal removal, which are often required to meet strict regulatory limits for API intermediates. This simplification of the downstream processing workflow reduces solvent consumption, waste disposal costs, and labor hours, leading to substantial operational savings. Furthermore, the catalysts used are organic small molecules that are generally cheaper to synthesize and can potentially be recovered and reused, further enhancing the economic efficiency of the process compared to single-use metal catalysts.
- Enhanced Supply Chain Reliability: The reliance on robust, shelf-stable reagents like aryl boronic acids and chiral diols ensures consistent batch-to-batch quality and availability. Unlike air-sensitive organometallic reagents that require special storage and handling protocols, the materials in this process are forgiving and easy to transport, reducing logistical complexities. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream customers, thereby strengthening the supplier-client relationship and reducing the risk of supply chain disruptions.
- Scalability and Environmental Compliance: The mild reaction conditions (60°C, atmospheric pressure equivalent) and the use of common solvents like toluene make this process highly scalable from gram to kilogram and ton scales without requiring specialized high-pressure or cryogenic reactors. The metal-free nature of the reaction also aligns with green chemistry principles, reducing the generation of hazardous heavy metal waste. This facilitates easier compliance with increasingly stringent environmental regulations and simplifies the permitting process for manufacturing facilities, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric conjugate addition technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific target molecules and for procurement teams assessing the supply chain implications.
Q: What are the key advantages of this metal-free catalytic system?
A: The primary advantage is the elimination of toxic transition metals like copper or rhodium, which simplifies purification and ensures the final product meets stringent pharmaceutical heavy metal limits without expensive scavenging steps.
Q: What is the typical enantioselectivity achieved with this method?
A: The method consistently achieves high enantiomeric excess (ee), often exceeding 90% and reaching up to 96% ee depending on the specific substrate and catalyst combination used.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction utilizes readily available organoboronic acids and robust chiral diol catalysts under mild conditions (60°C), making it highly suitable for scale-up in commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Imidazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric conjugate addition technology disclosed in patent CN114920702A for the production of high-value chiral building blocks. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, guaranteeing that every batch of optically active imidazolone compounds meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this metal-free route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global market with reliable, high-purity chemical solutions.
